
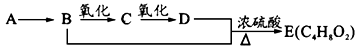
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| Cu |
| �� |
| Cu |
| �� |
| Ũ���� |
| �� |
| Ũ���� |
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| Cu |
| �� |
| Cu |
| �� |
| ϡ���� |
| �� |
| ϡ���� |
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��9�֣���֪����ȩ��һ�������¿ɱ���������Ϊ���ᡣA��ʯ���ѽ����Ҫ����֮һ������������ں���һ������ʯ�ͻ�����չˮƽ�ı�־���������л���֮���ת����ϵ��
��1��A�Ľṹ��ʽΪ ��������Ϊˮ���� ��
��2��B+D��E�ķ�Ӧ����Ϊ ��
��3��д��B��C��B+D��E��������ѧ��Ӧ����ʽ��
B��C�� ��
B+D��E�� ��
��4����ȥE��������������D���ʣ������Լ��������ǣ� ��
�鿴�𰸺ͽ���>>
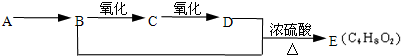
��Ŀ�����л�ѧ ��Դ��2010�꽭��ʡ�����и�һ��ѧ����ĩ���Ի�ѧ���� ���ͣ������
��9�֣���֪����ȩ��һ�������¿ɱ���������Ϊ���ᡣA��ʯ���ѽ����Ҫ����֮һ������������ں���һ������ʯ�ͻ�����չˮƽ�ı�־���������л���֮���ת����ϵ��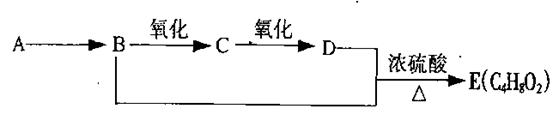
��1��A�Ľṹ��ʽΪ ��������Ϊˮ���� ��
��2��B+D��E�ķ�Ӧ����Ϊ ��
��3��д��B��C��B+D��E��������ѧ��Ӧ����ʽ��
B��C�� ��
B+D��E�� ��
��4����ȥE��������������D���ʣ������Լ��������ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��ӱ�ʡ��һ��ѧ����ĩ���Ի�ѧ���� ���ͣ������
��10�֣���֪����ȩ��һ�������¿ɱ���������Ϊ���ᡣA��ʯ���ѽ���Ҫ����֮һ������������ں���һ������ʯ�ͻ�����չˮƽ�ı�־���������л���֮���ת����ϵ��

��1��A�Ľṹ��ʽΪ ��������Ϊˮ���� ��
��2��B+D��E�ķ�Ӧ����Ϊ ��
��3��д��B��C��B+D��E��������ѧ��Ӧ����ʽ��
B��C�� ��
B+D��E�� ��
��4����γ�ȥE��������������D���ʣ������������̣� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com