
�̷�(FeSO4��7H2O)���ᷨ����һ��ϡ�н�����Ʒ�����в����ĸ���Ʒ����Ʒ���Ϊ����ɫ����ɫ�ᾧ���塣���������ɵ��ڼ���ˮ�е�pH����ˮ���������л���ϣ������ٳ�������ҪӦ����ˮ�ʾ�����ҵ��ˮ������ͬʱ����ɱ�����á�
��1��98% 1.84 g/cm3��Ũ������ϡ�����У��ܶ��½�����ϡ����50%ʱ���ܶ�Ϊ1.4g/cm3��50%���������ʵ���Ũ��Ϊ (������λС��)��50%��������30%������������ϣ�������Ũ��Ϊ (��>��<��=")40%" ��
��2��ʵ��������20%��������(100�˷������ẬSO3 20��)����ϡ���ᣬ����SO3��nH2O��ʾ20%�ķ������ᣬ��n=____________(������λС��)��
��3���̷��ڿ��������ױ���������Ϊ����������ȡ7.32�˾�������ϡ�������������BaCl2��Һ�����˵ó���9.32�ˣ���ͨ��112mL(��״��)����ǡ�ý�Fe2����ȫ�������Ʋ⾧��Ļ�ѧʽΪ ��
��4����������泥�(NH4)2SO4��FeSO4��6H2O��(�׳�Ī����)�����̷��ȶ����ڷ�����ѧ�г���������Fe2+�ı���Һ���ô�Fe2+�ı���Һ���Բⶨʣ��ϡ�����������ȡ8.64��Cu2S��CuS�Ļ������200 mL 2 mol/Lϡ������Һ������������Ӧ���£�
10NO3-��3Cu2S��16H��=6Cu2����10NO����3SO42-��8H2O
8NO3-��3CuS��8H��=3Cu2����3 SO42-��8NO��+ 4H2O
ʣ���ϡ����ǡ����V mL 2 mol/L (NH4)2Fe(SO4)2��Һ��ȫ��Ӧ��
��֪��NO3-��3Fe2����4H��= NO����3Fe3+��2H2O
�� Vֵ��Χ ��
�� ��V=48���Լ���������CuS���������� (������λС��)��
��1��7.14 mol��L��1 ; ��(��2��) ��2��0.77(2��)
��3��FeSO4��Fe2(SO4)3��10H2O (3��)����4����42��60mL(3��)����0.33��1/3(3��)
���������������1���ܶ�Ϊ1.4g/cm3��50%���������ʵ���Ũ��Ϊ��
c�� =7.14 mol��L��1��
=7.14 mol��L��1��
50%��������30%����������(�����ʻ�Ϊ1���ܶȷֱ��Ǧ�1�ͦ�2)��ϣ�������Ũ��Ϊ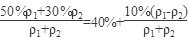 ��40%��
��40%��
��2����Ϊ20%��������Ϊ80��H2SO4��SO3 20�ˣ�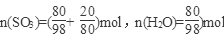 ��
��
����SO3��nH2O��ʾ20%�ķ������ᣬ��n=0.77��
��3��������Һ����������BaCl2��Һ�����˵ó���9.32�ˣ�n(SO42-)=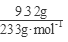 =0.04mol����ͨ��112mL(��״��)����ǡ�ý�Fe2����ȫ����(2Fe2��+Cl2==2Fe3++2Cl-)��n(Fe2+)=2��
=0.04mol����ͨ��112mL(��״��)����ǡ�ý�Fe2����ȫ����(2Fe2��+Cl2==2Fe3++2Cl-)��n(Fe2+)=2��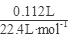 =0.01mol���ɵ���غ�n(Fe3+)=0.02mol�� 7.32�˾��庬��0.01molFeSO4Ϊ1.52g��0.01mol Fe2(SO4)3Ϊ4.00g������H2OΪ1.80g��0.1mol������Ļ�ѧʽFeSO4��Fe2(SO4)3��10H2O��
=0.01mol���ɵ���غ�n(Fe3+)=0.02mol�� 7.32�˾��庬��0.01molFeSO4Ϊ1.52g��0.01mol Fe2(SO4)3Ϊ4.00g������H2OΪ1.80g��0.1mol������Ļ�ѧʽFeSO4��Fe2(SO4)3��10H2O��
��4�������������ȫ��ΪCuS����n��CuS��=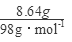 =0.09mol����Ҫ��������ʵ���Ϊy��
=0.09mol����Ҫ��������ʵ���Ϊy��
8NO3-+3CuS + 8H+==3Cu2++3SO42-+8NO��+4H2O��
3 8
0.09mol y
y=0.24mol
ʣ�����������ʵ���Ϊ��0.4mol-0.24mol=0.16mol��
0.112mol����ͣ�NH4��2Fe��SO4��2��Һ��ȫ��Ӧ��
NO3-+3Fe2++4H+==NO��+3Fe3++2H2O
3mol 4
1��10-3VL��2mol/L 0.16mol�����V=60��
����Vֵ��ΧΪ��42��V��60��
����V=48����48mL��NH4��2Fe��SO4��2��Һ��ȫ��Ӧ��Ҫ��������ʵ���Ϊ��
NO3-+3Fe2+ + 4H+��NO��+3Fe3++2H2O
3mol 4
0.048L��2mol/L n
��ã�n=0.128mol��
����������ﷴӦ����������ʵ���Ϊ��0.4mol-0.128mol=0.272mol��
��Cu2S�����ʵ���xmol��CuS�����ʵ���Ϊymol��160x+96y=8.64g�٣�
10NO3-+3Cu2S+16H+��6Cu2++10NO��+3SO42-+8H2O
3 16
x 16x/3
8NO3-+3CuS+8H+��3Cu2++3SO42-+8NO��+4H2O��
3 8
y 8y/3
16x/3+8y/3=0.272��
�ɢ٢ڽ�ã�x��0.036; y��0.03
�������CuS������������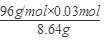 ��100%��33.33%��
��100%��33.33%��
���㣺��ѧ�뼼�������������Ũ�ȱ仯�ص㡢ͭ�����������ᷴӦ�ļ��㡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ͭ���仯�����������������й㷺Ӧ�ã�ͭ�ڻ������еij������ϼ���+l��+2����֪Cu2O��ϡ���ᷴӦ����Һ����ɫ��
��1����ҵ�Ͽ���Cu2S+O2 2Cu+SO2��Ӧ��ȡ��ͭ������32gCu2S����ʱ������ת����Ŀ�� ��
2Cu+SO2��Ӧ��ȡ��ͭ������32gCu2S����ʱ������ת����Ŀ�� ��
��2��������ͭ˿����������ϡ�����У��¶ȿ�����50�棬����H2O2����Ӧһ��ʱ������µ�60�棬�ٷ�Ӧһ��ʱ�����Ƶ�����ͭ���¶ȿ�����50��-60�������ԭ����˼ӿ췴Ӧ�����⣬���� ����CuSO4��Һ�м���һ������Na2SO3��NaCl��Һ���ȣ�����CuCl����,д������CuCl�����ӷ���ʽ ��
��3��ijС��ͬѧ����ͭ��Ũ���ᷴӦ��ʵ��ʱ������ͭƬ�����к�ɫ�������ɡ�
�ټ�ͬѧ���룺��ɫ������δ���ü��ܽ������CuO, �������û�ѧ����ʽ��ʾΪ��
Cu + H2SO4
����ͬѧ��Ϊ����ɫ�����CuO����ܺ���Cu2S��CuS.�����ɿ����� ������ĸ��
a.Cu2S��CuS���Ǻ�ɫ��
b.ͭ��Ũ�����ڼ��������·�Ӧ�������ܷų�O2
c.Ũ��������������ͭ�ǻ�ԭ����Cu�Ļ��ϼ�������S�Ļ��ϼ��½��ж��ֿ���
��4������Cu��Cu2O��CuO��ɵĻ�����У�����1 L 0.6 mol/L HNO3��Һǡ��ʹ������ܽ⣬ͬʱ�ռ���2240 mL NO���壨��״������Cu2O��ϡ���ᷴӦ�����ӷ���ʽ �����������������������H2���Ȼ�ԭ�����õ����������Ϊ g����������к�0.1 mol Cu�����û������ϡ�����ַ�Ӧ����������H2SO4�����ʵ���Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
(14 ��)���ǵؿ��к�����ߵĽ���Ԫ�أ��䵥�ʼ��Ͻ������������е�Ӧ�������㷺��
(1)���̼�Ȼ�ԭ-�Ȼ�����ʵ�����������Ʊ�������������ط�Ӧ���Ȼ�ѧ����ʽ���£�
Al2O3(s)+AlC13(g)+3C(s) ="3AlCl(g)+3CO(g)" ��H="a" kJ��mol��1
3AlCl(g)=2Al(l)+AlC13(g) ��H="b" kJ��mol��1
�ٷ�ӦAl2O3(s)+3C(s)=2Al(l)+3CO(g)�ġ�H= kJ��mol��1(�ú�a��b �Ĵ���ʽ��ʾ)��
��Al4C3�Ƿ�Ӧ�����е��м���Al4C3�����ᷴӦ(����֮һ�Ǻ�������ߵ���) �Ļ�ѧ����ʽΪ ��
(2)þ���Ͻ�(Mg17Al12 )��һ��DZ�ڵ�������ϣ�������������£���һ����ѧ�����ȵ�Mg��Al ������һ���¶���������á��úϽ���һ����������ȫ����ķ�Ӧ����ʽΪMg17Al122+17H2=17MgH2+12Al���õ��Ļ����Y(17MgH2 +12Al)��һ�������¿��ͷų�������
�������Ʊ�þ���Ͻ�(Mg17Al12)ʱͨ�������Ŀ���� ��
����6. 0 mol��L-1 HCl ��Һ�У������Y ����ȫ�ͷų�H2��1 mol Mg17 Al12��ȫ�����õ��Ļ����Y ������������ȫ��Ӧ���ͷų�H2�����ʵ���Ϊ ��
����0. 5 mol��L-1 NaOH ��1. 0 mol��L-1 MgCl2��Һ�У� ͼ8
�����Y ��ֻ�ܲ��ַų���������Ӧ������������ʵ�X������������ͼ��ͼ��ʾ(X����������������ж�ij��̬�����Ƿ���ڣ���ͬ��̬���ʳ�������������Dz�ͬ)��������NaOH ��Һ�У������Y �в�����������Ҫ������ (�ѧʽ)��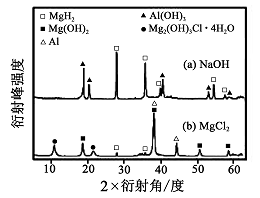
(3)�����������Խ��Al-AgO ��ؿ�����ˮ�¶�����Դ����ԭ����ͼ��ʾ���õ�ط�Ӧ�Ļ�ѧ����ʽΪ ��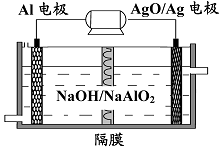
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
(6��) ��1L AlCl3��FeCl3�����Һ�м���2mol /LNaOH��Һ300mLʱ�������ij����������ֵ����������NaOH��Һ��������ʼ�ܽ⣬������NaOH��Һ��������ﵽ 350mLʱ���������ټ��٣���ԭ��Һ��FeCl3�����ʵ���Ũ��(��д���������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
�ú���A12O3��SiO2������FeO��xFe2O3�������Ʊ�A12(SO4)3��18H2O�������������£�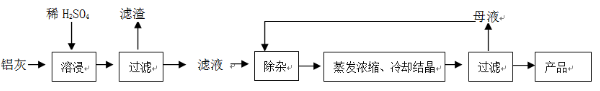
��һ�������£�MnO4- ����Mn2+��Ӧ����MnO2��
��֪�� �����������������pH
| | Al(OH)3 | Fe(OH)2 | Fe(OH)3 |
| ��ʼ����ʱ | 3.4 | 6.3 | 2.7 |
| ��ȫ����ʱ | 5.2 | 9.7 | 3.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��7�֣�����22g Mg��Al��Zn��Fe���ֻ��ý�����ĩ�Ļ������200mL����һ��������20%������Һǡ����ȫ��Ӧ���õ���ˮ��70g����Ҫ��д��������̣�
��1��������Һ��������
��2�����ɵ������������״������
��3���������ʵ���Ũ�ȣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
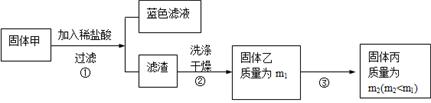
������Ʒ�������п�����������������Ʒ�ı�������һ�����ܵ�Fe3O4��ijѧϰС��Ϊ���о�������Ƭ���ֱ����������ʵ�������
�ٰ�һ����������Ƭ�ӹ��ɾ��ȷ�ĩ��
��ȡm g�÷�ĩ������28.00 mL 1 mol��L�������У�ǡ����ȫ��Ӧ�����ɱ�״���µ�����134.4 mL������Һ�е���KSCN��Һ������������
����ȡ���ݲ�ͬ�����ķ�ĩ���ּӼӵ���ͬ���(V)�����ʵ���Ũ�Ⱦ�Ϊl0.00 mol��L������������Һ�У���ַ�Ӧ����ȫ���ܽ⣬�йص�ʵ���������±���ʾ(����NO�������Ψһ��ԭ����)��
| ʵ����� | �� | �� | �� |
| �����ĩ����/g | 13.68 | 27.36 | 34.20 |
| ����������������״����/L | 2.912 | 5.824 | 6.720 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��16�֣�ͭ��Ũ���ᷴӦ��ʵ��װ����ͼ��ʾ��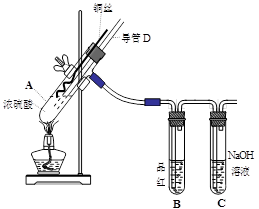
��1�� װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ�� ����Ӧ�������Թ�B�е������� ���Թ�C�������� ��
��2�� ����D���¶�(���߶�)Ӧλ�� ��Һ���ϡ�Һ���£�������D�������У���ʵ��������ų�װ���е�SO2���� ��
ʵ���з����Թ��ڳ��˲�����ɫ�����⣬��ͭ˿���滹�к�ɫ��������ɣ����п��ܺ���CuO��Cu2O��CuS��Cu2S��Ϊ̽���ijɷ֣����������µ�ʵ�顣
�������Ͽ�֪��Cu2O + 2HCl =CuCl2+ Cu + H2O�� 2Cu2O + O2���� 4CuO��2CuS+3O2����2CuO+2SO2��Cu2S+2O2����2CuO+SO2��CuS�� Cu2S��ϡHCl����Ӧ��
|
 ��3�� �������ڿ���������ʱ��ʹ�õ�ʵ���������˲����������żܡ��ƾ����⣬�������У� ��
��3�� �������ڿ���������ʱ��ʹ�õ�ʵ���������˲����������żܡ��ƾ����⣬�������У� ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������������Ҫ�������Σ���ũҵ������ũҩ����Ҫ��С����벡���������������ݼ����ڹ�ҵ������Ⱦɫ����������īˮ��ľ�ķ����ȡ�
��1�����Ƶ��̷���FeSO4��7H2O����dz��ɫ�ģ����ڿ����м��ױ�ɻ�ɫ������ɫ�ļ�ʽ������[Fe(OH)SO4]��д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
��2����֪FeSO4�ڲ�ͬ�����·ֽ�õ����ﲻͬ��������FeO��SO3��Ҳ������Fe2O3��SO3��SO2��
SO3�۵���16.8�棬�е���44.8�档
ij�о���ѧϰС����������װ�ý���ʵ��̽�����ڼ���������FeSO4�ķֽ�����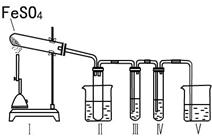
����װ�â�͢����������������Իش��������⣺
�٢�װ���ձ���ˮ���¶�Ӧ������ ��ѡ�0�桢25�桢50�桱����װ�â�������� ��
��װ�â��е��Լ������� ��ѡ����ţ���ͬ���������� ����֤����������к���SO2�� װ�â��е��Լ������� ��
| A��2 mol/LNa2CO3��Һ |
| B��Ʒ����Һ |
| C��0.5 mol/LBaCl2��Һ |
| D��0.5 mol/LBa(NO3)2 |
| �������� | Ԥ��ʵ������ | Ԥ��ʵ����� |
| ������һ����Һ�м��� �� | | �����к���Fe2O3 |
| ����һ����Һ�еμ�2�λ�ɫK3[Fe(CN)6]��Һ�� | ������ɫ���� | |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com