
(10��) ��0.1 mol��þ�������������100 mL 2mol/LH2SO4��Һ�У�Ȼ���ٵμ�1 mol/L NaOH��Һ����ش�
�����ڵμ�NaOH��Һ�Ĺ����У���������m�����NaOH��Һ�����V�仯����ͼ��ʾ����V1��140mLʱ,�������ĩ��n(Mg)������mol��V2������������mL��
�����ڵμ�NaOH��Һ�����У���ʹMg2����Al3���պó�����ȫ�������NaOH��Һ�����V(NaOH)��������mL��
�����������Ϊ0.1 mol������Mg�۵����ʵ�������Ϊa����100 mL 2 mol/L�������ܽ�˻������ټ���480 mL 1mol/L��NaOH��Һ�����ó�������Al(OH)3�������������a��ȡֵ��Χ�ǣ���������������������
(4)���μ�NaOH��Һ��V2mlʱ,ֹͣ�μ�NaOH��Һ����ʼ����Һ��ͨ������CO2,��д��������Ӧ�����ӷ���ʽ��������������������������������������������-����������

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(10��) ��0.1 mol��þ�������������100 mL 2mol/LH2SO4��Һ�У�Ȼ���ٵμ�1 mol/L NaOH��Һ����ش�
�����ڵμ�NaOH��Һ�Ĺ����У���������m�����NaOH��Һ����
��V�仯����ͼ��ʾ����V1��140mLʱ,�������ĩ��
n(Mg)������ mol��V2������������mL��
�����ڵμ�NaOH��Һ�����У���ʹMg2����Al3���պó�����ȫ�����
��NaOH��Һ�����V(NaOH)��������mL��
�����������Ϊ0.1 mol������Mg�۵����ʵ�������Ϊa����100 mL 2 mol/L�������ܽ�˻������ټ���480 mL 1mol/L��NaOH��Һ�����ó�������Al(OH)3�������������a��ȡֵ��Χ�ǣ���������������������
(4)���μ�NaOH��Һ��V2mlʱ,ֹͣ�μ�NaOH��Һ����ʼ����Һ��ͨ������CO2,��д��������Ӧ�����ӷ���ʽ��������������������������������������������-����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�����ʡ�����ص���ѧ��������ѧ�ȣ�������ѧ������������ѧ�Ծ����������� ���ͣ�������
(11��) ��֪������������ճ������м�Ϊ�������ᣬ��һ��������,CH3COOH��Һ�д��ڵ���ƽ�⣺CH3COOH CH3COO��+H+ ��H��0��
CH3COO��+H+ ��H��0��
(1)�����£��� pH =5��ϡ������Һ�У�c(CH3COO��)��____________(��ʽ�����ػ���)�����з����У�����ʹ0.10 mol��L��1 CH3COOH�ĵ���̶��������______?
a����������0.10 mol��L��1��ϡ���� b������CH3COOH��Һ
c����ˮϡ����0.010 mol��L��1 d����������������
e�����������Ȼ��ƹ��� f����������0.10 mol��L��1��NaOH��Һ
(2)����������пͶ��������pH������3�Ĵ����������Һ�У�������ַ�Ӧ����ֻ��һ����Һ����п��ʣ�࣬�����������������V(����)_________V(����)����Ӧ���������Ϊ��v(����)_________v(����)��(��д������������������)
(3)�����£������ΪVa mL��pHΪ3�Ĵ�����Һ�еμ�pH=11��NaOH��ҺVb mL����Һǡ�ó����ԣ���Va��Vb�Ĺ�ϵ�ǣ�__________________��
(4)�����£���0.1 mol/L�����0.1 mol/L��������Һ��ϣ�������ҺΪ���ԣ�������Һ�и����ӵ�Ũ�Ȱ��ɴ�С����Ϊ_______________________________��
(5)��֪��90��ʱ��ˮ�����ӻ�����ΪKw = 3.8��10��13���ڴ��¶��£���pH=3�������pH = 11������������Һ�������ϣ�������Һ�е�c(H+)=______________(������λ��Ч����)mol/L��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ��������ѧ�ȣ�������ѧ������������ѧ�Ծ��������棩 ���ͣ�������
(11��) ��֪������������ճ������м�Ϊ�������ᣬ��һ��������,CH3COOH��Һ�д��ڵ���ƽ�⣺CH3COOH CH3COO��+H+ ��H��0��
CH3COO��+H+ ��H��0��
(1)�����£��� pH =5��ϡ������Һ�У�c(CH3COO��)��____________(��ʽ�����ػ���)�����з����У�����ʹ0.10 mol��L��1 CH3COOH�ĵ���̶��������______。
a����������0.10 mol��L��1��ϡ���� b������CH3COOH��Һ
c����ˮϡ����0.010 mol��L��1 d����������������
e�����������Ȼ��ƹ��� f����������0.10 mol��L��1��NaOH��Һ
(2)����������пͶ��������pH������3�Ĵ����������Һ�У�������ַ�Ӧ����ֻ��һ����Һ����п��ʣ�࣬�����������������V(����)_________V(����)����Ӧ���������Ϊ��v(����)_________v(����)��(��д������������������)
(3)�����£������ΪVa mL��pHΪ3�Ĵ�����Һ�еμ�pH=11��NaOH��ҺVb mL����Һǡ�ó����ԣ���Va��Vb�Ĺ�ϵ�ǣ�__________________��
(4)�����£���0.1 mol/L�����0.1 mol/L��������Һ��ϣ�������ҺΪ���ԣ�������Һ�и����ӵ�Ũ�Ȱ��ɴ�С����Ϊ_______________________________��
(5)��֪��90��ʱ��ˮ�����ӻ�����ΪKw = 3.8��10��13���ڴ��¶��£���pH=3�������pH = 11������������Һ�������ϣ�������Һ�е�c(H+)=______________(������λ��Ч����)mol/L��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014�콭��ʡ��һ�ڶ����¿���ѧ�Ծ� ���ͣ������
(10��) ��0.1 mol��þ�������������100 mL 2mol/LH2SO4��Һ�У�Ȼ���ٵμ�1 mol/L NaOH��Һ����ش�
�����ڵμ�NaOH��Һ�Ĺ����У���������m�����NaOH��Һ����
��V�仯����ͼ��ʾ����V1��140mLʱ,�������ĩ��
n(Mg)������ mol��V2������������mL��
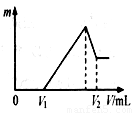
�����ڵμ�NaOH��Һ�����У���ʹMg2����Al3���պó�����ȫ�����
��NaOH��Һ�����V(NaOH)��������mL��
�����������Ϊ0.1 mol������Mg�۵����ʵ�������Ϊa����100 mL 2 mol/L�������ܽ�˻������ټ���480 mL 1mol/L��NaOH��Һ�����ó�������Al(OH)3�������������a��ȡֵ��Χ�ǣ���������������������
(4)���μ�NaOH��Һ��V2mlʱ,ֹͣ�μ�NaOH��Һ����ʼ����Һ��ͨ������CO2,��д��������Ӧ�����ӷ���ʽ��������������������������������������������-����������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com