
| ������ | K����Na����Fe2����Ba2����NH4+ |
| ������ | OH����NO3����I����HCO3����AlO2����HSO4�� |
 BaSO4����NH3����2H2O
BaSO4����NH3����2H2O BaSO4����NH3����2H2O��
BaSO4����NH3����2H2O��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Al3+��NO3�D��K+��SO42�� | B��Ca2+��H+��CO32����AlO2- |
| C��OH����SO42����NH4+��Al3+ | D��K+��Ca2+��HCO3-��OH- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ������ | K����Na����Fe2����Ba2����NH4+��Ca2�� |
| ������ | OH����NO3����I����HCO3����AlO2����HSO4�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ���ܴ������е������� | H+��Ag+��Mg2+��Al3+��NH4+��Fe3+ |
| ���ܴ������е������� | Cl-��Br-��I-��CO32����AlO2�� |
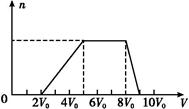
| Cl2�����(��״��) | 11.2 L | 22.4 L | 28.0 L |
| n(Cl-) | 2.5 mol | 3.5 mol | 4.0 mol |
| n(Br-) | 3.0 mol | 2.5 mol | 2.0 mol |
| n(I-) | x mol | 0 | 0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 HSO3-��H���ĵ��볣��Ka��1��10��2mol��L��1������¶���NaHSO3ˮ�ⷴӦ��ƽ�ⳣ��Kb��________mol��L��1������NaHSO3��Һ�м���������I2������Һ��
HSO3-��H���ĵ��볣��Ka��1��10��2mol��L��1������¶���NaHSO3ˮ�ⷴӦ��ƽ�ⳣ��Kb��________mol��L��1������NaHSO3��Һ�м���������I2������Һ��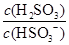 ��________(���������С�����䡱)��
��________(���������С�����䡱)���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
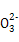 ��OH- 6�������е�3��,��֪����Һ����ɫ,������Һ�д������ڵ������ǣ� ��
��OH- 6�������е�3��,��֪����Һ����ɫ,������Һ�д������ڵ������ǣ� ��A��K+��OH-��C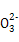 | B��Cu2+��H+��Cl- |
| C��K+��H+��Cl- | D��C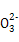 ��OH-��Cl- ��OH-��Cl- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ƽ�ⳣ���Ĵ�С���¶ȡ�Ũ�ȡ�ѹǿ���������й� |
| B�����¶�����ʱ������ĵ���ƽ�ⳣ��Ka��С |
| C��Ksp(AgCl)>Ksp(AgI)���ɴ˿����ж�AgCl(s)��I��(aq)=AgI(s)��Cl��(aq)�ܹ����� |
| D��Ka(HCN)<Ka(CH3COOH)��˵�����ʵ���Ũ����ͬʱ������������Աȴ���ǿ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 H����A2����
H����A2�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com