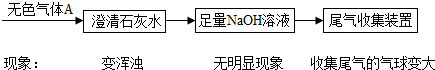
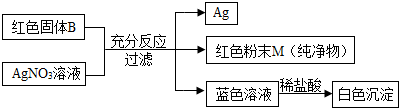
���أ�
ij��ѧ��ȤС����һ�λ�������ǣ���һ������ʯ��ĩ����ɽ���̽����������벢�ش��й����⣮
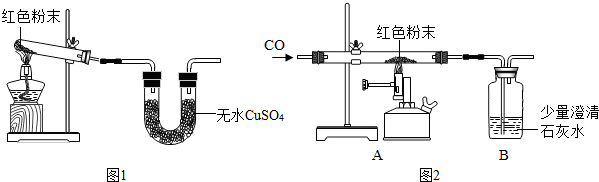
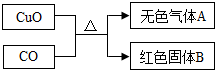
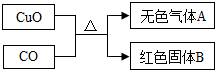
[��ʦ����]����������FeCO
3����������Fe
2O
3����������Fe
3O
4�����ֹ����ĩ�е�һ�ֻ�������ɣ�
[�������]������ĩ��Ҫ�ɷֿ����У���ֻ��FeCO
3����ֻ��Fe
2O
3����ֻ��Fe
3O
4����FeCO
3��Fe
2O
3����FeCO
3��Fe
3O
4����Fe
2O
3��Fe
3O
4��
[��������]��1��FeCO
3�����ֽ�FeCO
3FeO+CO
2��������������ᷴӦ�ų�CO
2��FeCO
3+2HCl=FeCl
2+CO
2��+H
2O��
��2������ʯ��ĩ�е����ʲ��μӷ�Ӧ��
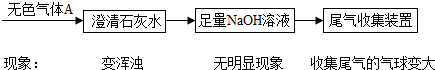
[̽��˼·]���������ʵ����ʲ�ͬ���ʵ��̽����ɷ֣�Ȼ����ѡ���ʵ����Լ��ͷ���ͨ��ʵ��ⶨ�京������װ���ڿ����е�ˮ������CO
2���Բ��ƣ�
[ʵ��̽��]
��1��ȡ����������ĩװ���Թ��У��������������ᣬû�����ݲ������Ӷ��ų������е�
�٢ܢ�
�٢ܢ�
��������е���ţ���
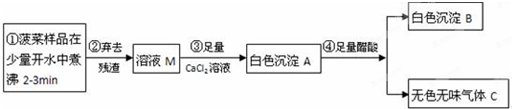
��2��Ϊ��һ��ȷ��������ĩ����ɣ���ȡ�÷�ĩ10.0gװ��Ӳ�ʲ������У���ͨ�������ͼ����ʵ�飮

��ʼʱ����ͨ��CO���壬��һ��ʱ����ټ���ʹ���ַ�Ӧ������Ӧ��ȫ��ֹͣ���ȣ��Լ���ͨCO����ֱ����������ȴ����Ӧǰ��������װ�ú����ʵ������������������±���
|
��Ӧǰ |
��Ӧ�� |
| a�� |
�����ܺ�������ĩ��������Ϊ70.0g |
�����ܺ������ʵ�������Ϊ67.6g |
| bƿ |
ϴ��ƿ����ʢ��Һ��������Ϊ200.0g |
ϴ��ƿ��ƿ�����ʵ�������Ϊ206.6g |
[��������]
��3��Ϊ�˷�ֹ��Һ��������ʵ�����ֹͣ����ǰ�Ƿ���Ҫ�ȶϿ�װ����a��b�����Ӵ���
����Ҫ
����Ҫ
������Ҫ����Ҫ����������
��Ϊһֱͨ��CO���壬B����Һ���ᵹ����Aװ����
��Ϊһֱͨ��CO���壬B����Һ���ᵹ����Aװ����
��
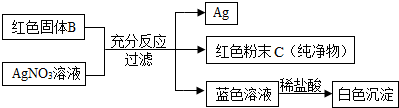
��4��bװ���п��ܻᷢ�������ѧ��Ӧ����д������1����Ӧ�Ļ�ѧ����ʽ��
CO2+Ca��OH��2=CaCO3��+H2O
CO2+Ca��OH��2=CaCO3��+H2O
��
[��˼����]
��5����ʵ���β���Ƿ��账�������账������ش���δ������粻�账������˵�����ɣ�
��Ҫ����bװ�ú��һȼ�ŵľƾ��ƽ�ʣ�������յ�
��Ҫ����bװ�ú��һȼ�ŵľƾ��ƽ�ʣ�������յ�
��
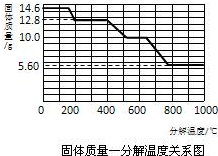
��6��ͨ������ȷ����������ʯ��������Ҫ�ɷ֣�ָ���ĸ���������ȷ�ģ���֪����ʯ�Ĵ���Ϊ80.0%����
�⣺

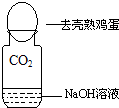 ij��ѧ��ȤС�齫CO2�ֱ�ͨ�����ʯ��ˮ������������Һ�У����ǹ۲쵽ǰ�߱���ǣ���������������
ij��ѧ��ȤС�齫CO2�ֱ�ͨ�����ʯ��ˮ������������Һ�У����ǹ۲쵽ǰ�߱���ǣ���������������