
 ��2013?�����ģ�⣩��������������Ȼ��ƣ�Ϊ�ⶨij������Ʒ��̼���Ƶĺ�����С����ȡ�ô�����Ʒ3.3g������ܽ���ˮ�У��ٵμ��Ȼ�����Һ����������������������Ȼ�����Һ��������ϵ��ͼ��ʾ��������������С�����һλ������
��2013?�����ģ�⣩��������������Ȼ��ƣ�Ϊ�ⶨij������Ʒ��̼���Ƶĺ�����С����ȡ�ô�����Ʒ3.3g������ܽ���ˮ�У��ٵμ��Ȼ�����Һ����������������������Ȼ�����Һ��������ϵ��ͼ��ʾ��������������С�����һλ������| 106 |
| x |
| 111 |
| y |
| 100 |
| 3.0g |
| 3.18g |
| 3.3g |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| 1H �� |
2He �� | ||||||
| 3Li � |
4Be �� |
5B �� |
6C ̿ |
7N �� |
8O �� |
9F �� |
10Ne �� |
| 11Na �� |
12Mg þ |
13Al �� |
14Si �� |
15P �� |
16S �� |
17Cl �� |
18Ar � |
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
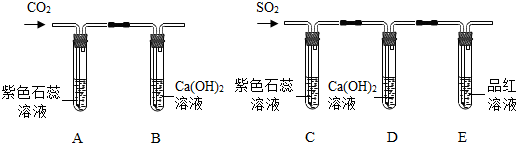
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com