������ʯ�����Ʋ�Ʒ֮һ������������Ҫȼ�ϣ���ش�
�����ͳ��ȼ�յIJ����Ƕ�����̼��ˮ���ɴ˿��ƶ����͵����Ԫ����һ������ Ԫ�أ�
��Ŀǰ��������β����Ⱦ�Ĵ�ʩ֮һ�����������м����������Ҵ����Ҵ���ȫȼ�յĻ�ѧ����ʽΪ ��
������β���к���CO��NO�������������ʶԴ�����Ⱦ�ķ���������������������װһ������ת��������ʹ���Ƿ�����Ӧ�������ֲ������ѭ�������壬������������ �� ��
���𰸡�
���������ݷ�Ӧ�������������ж����ʵ�Ԫ����ɣ����ݷ�Ӧ��������P�������غ㶨�ɿ�����д��ѧ����ʽ������CO��NO��������Ԫ�غʹ����еij������������
����⣺��1����Ӧǰ��Ԫ�ص�����䣬�����������̼��ˮ�е�̼Ԫ�غ���Ԫ���������ͣ����̼���⣮
��2���Ҵ���ȫȼ������ˮ�Ͷ�����̼��ע���Ҵ��Ļ�ѧʽ����ƽ����Ӧ�Ļ�ѧ����ʽΪ��C
2H
5OH+3O
2
2CO
2+3H
2O��
��3����֪һ����̼��һ�������ڴ��������������������ֲ������ѭ�������壬���ݻ�ѧ��Ӧǰ��Ԫ�������Ĺ��ɣ���Ӧ���к���̼������������Ԫ�أ�������Ԫ������ɴ����еĶ�����̼�͵��������������̼��������
�����������Ҫ���������غ㶨�ɺͽ��ܻ����ȷ�������ݣ�ֻ���������ܶ���ط��������������ȷ���жϣ�

 2CO2+3H2O��
2CO2+3H2O��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
 �й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
�й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����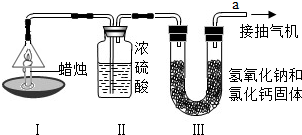
 �й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
�й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
 �й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
�й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����