�������ƣ�NaClO���Ͳ������ƣ�NaClO��������Ҫ�ĺ�����������
��l�� NaClO�У���Ԫ�صĻ��ϼ�Ϊ
��
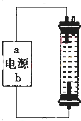
��2��ijͬѧ��ʵ����̽��������һ�ּ��û���������Һ��NaClO�����������������ͼ��װ�ã���ʯī���缫��ⱥ���Ȼ�����Һ��
��ͨ��ʱ��ΪʹCl
2����ȫ���գ��Ƶ��н�ǿɱ������������Һ�����Դ��b�缫����Ϊ
�������������������������������Ͷ��
��װ���з�����Ӧ���йػ�ѧ����ʽΪ
����ֵ�����Һ�и�����Ũ���ɴ�С˳��Ϊ
��
����˵��ʹ�ø�װ����������Һ��һ���ŵ��һ��ע������
��
��3����ѧ����С��Ϊ�ⶨijƷ�ơ�84����Һ����NaClO�ĺ�������λ�����Һ����NaCIO������������������ʵ�飮��֪��ClO
-+2I
-+2H
+=Cl
-+I
2+H
2O I
2+2S
2O
32-=2I
-+S
4O
62-ȡ10mL������Һ����ƿ�У�Ѹ�ټ��������������ữ�ĵ⻯����Һ���ܷ⡢��ֻ�ϣ��������ü����ӣ�Ȼ����뼸�ε�����Һ����c mol/L����������ƣ�Na
2S
2O
3������Һ�ζ���ƽ����ȥVmL�����������Һ����á�84����Һ����NaCIO�ĺ���Ϊ
g/mL���ú�c��V��ʽ�ӱ�ʾ����
��4�������ǹ������ⷨ�����������ƵĹ�������ͼ��

��֪����i��NaClO
2 ���ܽ�����¶����߶������ʵ������¿ɽᾧ������NaClO
2.3H
2O��
��ii����ClO
2�ֽⱬը��һ����ϡ����������ϡ�͵�10%���½ϰ�ȫ��
��iii�� HClO
2�ɿ�����ǿ�ᣮ
�ٷ������з�����Ӧ�����ӷ���ʽΪ
�ڷ������й�����������ÿ�����
��
���������ڵķ�Ӧ�Ļ�ѧ����ʽΪ
������������Ϊ��ֹ����ԭ��NaCI�����û�ԭ���Ļ�ԭ��Ӧ���У����⣬������ѡ��Ļ�ԭ��
������ţ���
A��Na
2O
2 B��Na
2S C��FeCl
2�۴���Һ�еõ�NaClO
2.3H
2O�־����ʵ�����������
������ţ���
A������ B������ C������ D������ E����ȴ�ᾧ��

 �������ƣ�NaClO���Ͳ������ƣ�NaClO��������Ҫ�ĺ�����������
�������ƣ�NaClO���Ͳ������ƣ�NaClO��������Ҫ�ĺ�����������



