
æĘѧ¼ŅĢį³ö¹¤ŅµÉś²ś»ŲŹÕĄūÓĆæÕĘųÖŠCO2µÄ¹¹Ļė£ŗ°ŃæÕĘų“µČė±„ŗĶĢ¼ĖįÄĘČÜŅŗÖŠ£¬ČÜŅŗæÉĪüŹÕCO2Éś³ÉĢ¼ĖįĒāÄĘ£»¼ÓČČĢ¼ĖįĒāÄĘ¹ĢĢåÓÖ·Ö½ā·Å³öCO2£»ŌŚŗĻ³ÉĖžÄŚCO2ÓėĒāĘų·“Ӧɜ³É¼×“¼£ØCH3OH£©ŗĶĖ®”£¼ņŅŖµÄÉś²śĮ÷³ĢČēĻĀĶ¼ĖłŹ¾£ŗ
[׏ĮĻæØʬ] Ģ¼ĖįĒāÄĘ·Ö½āĪĀ¶ČŹĒ270”ę£¬Ģ¼ĖįÄĘŌŚ856”ęČŪ»Æ£¬µ«»¹Ī““ļµ½·Ö½āĪĀ¶Č”£
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ĪüŹÕ³ŲÖŠ·¢ÉśĮĖ»ÆŗĻ·“Ó¦£¬»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø2£©»ŲŹÕĄūÓĆæÕĘųÖŠµÄCO2£¬½µµĶ“óĘųÖŠCO2ÅØ¶Č£¬ÓŠĄūÓŚ¼õ»ŗ ”£
£Ø3£©ŗĻ³ÉĖžÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø4£©øĆĮ÷³ĢÖŠÄÜŃ»·ĄūÓƵÄĪļÖŹŹĒ £ØĢī»ÆѧŹ½£©”£
£Ø1£©Na2CO3+2HCl==2NaCl+H2O+CO2”ü£Ø2·Ö£©£Ø2£©ĪĀŹŅŠ§Ó¦£Ø1·Ö£©
£Ø3£©CO2+3H2 CH3OH+H2O£Ø2·Ö£©
CH3OH+H2O£Ø2·Ö£©
£Ø4£© Na2CO3”¢H2O£Ø2·Ö£¬Ćæ»Ų“šŅ»ÖÖ£¬¼“øųŅ»·Ö£©
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©“ÓĶ¼ÖŠæÉÖŖ£ŗŌŚĪüŹÕ³ŲÖŠĢ¼ĖįÄĘÓėæÕĘųÖŠµÄ¶žŃõ»ÆĢ¼·¢ÉśĮĖ·“Ó¦£¬Éś³ÉĢ¼ĖįĒāÄĘ”£øł¾ŻĢ¼ĖįĒāÄʵÄ×é³ÉŌŖĖŲæÉÖŖ£¬·“Ó¦Īļ»¹Ó¦ÓŠŗ¬ĒāŌŖĖŲµÄĪļÖŹĖ®”£ĖłŅŌ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖNa2CO3+2HCl==2NaCl+H2O+CO2”ü”£
£Ø2£©¶žŃõ»ÆĢ¼ŹĒŌģ³ÉĪĀŹŅŠ§Ó¦µÄÖ÷ŅŖĘųĢ壬ĖłŅŌ»ŲŹÕĄūÓĆæÕĘųÖŠµÄCO2£¬½µµĶ“óĘųÖŠCO2ÅØ¶Č£¬ÓŠĄūÓŚ¼õ»ŗĪĀŹŅŠ§Ó¦”£
£Ø3£©øł¾ŻĢāŅāæÉÖŖ£ŗŌŚŗĻ³ÉĖžÄŚ·¢Éś·“Ó¦µÄ·“Ó¦ĪļĪŖCO2ÓėĒāĘų£»·“Ó¦Ģõ¼žĪŖ £»Éś³ÉĪļĪŖ¼×“¼£ØCH3OH£©ŗĶĖ®”£
£»Éś³ÉĪļĪŖ¼×“¼£ØCH3OH£©ŗĶĖ®”£
£Ø4£©øĆĮ÷³ĢµÄÄæµÄŹĒ¹¤ŅµÉś»ŲŹÕĄūÓĆæÕĘųÖŠCO2£¬ĖłŅŌÄÜŃ»·ĄūÓƵÄĪļÖŹŹĒNa2CO3ŗĶH2O”£
æ¼µć£ŗĢ¼ĖįÄĘÓėĢ¼ĖįĒāÄʵĊŌÖŹ”¢»Æѧ·½³ĢŹ½µÄŹéŠ“


 ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø
ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
ijŠĖȤŠ”×éµÄĶ¬Ń§ĪŖĢ½¾æ½šŹōµÄ»ī¶ÆŠŌ”¢ŅŌ¼°ĶŠāŹ“µÄĢõ¼ž½ųŠŠĮĖŅŌĻĀĻµĮŠ
ŹµŃ飬׊Ļø·ÖĪöŗó»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ŹµŃéŅ»£ŗŠ”Ć÷°“ĻĀĶ¼1ĖłŹ¾ŅŌ”°½šŹō±ķĆę²śÉśĘųÅŻµÄæģĀż”±ĪŖ±ź×¼Ą“ÅŠ¶ĻŠæ”¢ĀĮ”¢Ģś
µÄ»ī¶ÆŠŌĒæČõ£¬ÓŠĶ¬Ń§ČĻĪŖ²»¹»ŗĻĄķ£¬ĄķÓÉŹĒ________________________________”£ĀĮÓė
Ļ”ŃĪĖįøÕ½Ó“„Ź±·“Ó¦»ŗĀż£¬Ņ»¶ĪŹ±¼äŗó¾ēĮŅ£¬²śÉśĘųÅŻĖŁĀŹĖę׿Óæģ£¬ĘäŌŅņæÉÄÜŹĒ
_________________________________________ӣ
£Ø2£©ŹµŃ鶞£ŗĪŖĢ½¾æĢś”¢Ķ”¢ŅųČżÖÖ½šŹō»ī¶ÆŠŌµÄĖ³Šņ£¬Š”ŗģĢį³ö°“ÉĻĶ¼2½ųŠŠĮ½²½ŹµŃ飬
¼“æÉÖ¤Ć÷ČżÖÖ½šŹō»ī¶ÆŠŌĖ³Šņ”£ÄćČĻĪŖĘä·½°øæÉŠŠĀš£æ_______£ØĢī”°æÉŠŠ”±»ņ”°²»æÉŠŠ”±£© ”£
a. ŹŌ¹Ü¢ŁÖŠĻÖĻóŹĒ________________________________________________”£
b. ŹŌ¹Ü¢ŚÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ________________________________________”£
£Ø3£©ŹµŃéČż£ŗŠ”·¼”¢Š”ĮĮĮ½Ī»Ķ¬Ń§·¢ĻÖĶÖŹŃŪ¾µ¼ÜÉĻ³öĻÖĮĖŅ»Š©ĀĢÉ«µÄŠā×Õ”£¾²éŌÄ׏ĮĻµĆÖŖ£¬ĶŠāµÄ»Æѧ³É·ÖŹĒ¼īŹ½Ģ¼ĖįĶ ”£ĖūĆĒŅĄÕÕøÖĢśÉśŠāµÄĢ½¾æ·½·ØÉč¼ĘČēĻĀŹµŃé£ØĻĀĶ¼3ĖłŹ¾£©”£Ēė»Ų“š£ŗ
”£ĖūĆĒŅĄÕÕøÖĢśÉśŠāµÄĢ½¾æ·½·ØÉč¼ĘČēĻĀŹµŃé£ØĻĀĶ¼3ĖłŹ¾£©”£Ēė»Ų“š£ŗ
a. ĶĘ²āŅ»¶ĪŹ±¼äŗó»į³öĻÖĶŠāµÄŹĒ________ŹŌ¹ÜÄŚµÄĶʬ”££ØĢī”°¢Ł”±”¢”°¢Ś”±»ņ”°¢Ū”±£©
b. Š”·¼Ķ¬Ń§ČĻĪŖŅŖÅŖĒåµ¼ÖĀĶŠāŹ“µÄŅņĖŲ£¬»¹Ó¦øĆ²¹³äŹµŃ飬ČēÉĻĶ¼4ĖłŹ¾µÄŹµŃ飬ĘäÄæ
µÄŹĒ________________________________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
µĖ®×ŹŌ“µÄȱ·¦ŅŃ³ÉĪŖÖĘŌ¼Éē»į·¢Õ¹µÄÖŲŅŖŅņĖŲ£¬“ÓŗĘ嫵Äŗ£ŃóĄļ»ńČ”µĖ®£¬¶Ō½ā¾öµĖ®Ī£»ś¾ßÓŠÖŲŅŖŅāŅ唣ijŠ”×éÕė¶ŌÕāŅ»ĻÖדĢį³öĪŹĢā£¬²¢ŌŚŹµŃéŹŅ½ųŠŠĮĖŅŌĻĀĢ½¾æ”£
”¾Ģį³öĪŹĢā”æ ”£
”¾ŹµŃéÓĆĘ·”æ¾Ę¾«µĘ”¢ŹŌ¹Ü”¢µ„æ×ĻšĘ¤Čū”¢²£Į§µ¼¹Ü”¢Ģś¼ÜĢØ”¢ÉÕ±”¢½ŗĶ·µĪ¹Ü”¢ŗ£Ė®”¢ĻõĖįŅųČÜŅŗ”£
”¾ŹµŃé¹ż³Ģ”æ
¢Ł°“ČēĶ¼ĖłŹ¾×é×°ŅĒĘ÷²¢¼ģ²é×°ÖĆĘųĆÜŠŌ”£
¢ŚĻņ“óŹŌ¹ÜÖŠ¼ÓČė5 mL”«10 mLŗ£Ė®£¬ČūŗĆĻšĘ¤Čū£¬ÓĆ¾Ę¾«µĘ¼ÓČČ”£¼ÓČČŅ»¶ĪŹ±¼äŗ󣬊”ŹŌ¹ÜÄŚŹÕ¼Æµ½ŗÜÉŁŅŗĢ壬ŹŌ¹ÜæŚÓŠ“óĮæ°×Īķ”£
øĆŠ”×éĶ¬Ń§½ØŅé¶ŌøĆ×°ÖĆ½ųŠŠøĽų£¬ÄćČĻĪŖøĽųµÄ“ėŹ©ĪŖ£ŗ ”£
¢ŪĻņŠ”ŹŌ¹ÜĄļµÄĄäÄżĖ®ÖŠµĪ¼Ó¼øµĪĻõĖįŅųČÜŅŗ£¬ĻÖĻóĪŖ£ŗ ”£
”¾µĆ³ö½įĀŪ”æ
ĶعżÉĻŹöŹµŃéĢ½¾æ£¬ÄćµĆ³öµÄ½įĀŪŹĒ£ŗ ”£
”¾·“Ė¼½»Į÷”æ
¢Ł¶Ōŗ£Ė®µ»ÆŗóŹ£Óą×ŹŌ“£¬ĒėĢį³öŅ»ÖÖŗĻĄķĄūÓƵēėŹ©£ŗ ”£
¢Ś”°ÕōĮó·Øµ»Æŗ£Ė®”±µÄĖ¼Ā·ŹĒĻė°ģ·Ø½«Ė®“Ó»ģŗĻĪļÖŠ·ÖĄė³öĄ“”£ĒėĮŠ¾ŁŅ»ÖÖÓėÉĻŹöŌĄķĻąĶ¬µÄ»ģŗĻĪļ·ÖĄėµÄĄż×Ó£ŗ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
ѧĻ°ĒāŃõ»ÆÄʵĊŌÖŹÖ®ŗó£¬ÖŖµĄĒāŃõ»ÆÄĘČÜŅŗÖŠŗ¬ÓŠH2O”¢Na+”¢OH”Ŗ£¬ÄÜŹ¹ĪŽÉ«·ÓĢŖŹŌŅŗ±äŗģÉ«£¬Ä³æĘ¼¼Š”×éĻė½ųŅ»²½Ģ½¾æŹĒÄÄŅ»ÖÖĮ£×ÓŹ¹ĪŽÉ«·ÓĢŖŹŌŅŗ±äŗģÉ«”£
”¾Ģį³ö²ĀĻė”æ²ĀĻė¢Ł£ŗæÉÄÜŹĒH2O£» ²ĀĻė¢Ś£ŗæÉÄÜŹĒNa+£» ²ĀĻė¢Ū£ŗæÉÄÜŹĒOH”Ŗ”£
”¾ŹµŃéŃéÖ¤”æ
| Źµ Ńé ²Ł ×÷ | Źµ Ńé ĻÖ Ļó | ½į ĀŪ |
| ¢ŁÓĆŹŌ¹ÜȔɣĮæÕōĮóĖ®£¬µĪČė1-2µĪĪŽÉ«·ÓĢŖŹŌŅŗ£¬Õńµ“ | ŹŌ¹ÜÄŚČÜŅŗČŌĪŖĪŽÉ« | Ė®·Ö×Ó²»ÄÜŹ¹ĪŽÉ«·ÓĢŖŹŌŅŗ±äŗģÉ« |
| ¢ŚÓĆŹŌ¹ÜȔɣĮæĀČ»ÆÄĘČÜŅŗ£¬µĪČė1-2µĪĪŽÉ«·ÓĢŖŹŌŅŗ£¬Õńµ“ | | |
| ¢ŪÓĆŹŌ¹ÜȔɣĮæĒāŃõ»ÆÄĘČÜŅŗ£¬µĪČė1-2µĪĪŽÉ«·ÓĢŖŹŌŅŗ£¬Õńµ“ | | |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
³õÖŠ»ÆѧæĪÉĻ£¬Ķ¬Ń§ĆĒĄūÓĆĻĀĮŠŹµŃéĢ½¾æ¼īµÄ»ÆѧŠŌÖŹ£ŗ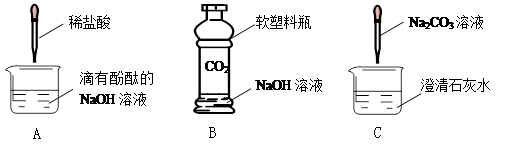
1£©AŹµŃéÖŠ¹Ū²ģµ½µÄĻÖĻóĪŖ £¬ŌŅņŹĒ£ØÓĆ»Æѧ·½³ĢŹ½±ķŹ¾£© ”£
2£©BŹµŃéÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
3£©CŹµŃéÖŠ¹Ū²ģµ½µÄĻÖĻóŹĒ ”£
4£©ŹµŃé½įŹųŗó£¬Ķ¬Ń§ĆĒ½«·ĻŅŗµ¹ČėĶ¬Ņ»Ö»·ĻŅŗø×ÖŠ£¬×īŗó·¢ĻÖ·ĻŅŗ»ė×Ē²¢ĻŌŗģÉ«”£
”¾Ģį³öĪŹĢā”æ ·ĻŅŗÖŠŗ¬ÓŠŹ²Ć“ĪļÖŹ£æ
”¾¼ŁÉč²ĀĻė”æ Ķعż·ÖĪöÉĻŹöŹµŃ飬Ķ¬Ń§ĆĒ²ĀĻė£ŗ·ĻŅŗÖŠ³ż·ÓĢŖ”¢Ģ¼ĖįøĘ¼°Ė®ŅŌĶāŅ»¶Øŗ¬ÓŠ ”£
ĄĻŹ¦ĪŹ£ŗ·ĻŅŗÖŠ»¹æÉÄÜÓŠŹ²Ć“ĪļÖŹ£æŅż·¢Ķ¬Ń§ĆĒČČĮŅĢÖĀŪ£¬ĻĀĆęŹĒ“ó¼ŅµÄ·¢ŃŌ£ŗ
| ѧɜ¼× | æÉÄÜÓŠCa(OH)2”¢NaOH | ѧɜ±ū | æÉÄÜÓŠCa(OH)2”¢CaCl2 |
| ѧɜŅŅ | æÉÄÜÓŠNaOH”¢Na2CO3 | ѧɜ¶” | æÉÄÜÓŠNa2CO3 |
| ѧɜĪģ | æÉÄÜÓŠNaOH | ”” | ”””” |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
ŹŠ³”ÉĻµÄŹķʬ”¢ĻŗĢõµČŹ³Ę·Ņ»°ć²ÉÓĆ³äĘų°ü×°£¬ĖüŹ¹ÓƵďĒŹ²Ć“ĘųĢåÄŲ£æŠ”Ć÷ŗĶŠ”ŗģ¶ŌŹŠ³”ÉĻĻśŹŪµÄŅ»ÖÖĻŗĢõ·¢ÉśĮĖŠĖȤ£¬ŅņĪŖĻŗĢõ°ü×°ŌŚ³äĀśĘųĢåµÄŠ”ĖÜĮĻ“üÄŚ£¬“üÄŚµÄĘųĢå³åµĆ¹Ä¹ÄµÄ”£ĖūĆĒČĻĪŖÕāÖÖ³äĘų°ü×°¼¼Źõ£¬Ö÷ŅŖŹĒĪŖĮĖŹ¹Ź³Ę·Äܹ»½Ļ³¤Ź±¼äµÄ±£ĻŹ”¢±£ÖŹ”£ÄĒĆ“£¬ÕāŹĒŹ²Ć“ĘųĢåÄŲ£æ
Š”ŗģ²ĀĻėŹĒµŖĘų£¬Š”Ć÷²ĀĻėŹĒ¶žŃõ»ÆĢ¼”£
£Ø1£©ĒėÄć°ļÖśĖūĆĒÉč¼ĘŅ»øö¼ņµ„µÄŹµŃé·½°ø£¬Ą“ÅŠ¶ĻŠ”Ć÷µÄ²ĀĻėŹĒ·ńÕżČ·£¬¼ņŅŖŠ“³ö²Ł×÷²½Öč£ŗ £¬
£Ø2£©Š”Ć÷µÄ²ĀĻėČē¹ūÕżČ·£¬ĻÖĻóÓ¦ŹĒ ”£
£Ø3£©ÄćČĻĪŖŹ³Ę·³äĘų°ü×°£¬¶ŌĖł³äĘųĢåµÄŅŖĒóŹĒ£ŗ £ØŠ“Ņ»µć£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
ĻĀĆęŹĒijŠĖȤŠ”×éĶ¬Ń§×öµÄ2øöŠ”ŹµŃ锣øł¾ŻŹµŃéÄŚČŻ»Ų“šĻĀĮŠĪŹĢā£ØŅĒĘ÷µÄ¹Ģ¶Ø×°ÖĆŅŃ¾Ź”ĀŌ)”£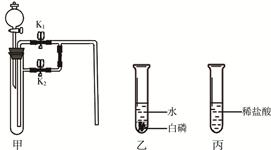
£Ø1£©Čō¼×ÖŠŹ¢ÓŠ¶žŃõ»ÆĆĢ£¬Į¬½Ó¼×ŅŅ×°ÖĆ£¬¹Ų±ÕK1“ņæŖK2£¬“Ó·ÖŅŗĀ©¶·Ļņ¼×ÖŠ¼ÓČėŹŹĮæĖ«ŃõĖ®£¬¹Ų±Õ·ÖŅŗĀ©¶·»īČū£¬¹Ū²ģµ½ŅŅÖŠµ¼¹ÜæŚÓŠĘųÅŻĆ°³ö£¬°×Įײ»Č¼ÉÕ£»¹Ų±ÕK2“ņæŖK1£¬ŌņŅŅÖŠµÄĻÖĻóŹĒ £¬ĒŅ°×Į×Č¼ÉÕ”£
¢Ł¼×ÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ ”£
¢ŚŹµŃé¹ż³ĢÖŠ£¬°×ĮײśÉśĻÖĻó²īŅģµÄŌŅņŹĒ ”£
£Ø2£©Čō¼×ÖŠ³äĀś¶žŃõ»ÆĢ¼£ØK1”¢K2¹Ų±Õ£©£¬Į¬½Ó¼×±ū£¬“Ó·ÖŅŗĀ©¶·Ļņ¼×ÖŠ¼ÓČėŹŹĮæµÄ³ĪĒåŹÆ»ŅĖ®£¬¹Ų±Õ·ÖŅŗĀ©¶·»īČū£¬Ņ»¶ĪŹ±¼äŗ󣬓ņæŖK1£¬ÕūøöŹµŃé¹ż³ĢÖŠ¹Ū²ģµ½¼×ÖŠµÄĻÖĻóŹĒ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
Ä³ŃŠ¾æŠ”×é·¢ĻÖ£¬Ī¬CÅŻĢŚĘ¬£Ø±£½”Ņ©Ę·£¬Ö÷ŅŖ³É·Ö¼ūĶ¼1£©ČÜÓŚĖ®£¬ÓŠŠķ¶ąĘųÅŻ²śÉś£ØČēĶ¼2£©”£øĆŠ”×éĶ¬Ń§Ģ½¾æøĆĘųĢåµÄ³É·Ö”£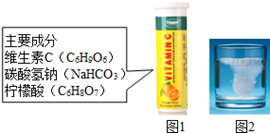
”¾²ĀĻėÓė¼ŁÉč”æ
Š”»ŖĖµ£ŗøĆĘųĢåæÉÄÜŹĒCO2”¢O2”¢CO”¢H2”¢N2”£
Š”Ć÷Ėµ£ŗ²»æÉÄÜŗ¬ÓŠN2£¬ŅņĪŖ ”£
Š”·¼Ėµ£ŗ²»æÉÄÜŗ¬ÓŠCOŗĶH2£¬ŅņĪŖ“ÓŅ©Ę·°²Č«½Ē¶Čæ¼ĀĒ£¬H2”¢COŅ×Č¼Ņ×±¬£¬Ķ¬Ź±COŅ²”” ”””£
øĆŠ”×éĶ¬Ń§ČĻĪŖ£ŗøĆĘųĢåæÉÄÜŗ¬ÓŠCO2”¢O2ÖŠµÄŅ»ÖÖ»ņĮ½ÖÖ”£
”¾½ųŠŠŹµŃé”æ
| ŹµŃ鱹ŗÅ | ŹµŃé²Ł×÷ | ŹµŃéĻÖĻó |
| ¢Ł | ½«ĘųĢåĶØČė³ĪĒåµÄŹÆ»ŅĖ®ÖŠ | ³ĪĒåŹÆ»ŅĖ®±ä»ė×Ē |
| ¢Ś | ½«“ų»šŠĒµÄľĢõÉģČėøĆĘųĢåÖŠ | “ų»šŠĒµÄľĢõƻӊø“Č¼ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗ³õÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢ½¾æĢā
»ÆѧæĪŗ󣬻ÆѧŠĖȤŠ”×éµÄĶ¬Ń§ŌŚÕūĄķŹµŃéץŹ±£¬·¢ĻÖÓŠŅ»ĘæĒāŃõ»ÆÄĘČÜŅŗƻӊČūĻšĘ¤Čū£¬Õ÷µĆĄĻŹ¦Ķ¬Ņāŗó£¬æŖÕ¹ĮĖŅŌĻĀĢ½¾æ£ŗ
[Ģį³öĪŹĢā1]øĆĒāŃõ»ÆÄĘČÜŅŗŹĒ·ń±äÖŹĮĖÄŲ£æ
[ŹµŃéĢ½¾æ1]
| ŹµŃé²Ł×÷ | ŹµŃéĻÖĻó | ŹµŃé½įĀŪ |
| ȔɣĮæøĆČÜŅŗÓŚŹŌ¹ÜÖŠ£¬ĻņČÜŅŗÖŠµĪ¼ÓĻ”ŃĪĖį£¬²¢²»¶ĻÕńµ“£® | ÓŠĘųÅŻĆ°³ö£® | ĒāŃõ»ÆÄĘČÜŅŗŅ»¶Ø±äÖŹĮĖ£® |
| ŹµŃé²½Öč | ŹµŃéĻÖĻó | ŹµŃé½įĀŪ |
| £Ø1£©Č”ÉŁĮæøĆČÜŅŗÓŚŹŌ¹ÜÖŠ£¬ĻņČÜŅŗÖŠµĪ¼Ó¹żĮæµÄĀČ»ÆøĘČÜŅŗ£¬²¢²»¶ĻÕńµ“£® | ÓŠ”” ””Éś³É£® | ĖµĆ÷ŌČÜŅŗÖŠŅ»¶ØÓŠĢ¼ĖįÄĘ£® |
| £Ø2£©Č”²½Öč£Ø1£©ŹŌ¹ÜÖŠµÄÉŁĮæÉĻ²ćĒåŅŗ£¬µĪ¼Ó·ÓĢŖČÜŅŗ£® | ČÜŅŗ±äŗģÉ«£® | ĖµĆ÷ŌČÜŅŗÖŠŅ»¶ØÓŠ”” ””£® |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com