
����Ŀ��I.����β���dz��е���Ҫ������Ⱦ��о���������β����Ϊ������������Ҫ����������ȼ������ʱ������Ӧ��N2��g��+O2��g��![]() 2NO��g�����÷�Ӧ�ǵ�������β���к���NO��ԭ��֮һ��T��ʱ����5L�ܱ������г���6.5 mol N2��7.5 molO2����5 minʱ��Ӧ�ﵽƽ��״̬����ʱ������NO�����ʵ�����5mol��
2NO��g�����÷�Ӧ�ǵ�������β���к���NO��ԭ��֮һ��T��ʱ����5L�ܱ������г���6.5 mol N2��7.5 molO2����5 minʱ��Ӧ�ﵽƽ��״̬����ʱ������NO�����ʵ�����5mol��
��1��5 min�ڸ÷�Ӧ��ƽ������v��NO��=___________����T��ʱ���÷�Ӧ��ƽ�ⳣ��ֵΪ_________��
��2����Ӧ��ʼ���ﵽƽ��Ĺ����У����������и�����仯���ǣ�����ţ�___________��
a�����������ܶ� b����������ѹǿ
c������Ӧ���� d����λʱ���ڣ�N2��NO��������֮��
��3����֪������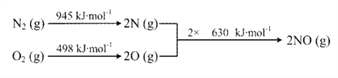
���� N2(g) + 2O2(g) === 2NO2(g) ��H= + 68 kJ��mol��1
����Ȼ�ѧ����ʽ��˵���¶ȶ���NO����NO2ƽ��ת���ʵ�Ӱ�죺_____________________��
II. ��pm2.5��������Ҫ�ɷ���SO2��NOx��CxHy������������ȡ�
��4����������������������_____________
��5��NaClO2��Һ��������SO2��NO����NaClO2��Һ��ͨ�뺬��SO2��NO�����壬��Ӧ�¶�Ϊ323 K��NaClO2��ҺŨ��Ϊ5��103mol��L1����Ӧһ��ʱ�����Һ������Ũ�ȵķ���������±�
���� | SO42 | SO32 | NO3 | NO2 | Cl |
c/��mol��L1�� | 8.35��104 | 6.87��106 | 1.5��104 | 1.2��105 | 3.4��103 |
��д��NaClO2��Һ������������Ҫ��Ӧ�����ӷ���ʽ________________________
����ѹǿ��NO��ת����______�����ߡ������䡱���͡�����
����ʵ������֪������Ӧ���ʴ���������Ӧ���ʣ���������������С��������ԭ�����SO2�ܽ��Դ���NO����������___________________
��6����ͼ���װ�ÿɽ�SO2��NOת��Ϊ(NH4)2SO4��
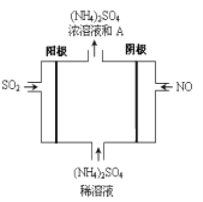
�������ĵ缫��Ӧʽ��______________________________
��SO2��NOͨ����װ���е������Ϊ___________________
���𰸡� 0.2mol/��Lmin�� 1.25 cd 2NO(g) �� O2(g) ![]() 2NO2(g) ��H = ��115 kJ��mol��1���¶�������ƽ�����淴Ӧ�����ƶ���NOת��ΪNO2��ƽ��ת���ʽ��� SO2��NOx 4NO+3ClO2��+2OH-=4NO3��+3Cl��+2H2O ��� SO2�ij�ʼŨ�ȴ���NO(���������� NO+5e-+6H+ =NH4++H2O 5:2
2NO2(g) ��H = ��115 kJ��mol��1���¶�������ƽ�����淴Ӧ�����ƶ���NOת��ΪNO2��ƽ��ת���ʽ��� SO2��NOx 4NO+3ClO2��+2OH-=4NO3��+3Cl��+2H2O ��� SO2�ij�ʼŨ�ȴ���NO(���������� NO+5e-+6H+ =NH4++H2O 5:2
��������(1) T��ʱ����5L�ܱ������г���6.5 mol N2��7.5 molO2����5 minʱ��Ӧ�ﵽƽ��״̬����ʱ������NO�����ʵ�����5mol���������������ݿ���֪����
N2��g��+O2��g��![]() 2NO��g��
2NO��g��
��ʼŨ�� 1.3 1.5 0
ת��Ũ�� 0.5 0.5 1
ƽ��Ũ�� 0.8 1 1
��![]() ��
��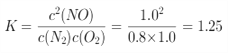 ����ȷ����0.2mol/��Lmin���� 1.25��
����ȷ����0.2mol/��Lmin���� 1.25��
(2)��������������������������������������������ܶȲ�����a��������������ܵ����ʵ������������������������������ѹǿ������b�������淴Ӧ��������Ӧ��Ũ�Ƚ���������Ӧ����������c��ȷ���淴Ӧ��������Ӧ��Ũ�Ƚ���������Ӧ�����������������Ũ���������淴Ӧ�����������ʵ�λʱ������N2����������С��NO����������������λʱ������ N2��NO��������֮�ȼ�С��d��ȷ����ȷ����cd��
��3������H=��Ӧ��ϼ����յ�������-������ϼ��ų�������=945+498-2��630=173 kJ��mol��1��N2(g) + O2(g) === NO(g) ��H=+173 kJ��mol��1��N2(g) + 2O2(g) === 2NO2(g) ��H= + 68 kJ��mol��1�����ݸ�˹���ɿɵã�2NO(g) �� O2(g) ![]() 2NO2(g) ��H = ��115 kJ��mol��1���¶�������ƽ�����淴Ӧ�����ƶ���NOת��ΪNO2��ƽ��ת���ʽ�������ȷ����2NO(g) �� O2(g)
2NO2(g) ��H = ��115 kJ��mol��1���¶�������ƽ�����淴Ӧ�����ƶ���NOת��ΪNO2��ƽ��ת���ʽ�������ȷ����2NO(g) �� O2(g) ![]() 2NO2(g) ��H = ��115 kJ��mol��1���¶�������ƽ�����淴Ӧ�����ƶ���NOת��ΪNO2��ƽ��ת���ʽ�����
2NO2(g) ��H = ��115 kJ��mol��1���¶�������ƽ�����淴Ӧ�����ƶ���NOת��ΪNO2��ƽ��ת���ʽ�����
II.��4��������������������������������Ҫԭ��������������γ������������SO2��NOx����ȷ�𰸣�SO2��NOx��
��5�����������ƾ�������������![]() ��Һ�ʼ���,��
��Һ�ʼ���,��![]() ��Һ������������Ҫ��Ӧ�����ӷ���ʽΪ
��Һ������������Ҫ��Ӧ�����ӷ���ʽΪ![]() ����ȷ����
����ȷ����![]() ������Ӧ�������С����������ѹǿ��NO��ת�����������ȷ����
������Ӧ�������С����������ѹǿ��NO��ת�����������ȷ����![]() �������
�������
���������Ϣ��֪������Ӧ���ʴ���������Ӧ����������SO2�ܽ��Դ���NO����������SO2�ij�ʼŨ�ȴ���NO����ȷ�𰸣����ڣ�SO2�ij�ʼŨ�ȴ���NO(����������)��
��6���ٵ���������������ԭ��Ӧ��NO�õ�������笠�����Ӧ���ӷ���ʽΪ��NO+5e-+6H+ =NH4++H2O����ȷ����NO+5e-+6H+ =NH4++H2O��
��SO2ʧ�����γ���������Ӧ����ʽΪ:5SO2+2NO+8H2O=(NH4)2SO4+4H2SO4����SO2��NOͨ����װ���е������Ϊ5:2����ȷ����5:2��


 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����нṹ������һ��û�к��Dz��빹�ɵ��ǣ� ��
A. ��� B. ø C. ATP D. Ҷ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������˵������ȷ���ǣ� ��
A.���й��ۼ��Ļ�����һ���ǹ��ۻ�����
B.�ǽ���Ԫ��֮�䲻�����γ����ӻ�����
C.������Ӽ�һ�����ڷ��Ӽ����������������ڲ�һ�������ۼ�
D.���ֲ�ͬ�ķǽ���Ԫ���γɵĻ�����ֻ���ڼ��Թ��ۼ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ķ������������������ϣ�
����һ��
���϶���
���� | �۵�/�� | �е�/�� | �ܶ�/g��cm-3 | �ܽ��� |
�Ҷ����� C2H6O2�� | -11.5 | 198 | 1.11 | ������ˮ���Ҵ� |
��������C3H8O3�� | 17.9 | 290 | 1.26 | �ܸ�ˮ���ƾ�������Ȼ��� |
�ش��������⣨��д��ţ���
A������ | B����ȡ�� | C�����ܽ⡢�ᾧ���������ķ��� | D����Һ�� |
��1�� ��������Ȼ��ƺʹ���Ļ�����з�����������Ӧ��__________��
��2�����Ҷ����ͱ�������������ѷ�����__________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����500![]() ��
��![]() ��
��![]() �Ļ����Һ�ֳ�5�ȷݣ�ȡһ�ݼ��뺬
�Ļ����Һ�ֳ�5�ȷݣ�ȡһ�ݼ��뺬![]()
![]() �����Ƶ���Һ��ǡ��ʹ��������ȫ��������ȡһ�ݼ��뺬
�����Ƶ���Һ��ǡ��ʹ��������ȫ��������ȡһ�ݼ��뺬![]()
![]() ����������Һ��ǡ��ʹ��������ȫ��������û����Һ�м�����Ũ��Ϊ
����������Һ��ǡ��ʹ��������ȫ��������û����Һ�м�����Ũ��Ϊ
A. ![]() B.
B. ![]()
C. ![]() D.
D. ![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������Ũ�Ⱦ�Ϊ0.1 mol��L��1��������Һ�������ᡢ�ڴ��ᡢ���������ơ����Ȼ�李��ݴ���李���������李��߰�ˮ����ش��������⣺
��1���١��ڡ��ۡ���������Һ����ˮ�������H��Ũ���ɴ�С��˳����(�����)___________��
��2���ܡ��ݡ��ޡ���������Һ��NH![]() Ũ���ɴ�С��˳����(�����)_______________��
Ũ���ɴ�С��˳����(�����)_______________��
��3�����ۺܰ͢������1��2��Ϻ��Һ�и�����Ũ���ɴ�С��˳���ǣ�__________________��
��4����֪t ��ʱ��KW��1��10��13����t ��(�����������������)________25�档��t ��ʱ��pH��11��NaOH��Һa L��pH��1��H2SO4��Һb L���(���Ի�Ϻ���Һ����ı仯)�������û����Һ��pH��2����a��b��________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ҹ�Ůҩѧ������������ʮ����ǰ�����������أ���Ч����ű�����ߵ������ʣ��Ӷ����2015��ŵ��������ѧ��ҽѧ���������صķ��ӽṹ��ͼ��ʾ�������й������ص��й�˵����ȷ����( )
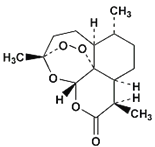
A������������̼ԭ�ӿ��ܴ���ͬһƽ����
B�������к���������ֻ������
C�������صĻ�ѧʽ��C15H22O5
D�������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��C��P��Cl��Fe��Ԫ�ؼ��仯��������Ҫ��Ӧ�ã��ش��������⣺
(1)Cԭ�ӵļ۵��ӹ������ʽΪ__________________ ��
(2)CC14���ӵĿռ乹����______________��������ԭ�Ӳ�ȡ______________�ӻ�����CC14��Ϊ�ȵ������һ��������__________________ (��д���ӷ��ţ���
(3)PC13����________________���ӣ�����ԡ��Ǽ��ԡ�����
(4)FeO��NiO�ľ���ṹ����NaCl����ṹ��ͬ������Fe2+��Ni2+�����Ӱ뾶�ֱ�Ϊ 7.8��l0-2nm��6.9��l0-2nm�����۵� FeO_________NiO (�� ��>������ <����=������ԭ����___________________��
(5)��֪FeCl3�ķе㣺319�棬�۵㣺306�棬��FeCl3�ľ�������Ϊ______________��
(6)��֪Fe��������ͼ��ʾ�����ֳ����ѻ���ʽ��

�����������������ܶѻ�����___________(�a����b������������Fe��a��ʽ���ܶѻ���ԭ�Ӱ뾶Ϊrpm��NA��ʾ�����ӵ�������ֵ������Fe���ܶ�Ϊ________g��cm-3 (�г���ʽ���ɣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����1���뽫����5�����ʣ�N2O��FeSO4��Fe(NO3)3��HNO3��Fe2(SO4)3�ֱ����������Ӧ�ĺ����ϣ����һ����ѧ����ʽ����ƽ��
________��________===________��________��________��________H2O
��2����Ӧ���з���������Ӧ������Ϊ________������ԭ��Ԫ����________��
��3����Ӧ��1 mol�������õ�________mol���ӡ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com