
| ��� | ʵ��Ŀ�� | ̼��/g | ����/g | ����/% |
| �� | Ϊ����ʵ�������� | 0.5 | 2.0 | 90.0 |
| �� | ����Ũ�ȵ�Ӱ�� | 0.5 | | 36.0 |
| �� | | 0.2 | 2.0 | 90.0 |
| ʵ�鲽��ͽ���(��Ҫ��д�����������)�� |
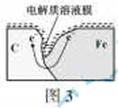 ��ԭ��Ӧ 2H2O+O2+4e-=4OH- ����4H++O2+4e-=2H2O��
��ԭ��Ӧ 2H2O+O2+4e-=4OH- ����4H++O2+4e-=2H2O��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
| ʵ���� | �� | �� | �� | �� |
| ϡHNO3�����mL�� | 100 | 200 | 300 | 400 |
| ʣ�����������g�� | 9.0 | 4.8 | 0 | 0 |
| NO�����L������� | 1.12 | 2.24 | 3.36 | 4.48 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ʵ�鷽�� | ʵ������ |
| ����ɰֽ�����þ�����ˮ��Ӧ������Ӧ����Һ�еμӷ�̪ | ��A������ˮ�棬�۳�С����ˮ�����ζ�����֮��ʧ����Һ��� |
| �������Ƶ�H2S������Һ�еμ����Ƶ���ˮ | ��B���������壬���ڿ�����ȼ�գ���Һ��Ϊdz��ɫ |
| ��������з�̪��Һ����ˮ��Ӧ | ��C����Ӧ��ʮ��ǿ�ң�������������ڿ�����ȼ�� |
| ��þ����2mol/L���ᷴӦ | ��D�����ҷ�Ӧ��������ȼ���� |
| ��������2mol/L���ᷴӦ | ��E�����ɰ�ɫ��״����������������ʧ |
| ����AlCl3��Һ�еμ�NaOH��Һ������ | ��F�����ɵ���ɫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
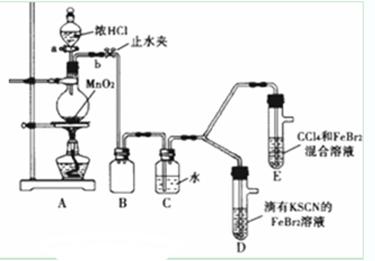
| ʵ����� | ʵ������ | ���� |
| ����a����Բ����ƿ�е�������Ũ���Ȼ��رջ���a����ȼ�ƾ��� | Dװ���У���Һ��� Eװ���У�ˮ����Һ�����CCl4�������Ա仯 | Cl2��Br2��Fe3+����������ǿ������˳��Ϊ�� ______________________ |
| ��SCN��2������±�ص������ƣ������ԣ�Cl2����SCN��2�� ��Cl2��Br2��Ӧ����BrCl�����ʺ�ɫ���Դ���ɫ�����е�Ϊ5�棬��ˮ����ˮ�ⷴӦ�� ��AgClO��AgBrO��������ˮ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

 Na2S2O3��aq�� ��III��
Na2S2O3��aq�� ��III���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| ѡ�� | ʵ������ | ʵ��Ŀ�Ļ�ʵ����� |
| A | ��ʢ��2 mL 0.1 mol/L AgNO3��Һ���Թ��еμ�5��0.1 mol/L NaCl��Һ���а�ɫ�������ɣ��������еμ�5��0.1 mol/L KI��Һ | ˵��һ�ֳ�����ת��Ϊ�ܽ�ȸ�С�ij��� |
| B | ��1 mL 20% ��������Һ�м���3��5��ϡ���ᣬˮԡ����5 min����ȴ���ټ�������Cu(OH)2����Һ������ | ֤�������ܷ���ˮ�ⷴӦ |
| C | ˮԡ����Ũ���ᡢŨ����ͱ��Ļ�����ֱ�������Һ��õ��Ĵֲ�Ʒ | �Ʊ��������� |
| D | ������,�ֱ���2֧�Թ��м�����ͬ�������ͬŨ�ȵ�Na2S2O3��Һ,�ٷֱ����������ͬŨ�ȵ�ϡ���� | �о�Ũ�ȶԷ�Ӧ���ʵ�Ӱ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
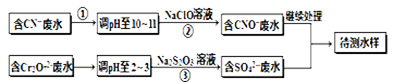
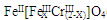 (��������,�������ֱ�ʾԪ�ؼ�̬)�ij���������1mol Cr2O72-�������a mol FeSO4?7H2O�����н�����ȷ���� ��
(��������,�������ֱ�ʾԪ�ؼ�̬)�ij���������1mol Cr2O72-�������a mol FeSO4?7H2O�����н�����ȷ���� ��| A��x ="0.5" ,a =8 | B��x ="0.5" ,a = 10 | C��x =" 1.5" ,a =8 | D��x =" 1.5" ,a = 10 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
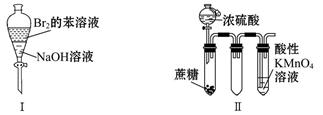
| A�����ã��ϲ���Һ��ɫ���ֲ��� |
| B��ʵ�������KMnO4��Һ�г������ݣ���ɫ�ޱ仯 |
| C��ʵ�����ϡ����Ƭ�̣���Һ�������ݲ��������ƿ��ʼ�ձ�����ɫ |
| D��ʵ��������������Һ�����ɫ��ֹͣ���ȣ�������ͨ����ϵʱ�ɲ��������ЧӦ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com