
��ͼ�Ǹ��ֲ�ͬ�ܶȵ�����������Ӧ����ͬ�¶ȣ�ʱ��Ҫ��ԭ����ķֲ�ͼ���������ͼ�������ش��й����⡣

(1)������������ȷ���ǣ�����ţ�_______________��
A.ͼ�к�������������ܶȣ�������ܶ�Խ����Ũ��ԽС��
B.����ԭ�����¶���ͬʱ����ͬŨ�ȵ����ᱻ��ԭ�IJ��ﲻ�ǵ�һ�ģ�ֻ����ijŨ��ʱ����ij�ֲ���Ϊ�����ѡ�
C.�����Ũ��Խ��ԭ������NԪ�صͻ��ϼ۲���Խ��
D.�������ܶȴ���1.3g��cm-3ʱ����ԭ������ҪΪNO��NO2
(2)����ͼ��ѡ��������ҩƷ��֤��������1.36 g��cm-3���ᷴӦ�����������к���NO��N2��O2�����������ɿ��ƣ�����װ�ú̶�װ��ʡ�ԣ�
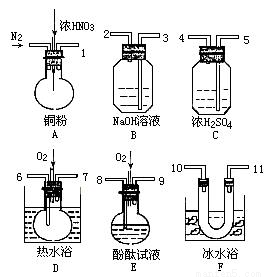
��֪����.NO+NO2+2OH-=2NO2-+H2O
��.�����£�NO2��N2O4��ϴ��ڣ���0��ʱ����ֻ����ɫ��N2O4Һ�������ڡ�
������������˳����������ӣ�����ӿڵı�ţ�Ϊ______________��
�ڷ�Ӧǰ��ͨ��N2��Ŀ����___________________��
��ȷ�������к���������__________________��
��װ��F��������________________��
������A�����Ļ��������NO2��NO������ֱ�ΪV1mL��V2mL��V1��V2����Ϊ0�����ұ���װ�������ܽ�������������ȫ���գ���װ����������Ҫ�������������Ϊ��________mL����ͬ״̬�£���
 С��ſ�ʱ��ҵϵ�д�
С��ſ�ʱ��ҵϵ�д� һ������ϵ�д�
һ������ϵ�д� �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д� �¸��̵�ѧϵ�д�
�¸��̵�ѧϵ�д� ����ͬѧһ����ʦȫ�źþ�ϵ�д�
����ͬѧһ����ʦȫ�źþ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�갲��ʡ�����и߶���ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ������
(NH4)2Fe(SO4)2•6H2O(Ī���Σ�dz��ɫ��ʽ��392)�ڶ��������г������궨������ء��ظ���ص���Һ�ı����ʣ���������ѧ�Լ���ҽҩ�Լ�����ұ�𡢵�Ƶȡ�
�ش��������⣺
��1��Ī�����ڿ����б����������ȶ���������¶���ڿ�����Ҳ����ʣ�����Ī�����Ƿ���ʵ��Լ���________��
��2��ȷ��ȡmg������Ī���Σ�����ƿ�м���20mLˮ����ܽ⣬��ij����K2Cr2O7��Һ�ζ����յ㣮�ظ�����3�Σ�����й��������£�
ʵ����� | ��ʼ����/mL | �յ����/mL |
I | 2.50 | 22.58 |
�� | 1.00 | 23.12 |
�� | 0.00 | 19.92 |
��K2Cr2O7��ҺӦ�÷���________ʽ�ζ����У�
��д���ζ������з�Ӧ�����ӷ���ʽ��________��
������K2Cr2O7��Һ�����ʵ���Ũ��Ϊ________mol/L(�ú�M�Ĵ���ʽ��ʾ)
��3��ij������ͨ��ʵ�����Ī���ξ������ʱ�ķֽ���
�ټ�ͬѧ������룺�ֽ���������N2��Fe2O3��SO3��H2O�������ʣ����Ƿ�ͬ�Ⲣ˵�����ɣ�________��
����ͬѧ�������ͼװ�ã�����Aװ���еĹ����Ϊ����ɫ�����������к���________��Cװ���к�ɫ��ȥ��˵����������к���________��Cװ�ú�Ӧ����β������װ��D��D��ʢ�е��Լ�������________(дһ�ּ���)��
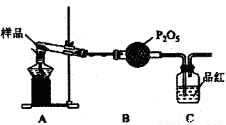
�۱�ͬѧ����������װ��֤���ֽ�����к��а�����ֻ�����B��C�е��Լ����ɣ����������Լ�ΪB________��C________��
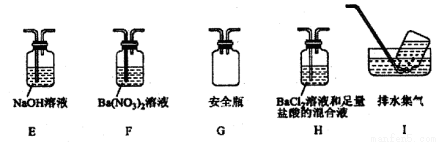
�ܶ�ͬѧ��ΪĪ���ηֽ���ܻ�����N2��SO3���������װ����ͼ2��ѡ���Ҫ��װ�ü���֤��������ȷ������˳�������������A��________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꽭��ʡ��һ��ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ������
��1��A��B��C��D�����ֶ�����Ԫ�أ���֪A��Cͬ���壬B��Dͬ���ڣ�A����̬�⻯���C����̬�⻯���ȶ���B�������ӱ�D��������������ǿ����B�������ӱ�C����������һ�����Ӳ㣬����A��B��C��D���𣬰��ɴ�С��˳������
��ԭ��������____________________�� ��ԭ�Ӱ뾶��____________________��
��2�������л�������: H2O2��Na2O��I2��NaCl��CO2��NH4Cl��Na2O2���û�ѧʽ�ش��������⣺
��ֻ�м��Թ��ۼ��Ĺ��ۻ�������____________���ڼ������Ӽ����зǼ��Լ��Ļ�������_________��
��3����aAn+��bB2-�������ӵĺ�����Ӳ�ṹ��ͬ����a����_________����ѡ�
A�� b+n+2 B��b+n-2 C��b-n-2 D��b-n+2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���Ĵ�ʡ�㰲���������ڽ���üɽ����2017������ڶ�����Ͽ������ۻ�ѧ�Ծ� ���ͣ��ƶ���
ͨ������;�����Ժϳɾ��е����Եĸ߷��Ӳ���F��

��֪��
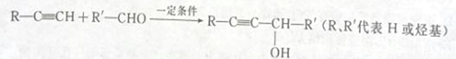
(1)����C���������ŵ�����Ϊ____________���Լ�X�Ľṹ��ʽ��___________��
(2)����Y��________________
(3)��д����Ӧ�ڵĻ�ѧ����ʽ��__________________
(4)C��H2��1��2��Ӧ���ò����ϵͳ������________________
(5)��Ӧ�Ļ�ѧ��Ӧ����Ϊ________����Ӧ�ݵĻ�ѧ����ʽ_____________��
(6)��Ӧ�ٳ��ñ���ʳ��ˮ����H2O����Ŀ����_________________
(7)д����������������B������ͬ���칹��_______________��
������������Һ��Ӧ �ں˴Ź���������3�����շ�
(8)���������ϳ���Ϣ���Ա�Ȳ����ȩ������Ϊԭ�ϣ����Լ���ѡ�����·�ߺϳ����ᡪ
 _________________��
_________________��
�ϳ�·��ʾ�����£�CH2=CH2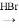 CH3CH2Br
CH3CH2Br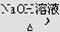 CH3CH2OH
CH3CH2OH
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ���Ĵ�ʡ�㰲���������ڽ���üɽ����2017������ڶ�����Ͽ������ۻ�ѧ�Ծ� ���ͣ�ѡ����
G�Ǻϳ�˫��A��ԭ�ϣ���ṹ��ʽ��ͼ������G˵������ȷ����
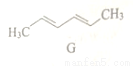
A. ����ʽΪC6H10
B. ����ϩ��ͬϵ��
C. ���Է����ӳɷ�Ӧ
D. ������ˮ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ�����и�һ��ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
��һ��������ͭ�ۼ��뵽100mLijŨ�ȵ�ϡ�����У���ַ�Ӧ��������ʣ��m1gͭ�ۣ���ʱ���ռ���NO����448mL(��״��)��Ȼ��Ȼ��������������м�������ϡ���������ٷ�ӦΪֹ������ʣ��ͭ��m2g����(m1-m2)Ϊ����λ��g��
A. 5.76 B. 3.84 C. 1.92 D. ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ�����и�һ��ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
���и����������Ϸ�Ӧ�������������ɣ��������г������ɵ���
�ٽ�����Ͷ�뵽FeCl3��Һ �ڹ���Na2O2Ͷ��������Һ
��Ba(HCO3)2��Һ��NaHSO4��Һ ������SO2ͨ��BaCl2��Һ
A. �٢� B. �٢� C. �ڢ� D. �٢�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2017�콭��ʡ��ͨ�и����߿�ȫ��ģ�⣨�壩��ѧ �Ծ��������棩 ���ͣ������
���÷Ͼ�п��Ƥ�Ʊ�����Fe3O4�������Ӽ�������ZnO��һ���Ʊ�����ͼ���£�
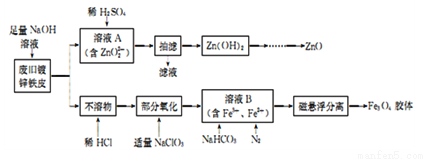
��1����NaOH��Һ�����Ͼ�п��Ƥ������Ӧ�Ļ�ѧ����ʽΪ____________��
��2��������ҺA�еμ�ϡH2SO4���������ܽ������Zn(OH)2���ɸ��õķ�����____________��
����Zn(OH)2������ZnOǰ�����ϴ�ӣ��������ϴ���Ƿ�ɾ��ķ�����______________��
��3�������м���NaC1O3������Ӧ�����ӷ���ʽΪ______________��
��4��������ҺB�Ƶ�Fe3O4�������ӵĹ�����ͨ��N2��ԭ����____________��
����ҺB����Ҫ���������ӷ�Ӧ����ʽΪ________________��
��ȷ����ҺB�к���Fe2+��ʵ�鷽����_____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�긣��ʡ���ݰ�һ�С�Ȫ��ʵ����ѧ��һ��ѧ����ĩ������ѧ�Ծ��������棩 ���ͣ�ѡ����
����������ΪN��R2?��������ΪA��������ӵĺ��������Ϊ�� ��
A. A?N+2 B. A?N?2 C. A+N?2 D. A+N+2
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com