

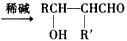 £ØR”¢R'ĪŖĢž»ł»ņĒāŌ×Ó£©
£ØR”¢R'ĪŖĢž»ł»ņĒāŌ×Ó£©
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| “߻ƼĮ |
| “߻ƼĮ |

 £ØR”¢R?ĪŖĢž»ł£©
£ØR”¢R?ĪŖĢž»ł£©







²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012½ģŗÓ±±Ź”Õż¶Ø֊ѧøßČżµŚČż“Īæ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£ØÓŠ»ś»Æѧ£©£Ø15·Ö£©ŅŅ“¼ŹĒŅ»ÖÖÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£
£Ø1£©ĘĻĢŃĢĒŌŚ¾Ę»ÆĆøµÄ×÷ÓĆĻĀ×Ŗ»ÆĪŖŅŅ“¼µÄ»Æѧ·½³ĢŹ½ŹĒ ”£
£Ø2£©Ļć¶¹ĖŲŹĒŅ»ÖÖÓĆĶ¾¹ć·ŗµÄĻćĮĻ£¬ĖüæÉŅŌĄūÓĆŅŅ“¼ŗĶBĶعżŅŌĻĀĶ¾¾¶ŗĻ³É”£ 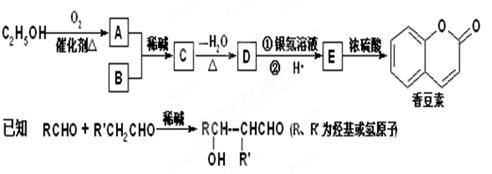
¢Ł DµÄ½į¹¹¼ņŹ½ŹĒ £»
¢Ś Ķź³ÉĻĀĮŠ»Æѧ·½³ĢŹ½£ŗ
EŌŚŅ»¶ØĢõ¼žĻĀ·¢Éś¼Ó³É¾ŪŗĻ·“Ó¦£ŗ £»
E”śĻć¶¹ĖŲ£ŗ £»
¢Ū BÓŠ¶ąÖÖĶ¬·ÖŅģ¹¹Ģ壬ĘäÖŠŗ¬ÓŠ±½»·ĒŅŗĖ“Ź²ÕńĒāĘ×ÓŠĖÄøö·åµÄĖłÓŠĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½ŹĒ £»
¢Ü ÓŠ¹ŲĻć¶¹ĖŲµÄĖµ·ØÕżČ·µÄŹĒ £ØĢī×ÖÄø£©”£
a.Ö»ÄÜÓėäåµ„ÖŹ·¢Éś¼Ó³É·“Ó¦£¬²»ÄÜ·¢ÉśČ”“ś·“Ó¦
b. 1molĻć¶¹ĖŲæÉÓė5molH2·¢Éś¼Ó³É·“Ó¦
c. 1molĻć¶¹ĖŲæÉÓė2molNaOH·¢Éś·“Ó¦
d. 1molĻć¶¹ĖŲĶźČ«Č¼ÉÕĻūŗÄ9.5molO2
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013-2014ѧğ°²»ÕŹ””°½ÄĻŹ®Š£”±øßČż3ŌĀĮŖæ¼Ąķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ŅŅ“¼ŹĒŅ»ÖÖÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£
¢ń£®ĘĻĢŃĢĒŌŚ¾Ę»ÆĆøµÄ×÷ÓĆĻĀ×Ŗ»ÆĪŖŅŅ“¼µÄ»Æѧ·½³ĢŹ½ŹĒ£ŗ__________________________________”£
¢ņ£®Ļć¶¹ĖŲŹĒŅ»ÖÖÓĆĶ¾¹ć·ŗµÄĻćĮĻ£¬æÉŅŌĄūÓĆŅŅ“¼ŗĶB£Ø·Ö×ÓŹ½ĪŖC7H6O2£©ĶعżŅŌĻĀĶ¾¾¶ŗĻ³É”£

£Ø1£©CÖŠŗ¬Ńõ¹ŁÄÜĶŵÄĆū³ĘĪŖ_______£»DµÄ½į¹¹¼ņŹ½ŹĒ_________£»D”śEÖŠ¢ŁµÄ·“Ó¦ĄąŠĶĪŖ______________”£
£Ø2£©¢ŁŅŅ“¼Éś³ÉAµÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ_________ ¢ŚÓÉEÉś³ÉĻć¶¹ĖŲµÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ______________”£
£Ø3£©BÓŠ¶ąÖÖĶ¬·ÖŅģ¹¹Ģ壬ĘäÖŠ±½»·ÉĻÖ»ÓŠŅ»øö²ąĮ“µÄĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½ŹĒ_______________”£
£Ø4£©ÓŠ¹ŲĻć¶¹ĖŲµÄĖµ·ØÕżČ·µÄŹĒ_____________________Ģī×ÖÄø£©”£
A£®Ö»ÄÜÓėäåµ„ÖŹ·¢Éś¼Ó³É·“Ó¦£¬²»ÄÜ·¢ÉśČ”“ś·“Ó¦
B£®1 molĻć¶¹ĖŲæÉÓė5 mol H2·¢Éś¼Ó³É·“Ó¦
C£®³¤ĘŚ·ÅÖĆĻć¶¹ĖŲ£¬ČŻŅ×Ńõ»Æ±äÖŹ
D£®1 molĻć¶¹ĖŲĶźČ«Č¼ÉÕĻūŗÄ9.5 mol O2
E .1 molĻć¶¹ĖŲæÉÓė2 mol NaOH·“Ó¦
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012ѧğŗÓ±±Ź”øßČżµŚČż“Īæ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£ØÓŠ»ś»Æѧ£©£Ø15·Ö£©ŅŅ“¼ŹĒŅ»ÖÖÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£
£Ø1£©ĘĻĢŃĢĒŌŚ¾Ę»ÆĆøµÄ×÷ÓĆĻĀ×Ŗ»ÆĪŖŅŅ“¼µÄ»Æѧ·½³ĢŹ½ŹĒ ”£
£Ø2£©Ļć¶¹ĖŲŹĒŅ»ÖÖÓĆĶ¾¹ć·ŗµÄĻćĮĻ£¬ĖüæÉŅŌĄūÓĆŅŅ“¼ŗĶBĶعżŅŌĻĀĶ¾¾¶ŗĻ³É”£

¢Ł DµÄ½į¹¹¼ņŹ½ŹĒ £»
¢Ś Ķź³ÉĻĀĮŠ»Æѧ·½³ĢŹ½£ŗ
EŌŚŅ»¶ØĢõ¼žĻĀ·¢Éś¼Ó³É¾ŪŗĻ·“Ó¦£ŗ £»
E”śĻć¶¹ĖŲ£ŗ £»
¢Ū BÓŠ¶ąÖÖĶ¬·ÖŅģ¹¹Ģ壬ĘäÖŠŗ¬ÓŠ±½»·ĒŅŗĖ“Ź²ÕńĒāĘ×ÓŠĖÄøö·åµÄĖłÓŠĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½ŹĒ £»
¢Ü ÓŠ¹ŲĻć¶¹ĖŲµÄĖµ·ØÕżČ·µÄŹĒ £ØĢī×ÖÄø£©”£
a.Ö»ÄÜÓėäåµ„ÖŹ·¢Éś¼Ó³É·“Ó¦£¬²»ÄÜ·¢ÉśČ”“ś·“Ó¦
b. 1molĻć¶¹ĖŲæÉÓė5molH2·¢Éś¼Ó³É·“Ó¦
c. 1molĻć¶¹ĖŲæÉÓė2molNaOH·¢Éś·“Ó¦
d. 1molĻć¶¹ĖŲĶźČ«Č¼ÉÕĻūŗÄ9.5molO2
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010Äź±±¾©ŹŠ¶«³ĒĒųøßČżµŚ¶ž“ĪÄ£Äāæ¼ŹŌ£ØĄķ×Ū£©»Æѧ²æ·Ö ĢāŠĶ£ŗĢīæÕĢā
£Ø14·Ö£©ŅŅ“¼ŹĒŅ»ÖÖÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£
£Ø1£©ĘĻĢŃĢĒŌŚ¾Ę»ÆĆøµÄ×÷ÓĆĻĀ×Ŗ»ÆĪŖŅŅ“¼µÄ»Æѧ·½³ĢŹ½ŹĒ ”£
£Ø2£©³£ĪĀ³£Ń¹ĻĀ£¬23gŅŅ“¼ĶźČ«Č¼ÉÕÉś³É¶žŃõ»ÆĢ¼ŗĶĖ®Ź±·ÅČČ680kJ£¬Ōņ±ķŹ¾øĆ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½ŹĒ ”£
£Ø3£©Ļć¶¹ĖŲŹĒŅ»ÖÖÓĆĶ¾¹ć·ŗµÄĻćĮĻ£¬ĖüæÉŅŌĄūÓĆŅŅ“¼ŗĶBĶعżŅŌĻĀĶ¾¾¶ŗĻ³É£¬ĘäÖŠBµÄ·Ö×ÓŹ½ĪŖC7H6O2”£

¢ŁDµÄ½į¹¹¼ņŹ½ŹĒ £»
¢ŚĶź³ÉĻĀĮŠ»Æѧ·½³ĢŹ½£ŗ
EŌŚŅ»¶ØĢõ¼žĻĀ·¢Éś¼Ó³É¾ŪŗĻ·“Ó¦£ŗ £»E”śĻć¶¹ĖŲ£ŗ £»
¢ŪBÓŠ¶ąÖÖĶ¬·ÖŅģ¹¹Ģ壬ĘäÖŠ±½»·ÉĻÖ»ÓŠŅ»øö²ąĮ“µÄĖłÓŠĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½ŹĒ
£»
¢ÜÓŠ¹ŲĻć¶¹ĖŲµÄĖµ·ØÕżČ·µÄŹĒ £ØĢī×ÖÄø£©”£
a.Ö»ÄÜÓėäåµ„ÖŹ·¢Éś¼Ó³É·“Ó¦£¬²»ÄÜ·¢ÉśČ”“ś·“Ó¦
b. 1molĻć¶¹ĖŲæÉÓė5molH2·¢Éś¼Ó³É·“Ó¦
c. 1molĻć¶¹ĖŲæÉÓė2molNaOH·¢Éś·“Ó¦
d. 1molĻć¶¹ĖŲĶźČ«Č¼ÉÕĻūŗÄ9.5molO2
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com