
��13�֣�![]() ����������Ҫ�ɷ���
����������Ҫ�ɷ���![]() ����
����![]() ��
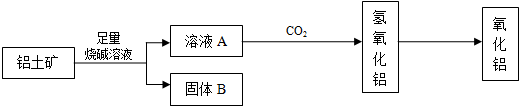
��![]() ��MgO�����ʣ�����ȡ���ֹ���Ʒ���������£�
��MgO�����ʣ�����ȡ���ֹ���Ʒ���������£�

��ش��������⣺
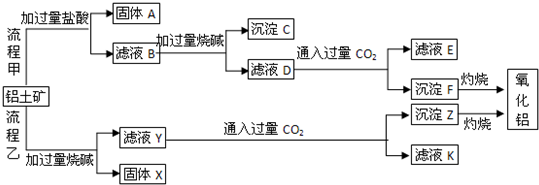
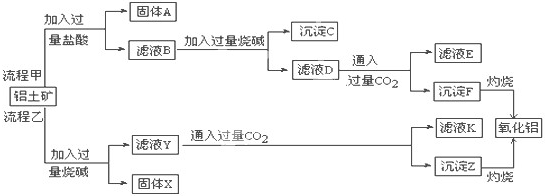
��1�����̼������������Al3+�ķ���ʽΪ_________.
��2�������Ҽ����ռ������Si![]() �����ӷ���ʽΪ________.
�����ӷ���ʽΪ________.
��3����֤��ҺB��![]() ����ȡ������Һ������________�����Լ����ƣ���
����ȡ������Һ������________�����Լ����ƣ���
��4����ҺE��K�����ʵ���Ҫ�ɷ���________(�ѧʽ)��д������Һ��һ����;________
��5����֪298Kʱ��![]() ���ݶȻ�����
���ݶȻ�����![]() =5.6��
=5.6��![]() ,ȡ��������ҺB,����һ�������ռ�ﵽ������Һƽ�⣬���PH=13.00������¶��²�������Һ�е�
,ȡ��������ҺB,����һ�������ռ�ﵽ������Һƽ�⣬���PH=13.00������¶��²�������Һ�е�![]() =_______.
=_______.
��1��Al2O3 + 6H�� ![]() 2Al3�� +3H2O
2Al3�� +3H2O
��2��SiO2 + 2OH�� ![]() SiO32 �� + H2O
SiO32 �� + H2O
��3�����軯�أ���������ء�������Һ�Ⱥ����𰸣���
��4��NaHCO3���ƴ���������ͷ۵Ⱥ����𰸣�
��5��5.6��10-10mol/L
���������⿼������������������ȡ�Ĺ������̡���1����HCl��Ӧ����Al3����ӦΪ��������Al2O3����2��SiO2��������NaOH������Na2SiO3����3������Fe3����������Ӧ�ܶ࣬����KSCN��Ѫ��ɫ���뱽������ɫ����OH���Ժ��ɫ�����ȡ���4������E��Kʱ��CO2���ǹ����ģ���Ӧ����NaHCO3����5��![]() ��
��![]() =5.6��10-12/(0.1)2=5.6��10-10��
=5.6��10-12/(0.1)2=5.6��10-10��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
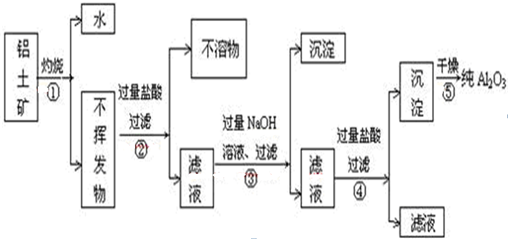
| A���ܢ� | B���ۢ� | C���ڢ� | D���٢� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com