
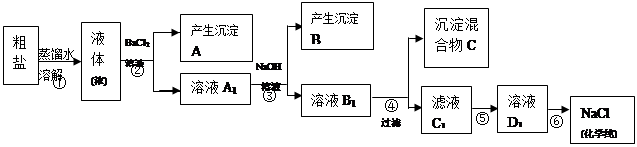



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

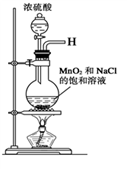
| | ʵ�鲽�� | Ԥ����������� |
| ����1 | ȡ��������Ư�����Թ��У��ȼ��� �ܽ���ٰ����ɵ�����ͨ�� �� | �� �������1������ �� �������2�����3������ |
| ����2 | ��ȷ��Ư�۱��ʣ�����ȡ��������Ư�����Թ��У��ȼ�������1 mol��L-1 HCl��Һ���ټ��� �� | �� �������2������ �� �������3������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ˮ | B��Na2CO3��Һ | C��ʯ����Һ | D������Cu(OH) 2����Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ŨNaOH��Һ | B��AgNO3��Һ | C����ˮ | D��NaCl��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ֽ��������������������Թ��ڱ��ϵĺ�ɫ���ʿ���ϡ����ϴ�� |
| B��ʧȥ��ǩ����������Һ��ϡ���ᡢ����������Һ���Ȼ�����Һ��������̼�����Һ���� |
| C����CuCl2��Һ���������м������ɣ��õ���ˮCuCl2���� |
| D������ӻ����ϴ������ĺ�ɫ�����Ƿ���Fe2O3 �IJ�������Ϊ����Ʒ���������ˮ�ܽ�����ˡ�����Һ�еμ�KSCN��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
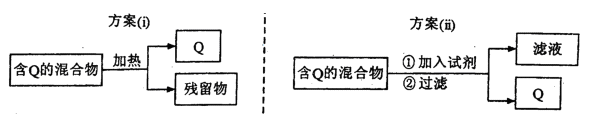
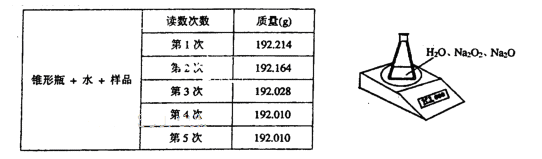

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

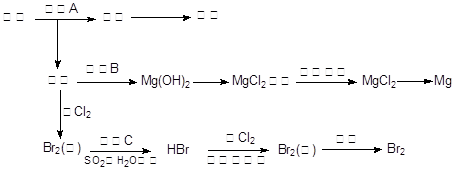
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com