
��ˮ���ۺ����ÿ����Ʊ�����þ����������ͼ��ʾ��
(1)���ڿ����м���MgCl2��6H2O�����ɵ���Mg(OH)Cl��MgO��д����Ӧ��Ӧ�Ļ�ѧ����ʽ�� ��
�õ�ⷨ��ȡ����þʱ����Ҫ��ˮ�Ȼ�þ���ڸ����HCl�����м���MgCl2��6H2Oʱ���ܵõ���ˮMgCl2����ԭ���� ��
(2)Mg(OH)2�����л��е�Ca(OH)2Ӧ������ȥ��д��ʵ�鲽�裺 ��
(3)ʵ�����ォ�����Ƴɾ��εĹ����У����ܽ⡢���ˡ�������������IJ����ж�Ҫ�õ����������ֱ�˵���������������ʹ�ò�������Ŀ�ġ�
�ܽ�ʱ�� ��
����ʱ�� ��
����ʱ�� ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���и�����������÷�Һ©���������
| A������ˮ | B�������屽 | C���Ҵ�����ˮ | D��������Ҵ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ����̼���̿�Ϊ��Ҫԭ������MnO2�Ĺ����������£� �й��������↑ʼ�����ͳ�����ȫ��pH���±���
�й��������↑ʼ�����ͳ�����ȫ��pH���±���
| �������� | Al��OH��2 | Fe��OH��3 | Fe��OH��2 | Cu��OH��2 | Pb��OH��2 | Mn��OH��2 |
| ��ʼ������pH | 3.3 | 1.5 | 6.5 | 4.2 | 8.0 | 8.3 |
| ������ȫ��pH | 5.2 | 3.7 | 9.7 | 6.7 | 8.8 | 9.8 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������±ʯ(K2SO4��MgSO4��2CaSO4��2H2O)���ڡ�������ˮ�д�������ƽ�⣺
K2SO4��MgSO4��2CaSO4��2H2O(s)  2Ca2����2K����Mg2����4
2Ca2����2K����Mg2����4 ��2H2O
��2H2O
Ϊ�ܳ�����ü���Դ���ñ���Ca(OH)2��Һ�ܽ���±ʯ�Ʊ�����أ������������£�
(1)������Ҫ�ɷ���________��________�Լ�δ����±ʯ��
(2)�û�ѧƽ���ƶ�ԭ������Ca(OH)2��Һ���ܽ���±ʯ����K����ԭ��_________________________________________________��
(3)�����ӡ������У��ȼ���________��Һ��������Ȳ������ˣ��ټ���________��Һ����ҺpH�����ԡ�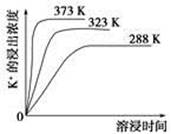
(4)��ͬ�¶��£�K���Ľ���Ũ�����ܽ�ʱ��Ĺ�ϵ��ͼ����ͼ�ɵã������¶����ߣ�
��________________________________________________________��
��________________________________________________________��
(5)�����Կ�����̼����Ϊ�ܽ��������ܽ������лᷢ����CaSO4(s)�� ?
? CaCO3(s)��
CaCO3(s)��
��֪298 Kʱ��Ksp(CaCO3)��2.80��10��9��
Ksp(CaSO4)��4.90��10��5������¶��¸÷�Ӧ��ƽ�ⳣ��K(������������λ��Ч����)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����ң���Ҫ�ɷ�ΪAl��Al2O3����������CuO��SiO2��FeO��Fe2O3���ʣ�Ϊԭ�ϣ����Ƶ�Һ��ۺ��Ȼ���Alm��OH��nCl3m-n�������IJ��ֹ�������ͼ��ʾ�����ֲ���Ͳ�������ȥ����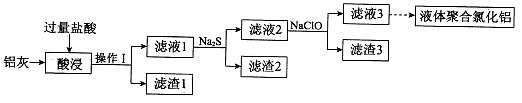
��֪ijЩ������������±���
��1������I�� ��Al2O3�����ᷴӦ�����ӷ���ʽ�� ��
��2������2Ϊ��ɫ���ú�ɫ���ʵĻ�ѧʽ�� ��
��3������Һ2�м���NaClO��Һ�����ٲ������ɫ��������ʱ��Һ��pHԼΪ3��7��NaClO�������� ��
��4������Һ3��pH����4��2��4��5������ˮ�ⷴӦ�õ�Һ��ۺ��Ȼ�������Ӧ�Ļ�ѧ����ʽ�� ��
��5������Һ3���Ҳ���Եõ�Һ��ۺ��Ȼ�����װ����ͼ��ʾ�������ӽ���Ĥֻ����������ͨ�����缫Ϊ���Ե缫����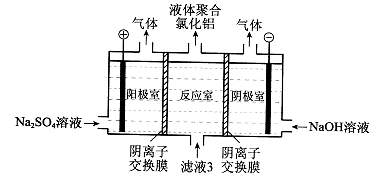
��д�������ҵĵ缫��Ӧ�� ��
�ڼ����ڷ�Ӧ�������ɾۺ���������ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�⻯�����Ʊ������л��⻯���ԭ�ϣ���ҽҩ��������̵����������ȡ���ҵ������м��ԭ���Ʊ�NaI������Ҫ��������ͼ�� 
��1��д����мת��ΪFe(OH)3��Ӧ�����ӷ���ʽ�� ��
��2���жϵ�����ȫ��Ӧ�ķ����� ��
��3������Һ�õ�NaI����IJ����� ��
��4���ⶨ��Ʒ��NaI�����ķ����ǣ�
a����ȡ3��000g��Ʒ�ܽ⣬��250mL����ƿ�ж��ݣ�
b����ȡ25��00mL������Һ����ƿ�У�
c����0��100mol ��Һ�ζ����յ㣬����
��Һ�ζ����յ㣬���� ��Һ�����ƽ��ֵΪ19��00mL��
��Һ�����ƽ��ֵΪ19��00mL��
�������ⶨ�������������У���Ҫ����Ƿ�©Һ�������� ��
����ʹ��ǰ�������ϴ�������� ��
��������Ʒ��NaI����������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ���������̿�������������Һ��ȡ����̣�����������Һ�Ʊ��ߴ�̼���̡���������淋Ĺ�����������(���̿����Ҫ�ɷ���MnO2�������й衢��������������������ؽ��������������)��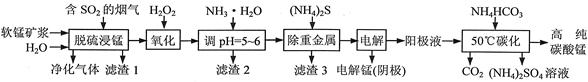
��1��һ���¶��£���������̡���Ҫ����ΪMnSO4���÷�Ӧ�Ļ�ѧ����ʽΪ ��
��2��������2������Ҫ�ɷֵĻ�ѧʽΪ ��
��3�������ؽ�����ʱʹ��(NH4)2S����ʹ��Na2S��ԭ���� ��
��4������⡱ʱ�ö��Ե缫�������ĵ缫��ӦʽΪ ��
��5����50��̼�����õ��ߴ�̼���̣���Ӧ�����ӷ���ʽΪ ����50��̼����ʱ�������NH4HCO3�����ܵ�ԭ���ǣ�ʹMnSO4���ת��ΪMnCO3�� �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������±ʯ(K2SO4��MgSO4��2CaSO4��2H2O)���ڡ�������ˮ�д�������ƽ�⣺
K2SO4��MgSO4��2CaSO4��2H2O(s) 2Ca2����2K����Mg2����4SO42����2H2O
2Ca2����2K����Mg2����4SO42����2H2O
Ϊ�ܳ�����ü���Դ���ñ���Ca(OH)2��Һ�ܽ���±ʯ�Ʊ�����أ������������£�
(1)������Ҫ�ɷ���________��________�Լ�δ����±ʯ��
(2)�û�ѧƽ���ƶ�ԭ������Ca(OH)2��Һ���ܽ���±ʯ����K����ԭ��
_____________________________________________________________��
(3)�����ӡ������У��ȼ���________��Һ��������Ȳ������ˣ��ټ���________��Һ����ҺpH�����ԡ�
(4)��ͬ�¶��£�K���Ľ���Ũ�����ܽ�ʱ��Ĺ�ϵ��ͼ����ͼ�ɵã������¶����ߣ�
��_______________________________________________________��
��_______________________________________________________��
(5)�����Կ�����̼����Ϊ�ܽ��������ܽ������лᷢ����CaSO4(s)��CO32�� CaCO3(s)��SO
CaCO3(s)��SO
��֪298 Kʱ��Ksp(CaCO3)��2.80��10��9��
Ksp(CaSO4)��4.90��10��5������¶��¸÷�Ӧ��ƽ�ⳣ��K(������������λ��Ч����)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������dz��õ�����������ҵ�������̿���Ҫ�ɷ���MnO2��Ϊԭ���Ʊ�������ؾ��塣��ͼ��ʵ�����Ʊ��IJ������̣�
������Ӧ�ڵĻ�ѧ����ʽ��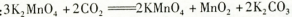
��֪��
��1���������̿�KClO3��KOH����ʱ�������ô�������ѡ����������������_______��
��Ӧ�ٵĻ�ѧ����ʽΪ______��
��2������Һ�еõ�KMnO4�����ʵ�����������________��ѡ����ĸ���ţ���ͬ����
A������ B������ C������ D������ E����ȴ�ᾧ
��3���Ʊ���������Ҫ�õ�������CO2���塣��ȡ��CO2�����ѡ�������Լ���_________��
A��ʯ��ʯ B��Ũ���� C��ϡ���� D������
��4��ʵ��ʱ����CO2����������KHCO3�����µõ���KMnO4��Ʒ�Ĵ��Ƚ��͡�ԭ����______ ��
��5������CO2��ͨ�������ѿ��ƣ���˶�����ʵ�鷽�������˸Ľ�������ʵ����ͨCO2��Ϊ���������ᡣ�������Ϸ�����ѡ����������________ ���õ��IJ�Ʒ���ȸ��ߡ�
A������ B��Ũ���� C��ϡ����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com