
��10��ɽ23����8�֣�A-G�dz��г��������ʡ�����EΪ��ɫ���塣��ͼΪ��Щ���ʵ�ת����ϵͼ�����в���������ͷ�Ӧ������ʡ�ԡ�ȫƷ�п���
��1��д���������ʵĻ�ѧʽ��E ��G ��
��2����ѧ��Ӧ�����������仯��B��ˮ��Ӧʱ�� �������ա��ų�����������
��3��ʵ���Ҳ���A��ϡ���ᷴӦ����ȡC��ԭ����
��
��4��д��D��F�Ļ�ѧ����ʽ ��
��5������Cu��O2��H2SO4��ԭ���Ƶ�CuSO4����Ӧ�Ĺ����ǣ�
�������ֱ�������
 ÿ��10���ӿ�����������������ϵ�д�
ÿ��10���ӿ�����������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��10��ɽ23����8�֣�A-G�dz��г��������ʡ�����EΪ��ɫ���塣��ͼΪ��Щ���ʵ�ת����ϵͼ�����в���������ͷ�Ӧ������ʡ�ԡ�ȫƷ�п���

��1��д���������ʵĻ�ѧʽ��E ��G ��
��2����ѧ��Ӧ�����������仯��B��ˮ��Ӧʱ�� �������ա��ų�����������
��3��ʵ���Ҳ���A��ϡ���ᷴӦ����ȡC��ԭ����
��
��4��д��D��F�Ļ�ѧ����ʽ ��
��5������Cu��O2��H2SO4��ԭ���Ƶ�CuSO4����Ӧ�Ĺ����ǣ�
�������ֱ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��ȫ���п�����ר����ר��������ʵļ��𡢼������ƶ�4 ���ͣ������
��10��ɽ23����8�֣�A-G�dz��г��������ʡ�����EΪ��ɫ���塣��ͼΪ��Щ���ʵ�ת����ϵͼ�����в���������ͷ�Ӧ������ʡ�ԡ�ȫƷ�п���
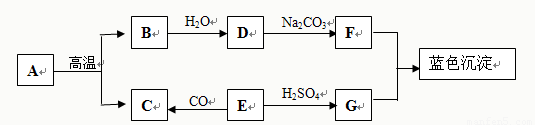
��1��д���������ʵĻ�ѧʽ��E ��G ��
��2����ѧ��Ӧ�����������仯��B��ˮ��Ӧʱ�� �������ա��ų�����������
��3��ʵ���Ҳ���A��ϡ���ᷴӦ����ȡC��ԭ����
��
��4��д��D��F�Ļ�ѧ����ʽ ��
��5������Cu��O2��H2SO4��ԭ���Ƶ�CuSO4����Ӧ�Ĺ����ǣ�
�������ֱ�������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com