

 Fe3O4+4H2£¬ŅŌ“Ė½ā“šøĆĢā£®
Fe3O4+4H2£¬ŅŌ“Ė½ā“šøĆĢā£® Fe3O4+4H2£¬
Fe3O4+4H2£¬ Fe3O4+4H2£¬
Fe3O4+4H2£¬ Fe3O4+4H2£®
Fe3O4+4H2£®

 ŌĶĮæģ³µĻµĮŠ“š°ø
ŌĶĮæģ³µĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| ||
| ||
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
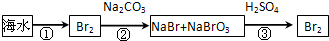
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014½ģÕć½Ź”ĪĀÖŻŹŠŹ®Š£ĮŖŗĻĢåøßČżÉĻѧʌʌ֊ĮŖæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
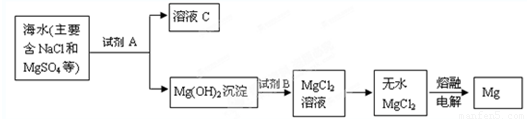
“Óŗ£Ė®ÖŠĢįČ”Ć¾ŹĒŹĄ½ēø÷¹ś»ńµĆĆ¾µÄÖ÷ŅŖĄ“Ō“£¬ŅŌĻĀŹĒÖĘČ”Ć¾µÄ¹ż³ĢÖŠÉę¼°µÄ¼øÖÖĪļÖŹµÄČܶȻż£¬øł¾ŻÄćĖłŃ§µÄÖŖŹ¶»Ų“šĻĀĮŠ¼øøöĪŹĢā£ŗ
|
ĪļÖŹ |
CaCO3 |
MgCO3 |
Ca(OH)2 |
Mg(OH)2 |
|
ČܶȻż |
2.8”Į10ØC9 |
6.8”Į10ØC6 |
5.5”Į10ØC6 |
1.8”Į10ØC11 |
(1)ŌŚ“Óŗ£Ė®ÖŠĢįČ”Ć¾Ź±£¬ĶłĶłÓƵ½±“æĒ(Ö÷ŅŖ³É·ŻŹĒĢ¼ĖįøĘ)£¬ÄćČĻĪŖ (Ģī”°ÄÜ”±»ņ”°²»ÄÜ”±)½«±“æĒŃŠÄ„³É·ŪÄ©Ö±½ÓĶ¶Čėŗ£Ė®ÖŠ£¬ĄķÓÉŹĒ £»Čē¹ū²»ÄÜÖ±½ÓĶ¶Čė£¬Ó¦½«±“æĒ×÷ŗĪ“¦Ąķ£¬ŹŌŠ“³ö»Æѧ·½³ĢŹ½ ”£
(2)ijĶ¬Ń§ŌŚŹµŃéŹŅĢõ¼žĻĀÄ£ÄāøĆÉś²ś¹ż³Ģ£¬µ«ŹµŃéŹŅĪŽŹÆ»Ņ£¬Ėū“ņĖćÓĆÉÕ¼ī“śĢę£¬ÄćČĻĪŖ (Ģī”°æÉŅŌ”±»ņ”°²»æÉŅŌ”±)µĆµ½Mg(OH)2”£ŌŚ¼ÓŹŌ¼ĮŹ±£¬Ī󽫓æ¼īČÜŅŗ¼ÓČėŗ£Ė®ÖŠ£¬ĖūĖ¼æ¼ĮĖŅ»ĻĀ£¬ÓÖŌŚµĆµ½µÄ»ģŗĻĢåĻµÖŠ¼ÓČė¹żĮæµÄÉÕ¼īČÜŅŗ£¬Äć¾õµĆĖū (Ģī”°ÄÜ”±»ņ”°²»ÄÜ”±)½«Mg2+×Ŗ»ÆĪŖMg(OH)2³Įµķ£¬ĄķÓÉŹĒ (ÓĆĄė×Ó·½³ĢŹ½±ķŹ¾)”£
(3)ŅŃÖŖŗ£Ė®ÖŠĆ¾Ąė×ÓÅضČĪŖ1.8”Į10ØC3mol”¤LØC1£¬ŌņŅŖŹ¹Ć¾Ąė×Ó²śÉś³Įµķ£¬ČÜŅŗµÄPH×īµĶÓ¦ĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015½ģ±±¾©ŹŠ¶«³ĒĒųøßŅ»ĻĀŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŹµŃéĢā
“Óŗ£Ė®ĢįČ”Ć¾ŗĶäåµÄĮ÷³ĢČēĻĀ£¬Ēė»Ų“šĻą¹ŲĪŹĢā”£
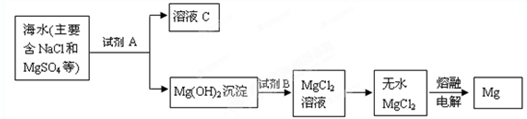
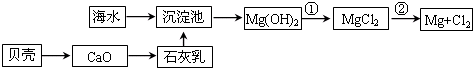
£Ø1£©“Óŗ£Ė®ÖŠĢįČ”Ć¾µÄĮ÷³ĢČēĻĀĶ¼ĖłŹ¾£ØæņĶ¼ÖŠŹĒÖ÷ŅŖ²śĪļ£©£ŗ
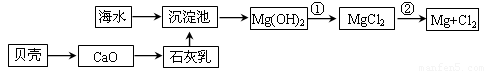
i.ŗ£Ė®½ųČė³Įµķ³ŲĒ°£¬¶Ōŗ£Ė®½ųŠŠ“¦ĄķÓŠŅŌĻĀĮ½ÖÖ·½°ø£ŗ
·½°øŅ»£ŗ½«É¹ŃĪŗóµÄĀ±Ė®ĶØČė³Įµķ³Ų£»
·½°ø¶ž£ŗ½«¼ÓČČÕō·¢ÅØĖõŗóµÄŗ£Ė®ĶØČė³Įµķ³Ų”£
ÄćČĻĪŖ·½°ø________øüŗĻĄķ£¬ĄķÓÉŹĒ_________________________________________”£
ii.·“Ó¦¢ŁµÄĄė×Ó·½³ĢŹĒ________________________________________________”£
·“Ó¦¢ŚµÄ»Æѧ·½³ĢŹ½ŹĒ_______________________________________________”£
£Ø2£©“Óŗ£Ė®ÖŠĢįČ”äåµÄĮ÷³ĢČēĻĀĶ¼ĖłŹ¾£ØæņĶ¼ÖŠŹĒÖ÷ŅŖ²śĪļ£©£ŗ


i.¹ż³Ģ¢ŁÖŠ£¬¼ÓČėµÄŹŌ¼ĮŹĒ___________”£
ii.¹ż³Ģ¢ŚÖŠ£¬Ļņ»ģŗĻŅŗÖŠ“µČėČČæÕĘų£¬½«ä哵³ö£¬ÓĆ“æ¼īĪüŹÕ£¬“µČėČČæÕĘųµÄÄæµÄŹĒ______________________________________________________________________”£
iii.¹ż³Ģ¢ŪÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ____________________________________________”£
iv.Čō×īÖÕµĆµ½µÄäåµ„ÖŹÖŠČŌČ»»ģÓŠÉŁĮæµÄCl2£¬Ōņ³żČ„øĆŌÓÖŹµÄ·½·ØŹĒ__________________________________________________£Ø½įŗĻĄė×Ó·½³ĢŹ½»Ų“š£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014½ģø£½ØŹ””¢¶žÖŠø߶žÉĻѧʌʌĩĮŖæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ÄæĒ°ŹĄ½ēø÷¹ś»ńµĆĆ¾µÄÖ÷ŅŖĄ“Ō““Óŗ£Ė®ÖŠĢįČ”£¬ŅŌĻĀŹĒĢįČ”Ć¾µÄ¹ż³ĢÖŠÉę¼°µ½µÄ¼øÖÖĪļÖŹ³£ĪĀĻĀµÄČܶȻż³£Źż£¬øł¾ŻÄćĖłŃ§µÄÖŖŹ¶»Ų“šĻĀĆęµÄ¼øøöĪŹĢā£ŗ?
|
ĪļÖŹ |
CaCO3 |
MgCO3 |
Ca(OH)2 |
Mg(OH)2 |
|
ČܶȻż |
2.8”Į10-9 |
6.8”Į10-6 |
5.5”Į10-6 |
1.8”Į10-11 |
(1)ŌŚ“Óŗ£Ė®ÖŠĢįČ”Ć¾Ź±£¬ĶłĶłÓƵ½±“æĒ£ØÖ÷ŅŖ³É·ÖŹĒCaCO3£©£¬ÄćČĻĪŖ (Ģī”°ÄÜ”±»ņ”°²»ÄÜ”±)Ķعż½«±“æĒŃŠÄ„³É·ŪÄ©Ö±½ÓĶ¶Čėŗ£Ė®ĄļÖʱøŗ¬Ć¾µÄ³Įµķ£¬ĄķÓÉŹĒ ”£Čē¹ū²»ÄÜÖ±½ÓĶ¶Čė£¬Ó¦½«±“æĒ×÷ŗĪ“¦Ąķ£¬ŹŌŠ“³ö»Æѧ·½³ĢŹ½£ŗ ”££ØČōµŚŅ»øöæÕøńĢī”°ÄÜ”±£¬“ĖæÕøń²»Ģī£»ČōĢī”°²»ÄÜ”±£¬ “ĖæÕøńÖ»ŠėµŚŅ»²½“¦ĄķµÄ»Æѧ·“Ó¦·½³ĢŹ½”££©
£Ø2£©ŅŃÖŖijµŲŗ£Ė®ÖŠµÄĆ¾Ąė×ÓµÄÅضČĪŖ1.8”Į10-3 mol”¤L-1,Ōņ³£ĪĀĻĀŅŖŹ¹Ć¾Ąė×Ó²śÉś³Įµķ£¬ČÜŅŗpH×īµĶÓ¦ĪŖ ”£
£Ø3£©ŹµŃéŹŅÖŠ³£ÓĆCaCO3ÖĘCO2£¬Ęä²śĪļÖ®Ņ»µÄĀČ»ÆøĘŹĒÓ¦ÓĆ¹ć·ŗµÄ»ÆѧŹŌ¼Į£¬æÉ×÷øÉŌļ¼Į”¢Ąä¶³¼ĮµČ”£ĪŖĮĖ²ā¶ØijĀČ»ÆøĘѳʷ֊øĘŌŖĖŲµÄŗ¬Į棬½ųŠŠČēĻĀŹµŃé£ŗ
£ØI£©×¼Č·³ĘČ”ĀČ»ÆøĘѳʷ0.2000g£¬·ÅČėÉÕ±ÖŠ£¬¼ÓČėŹŹĮæ6mol/LµÄŃĪĖįŗĶŹŹĮæÕōĮóĖ®Ź¹ŃłĘ·ĶźČ«Čܽā£¬ŌŁ¼ÓČė35mL 0.25mol/L £ØNH4£©2C2O4ČÜŅŗ£¬Ė®Ō”¼ÓČČ£¬Öš½„Éś³ÉCaC2O4³Įµķ£¬¾¼ģŃ飬Ca2+ŅŃ³ĮµķĶźČ«”£
£ØII£©¹żĀĖ²¢Ļ“µÓ£ØI£©ĖłµĆ³Įµķ”£
£ØIII£©¼ÓČė×ćĮæµÄ10% H2SO4ČÜŅŗŗĶŹŹĮæµÄÕōĮóĖ®£¬£ØII£©ÖŠ³ĮµķĶźČ«Čܽā£¬ČÜŅŗ³ŹĖįŠŌ£¬¼ÓČČÖĮ75”ę£¬³ĆČČÖšµĪ¼ÓČė0.05000 mol/L KMnO4ČÜŅŗ16.00mL£¬Ē”ŗĆĶźČ«·“Ó¦”£Ēė»Ų“š£ŗ
ŅŃÖŖµĪ¶Ø¹ż³Ģ·¢ÉśµÄ·“Ó¦ĪŖ2MnO4- + 5H2C2O4 + 6H+ ="=2" Mn2+ +10 CO2”ü+8 H2O£ØŅŃÅäĘ½£©
¢Ł0.05000 mol/L KMnO4ČÜŅŗ±ź×¼ČÜŅŗÓ¦ÖĆÓŚ £ØŃ”Ģī”°ĖįŹ½”±»ņ”°¼īŹ½”±£©µĪ¶Ø¹ÜÖŠ”£
¢ŚµĪ¶ØÖÕµćµÄĻÖĻóĪŖ ”£
¢ŪøĆĀČ»ÆøĘѳʷ֊øĘŌŖĖŲµÄÖŹĮæ°Ł·ÖŹżĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com