
ij���Ͻ�(Ӳ��)�к���þ��ͭ���裬Ϊ�˲ⶨ�úϽ������ĺ����������������ʵ�飺
(1)ȡ��Ʒa g����ȡʱʹ�õ���������Ϊ ��
(2)����Ʒ��������ϡ�����У����ˣ���Һ����Ҫ���� �������к��� �����ܽ����ʱʹ�õ���Ҫ���������� ��
(3)����Һ�м������NaOH��Һ�����ˣ�д���ò��������йصĻ�ѧ����ʽ ��
(4)�ڵ�(3)������Һ��ͨ������CO2�����ˣ�������������ˮϴ�����κ�ɲ��������������ټ���Ϊֹ����ȴ�����������Ϊb g���йط�Ӧ�Ļ�ѧ����ʽΪ ��
(5)����Ʒ���������������ı���ʽΪ ��
 �ܿ���ȫ��100��ϵ�д�
�ܿ���ȫ��100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��15�֣���FeCl3������Һ�ѳ�H2S��ķ�Һ��ͨ�����Ƶ�ѹ������������ijͬѧʹ��ʯī�缫���ڲ�ͬ��ѹ��x���µ��pH=1��0.1mol/LFeCl2��Һ���о���Һ������������¼���£�a��b��c������ѹֵ����
| ��� | ��ѹ/V | �������� | ������������ |
| I | x��a | �缫�������ֻ�ɫ�������ݲ��� | ��Fe3+����Cl2 |
| II | a��x��b | �缫�������ֻ�ɫ�������ݲ��� | ��Fe3+����Cl2 |
| III | b��x��0 | �����Ա仯 | ��Fe3+����Cl2 |
| ��� | ��ѹ/V | �������� | ������������ |
| IV | a��x��c | �����Ա仯 | ��Cl2 |
| V | c��x��b | �����Ա仯 | ��Cl2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�п��Ľ���Na��¶�ڿ����У���仯�������£�
��1����Ӧ��ķ�Ӧ�����������仯�Ĺ�ϵ���£�
�� ��Ӧ�� ���� ����Ӧ������ȡ������ȡ������ж���������������
�� 1 mol Na(s)ȫ��������Na2O(s)���Ȼ�ѧ����ʽ����������
��2����Ӧ����Na2O��ˮ�ķ�Ӧ�������ĵ���ʽ�� ��
��3����ɫ��ĩΪNa2CO3����������ˮ����Ϊ0.1 mol/L Na2CO3��Һ������˵����ȷ���� ������ĸ����
| A�������¶ȣ���Һ��pH���� |
| B��c(OH��)��c (H��)��c (HCO3��)��2 c (H2CO3) |
| C����������NaOH���壬c (CO32�D)��c (Na��)������ |
| D��c (Na��) > c (CO32�D) > c (HCO3�D) > c(OH�D) > c (H��) |
 Ni + 2NaCl����������Ӧʽ��_____��
Ni + 2NaCl����������Ӧʽ��_____���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������ͭ�Ƚ������仯�������ճ�������Ӧ�ù㷺�����������ʵ��ش����⡣
(1)�����к���һ����̼������X(Fe3C)��X�������Ŀ����и������գ������д��ԵĹ���Y����Y���ڹ����������Һ�д������ڵ��������� ��Y�����Ũ���ᷴӦ����Һ�к��е��εĻ�ѧʽΪ ��
(2)ij��Һ����Mg2����Fe2����Al3����Cu2�������ӣ������м��������NaOH��Һ���ˣ��������������գ��������պ�Ĺ���Ͷ�������ϡ�����У�������Һ��ԭ��Һ��ȣ���Һ�д������ٵ��������� ��
A��Mg2�� B��Fe2�� C��Al3�� D��Cu2��
(3)����������Ҫ�Ĺ�ҵ���ϣ��÷���м�Ʊ������������£�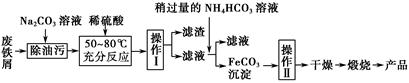
�ش��������⣺
�ٲ������������ ��������������� ��������ķ���Ϊ ��
����д������FeCO3���������ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������Ļ�����Ӧ�ù㷺����FeCl3������������ӡˢ��·ͭ�帯ʴ��������ֹѪ���ȡ�
(1)д��FeCl3��Һ��ʴӡˢ��·ͭ������ӷ���ʽ_______________________
(2)����(1)�еķ�Ӧ��Ƴ�ԭ��أ��뻭��ԭ��ص�װ��ͼ�����������������д���缫��Ӧʽ��
������Ӧ_______________________________
������Ӧ_______________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�о���ѧϰС���Na2O2���й�����̽�����£�����з�̪��ˮ��
Ͷ��һ������Na2O2���۲쵽���д������ݲ�������Һ��죬��һ�����Һ�ֱ�Ϊ��ɫ��
������ʵ������Һ��죬��һ�������ɫ��ԭ�ס�����ͬѧ����˲�ͬ�Ľ��ͣ�
��ͬѧ��Ϊ��Na2O2��ˮ��Ӧ�ų������������������ԣ�����̪������ʹ��Һ��ɫ��
��ͬѧ����Ϊ��Na2O2��ˮ��Ӧʱ������H2O2��H2O2��ǿ������ʹ��̪��ɫ��
(1)��ͬѧ���������ʵ����֤���Լ��IJ�������ȷ�ģ��ڵ��з�̪������������Һ�еμ�3%��H2O2��Һ����
������ͬѧ�IJ�����ȷ���ɹ۲쵽�������� ��
�ڼ�ͬѧ�����ͬѧ��������֤ʵ�������Ӧ����ʵ����ܸ�ֱ��֤��Na2O2��ˮ��Ӧ������H2O2����ͬѧ���Ҫ���ӵ�ʵ����ʲô��
(2)����Ƽ�ʵ��֤����ͬѧ�Ľ����Ƿ���ȷ(��������װ��ͼ��˵����Ҫ����)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1�����µ�⼼���ܸ�Чʵ��CO2(g) + H2O(g) ="CO(g)" + H2(g) +O2(g) ������ԭ��ʾ��ͼ���£�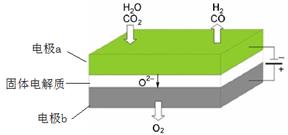
�ٵ缫b���� �����������ԭ������Ӧ��
��CO2�ڵ缫a�ŵ�ķ�Ӧʽ�� ��
��2����ҵ����ij����������Cu2O��Al2O3��Fe2O3��SiO2����ȡͭ�IJ����������£�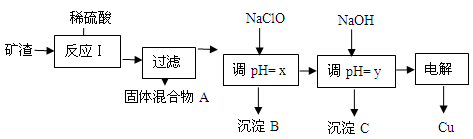
��֪�� Cu2O + 2H+ =" Cu" + Cu2+ + H2O
| ������ | Cu(OH)2 | Al(OH)3 | Fe(OH)3 | Fe(OH)2 |
| ��ʼ����pH | 5.4 | 4.0 | 1.1 | 5.8 |
| ������ȫpH | 6.7 | 5.2 | 3.2 | 8.8 |
 HClO��ClO������H+���Ӷ��ﵽ����pH��Ŀ��
HClO��ClO������H+���Ӷ��ﵽ����pH��Ŀ�� HClO+OH����OH������H+ ���Ӷ��ﵽ����pH��Ŀ��
HClO+OH����OH������H+ ���Ӷ��ﵽ����pH��Ŀ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������仯������������������з�������Ҫ�����á�
��1����һС�������Ͷ��ʢ��ˮ���ձ��У�������Ӧ�Ļ�ѧ����ʽΪ ��
��2���ơ����������ֽ���Ԫ�����γɵĸ����������о���ǿ�����Ե��� ���ѧʽ����ͬ�����������ͻ���ϵ��� ��
��3�����������������ᷴӦ���������ռ���Һ��Ӧ����д�����������ռ���Һ��Ӧ�����ӷ���ʽ�� ��
��4��ȡ����FeCl2��Һ����ͼ��ʾ������ʹ���NaOH��Һ��Ӧ�� �ɹ۲쵽�Թ��г��ְ�ɫ��״�������̶���ɻ���ɫ������ɺ��ɫ����д��������������صķ�Ӧ�Ļ�ѧ����ʽ�� �� ��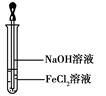
��5����ʢ���Ȼ������Ȼ��������Ȼ�ͭ�����Һ���ձ��м������ۺ�ͭ�ۣ���Ӧ���������н�������ܳ��ֵ��� ������ţ���
| A����ͭ���� | B��������ͭ | C������ͭ���� | D������ͭ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ִ�ѭ������Ҫ���ۺϿ��ǻ�����Ⱦ�;���Ч�档�ߴ������������ִ����ӹ�ҵ�IJ��ϣ�
����������������������Ҫ�ɷ�ΪFe2O3��Fe3O4��FeO��SiO2��Ϊԭ���Ʊ��ߴ������������Ŧ�Fe2O3������������ʾ��ͼ��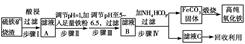
��1��������й���������������Ҫ�ɷ���________��
��2��������м������۵�Ŀ����________��
��3��������п�ѡ��________������Һ��pH��
| A��ϡ���� | B��˫��ˮ | C����ˮ | D�����������Һ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com