
ŅŃÖŖ25”ꏱ²æ·ÖČõµē½āÖŹµÄµēĄėĘ½ŗā³£ŹżŹż¾ŻČēĻĀ±ķ£ŗ
| »ÆѧŹ½ | CH3COOH | H2CO3 | HClO | |
| Ę½ŗā³£Źż | Ka=1.8”Į10-5 | Ka1=4.3”Į10-7 | Ka2=5.6”Į10-11 | Ka=3.0”Į10-8 |
»Ų“šĻĀĮŠĪŹĢā£ŗ
(1) ĪļÖŹµÄĮæÅØ¶Č¾łĪŖ0£®1mol”¤LŅ»1µÄĖÄÖÖČÜŅŗ£ŗ a£®CH3COONa b£®Na2CO3 c£®NaClO d£®NaHCO3£¬pHÓÉŠ”µ½“óµÄÅÅĮŠĖ³ŠņŹĒ___________ (ÓĆ×ÖÄø±ķŹ¾)
(2) ³£ĪĀĻĀ£¬0£®1mol”¤L”Ŗ1µÄCH3COOHČÜŅŗ¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠ£¬ĻĀĮŠ±ķ“ļŹ½µÄŹż¾Ż±ä“óµÄŹĒ_______________(ĢīŠņŗÅ)
 A£®c£ØH+£© B£®c(H+)£Æc(CH3COOH)
A£®c£ØH+£© B£®c(H+)£Æc(CH3COOH)
C£®c(H+)”¤c(OH”Ŗ) D£®c(OH-)£Æc(H+)
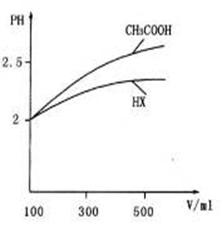
(3) Ģå»ż¾łĪŖl00mL pH=2µÄCH3COOHÓėŅ»ŌŖĖįHX£¬¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠpHÓėČÜŅŗĢå»żµÄ¹ŲĻµČēĶ¼ĖłŹ¾£¬ŌņHXµÄµēĄėĘ½ŗā³£Źż_______ (Ģī”°£¾”±”¢”°£¼”±»ņ”°=”±)CH3COOHµÄµēĄėĘ½ŗā³£Źż”£
(4) 25”ꏱ£¬ŌŚCH3COOHÓėCH3COONaµÄ»ģŗĻČÜŅŗÖŠ£¬Čō²āµĆpH=6£¬ŌņČÜŅŗÖŠ£ŗ
¢Ł c(CH3COO”Ŗ)”Ŗc(Na+)=_______ mol”¤L”Ŗ1(Ģī¾«Č·Öµ)
¢Ś c(CH3COO-)£Æc(CH3COOH)=_______ ”£
 ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø
ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| »ÆѧŹ½ | CH3COOH | H2CO3 | HClO | |
| Ę½ŗā³£Źż | Ka=1.8”Į10-5 | Ka1=4.3”Į10-7 | Ka2=5.6”Į10-11 | Ka=3.0”Į10-8 |

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
 ŅŃÖŖ25”ꏱ²æ·ÖČõµē½āÖŹµÄµēĄėĘ½ŗā³£ŹżŹż¾ŻČēĻĀ±ķ£ŗ
ŅŃÖŖ25”ꏱ²æ·ÖČõµē½āÖŹµÄµēĄėĘ½ŗā³£ŹżŹż¾ŻČēĻĀ±ķ£ŗ| »ÆѧŹ½ | CH3COOH | H2CO3 | HClO | |
| Ę½ŗā³£Źż | Ka=1.8”Į10-5 | Ka1=4.3”Į10-7 | Ka2=5.6”Į10-11 | Ka=3.0”Į10-4 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŅŃÖŖ25”ꏱ²æ·ÖČõµē½āÖŹµÄµēĄėĘ½ŗā³£ŹżŹż¾ŻČē±ķĖłŹ¾£ŗ
| »ÆѧŹ½ | CH3COOH | H2CO3 | HClO | |
| µēĄėĘ½ŗā³£Źż | Ka£½1.8”Į10£5 | Kal£½4.3”Į10£7 | Ka2£½5.6”Į10£11 | Ka£½3.0”Į10£8 |
»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)ĪļÖŹµÄĮæÅØ¶Č¾łĪŖ0.1mol”¤L£1µÄĖÄÖÖČÜŅŗ£»
a£®CH3COONa b£®Na2CO3 c£®NaClO d£®NaHCO3
pHÓÉŠ”µ½“óÅÅĮŠµÄĖ³ŠņŹĒ_______________(ÓƱąŗÅĢīŠ“)”£
(2)³£ĪĀĻĀ£¬0.1mol”¤L£1CH3COOHČÜŅŗ¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠ£¬ĻĀĮŠ±ķ“ļŹ½µÄŹż¾Ż±ä“óµÄŹĒ ”£
A£®c(H£«) B£®c(H£«)/c(CH3COOH) C£®c(H£«)”¤c(OH£)
D£®c(OH£)/c(H£«) E£®c(H£«)”¤c(CH3COO£)/c(CH3COOH)
(3)Ģå»ż¾łĪŖ100mL pH£½2µÄCH3COOHÓėŅ»ŌŖĖįHX£¬¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠpHÓėČÜŅŗĢå»żµÄ¹ŲĻµČēĶ¼ĖłŹ¾£¬ŌņHXµÄµēĄėĘ½ŗā³£Źż_____(Ģī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±)CH3COOHµÄµēĄėĘ½ŗā³£Źż£¬ĄķÓÉŹĒ_____________________”£

(4)25”ꏱ£¬CH3COOHÓėCH3COONaµÄ»ģŗĻČÜŅŗ£¬Čō²āµĆpH£½6£¬ŌņČÜŅŗÖŠ
c(CH3COO£)£c(Na£«)£½ mol”¤L£1(Ģī¾«Č·Öµ)”£
(5)±ź×¼×“æöĻĀ£¬½«1.12L CO2ĶØČė100mL 1mol”¤L£1µÄNaOHČÜŅŗÖŠ£¬ÓĆČÜŅŗÖŠĪ¢Į£µÄÅØ¶Č·ūŗÅĶź³ÉĻĀĮŠµČŹ½£ŗ
¢Łc (OH£)£½2c(H2CO3)£« £»
¢Śc(H£«)£«c(Na£«)£½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014½ģ½ĖÕŹ”ø߶ž5ŌĀѧ·ÖČĻ¶ØÄ£æé¼ģ²ā»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖ25”ꏱ²æ·ÖČõµē½āÖŹµÄµēĄėĘ½ŗā³£ŹżŹż¾ŻČē±ķĖłŹ¾£ŗ
|
»ÆѧŹ½ |
CH3COOH |
H2CO3 |
HClO |
|
|
µēĄėĘ½ŗā³£Źż |
Ka£½1.8”Į10£5 |
Kal£½4.3”Į10£7 |
Ka2£½5.6”Į10£11 |
Ka£½3.0”Į10£8 |
»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)ĪļÖŹµÄĮæÅØ¶Č¾łĪŖ0.1mol”¤L£1µÄĖÄÖÖČÜŅŗ£»
a£®CH3COONa b£®Na2CO3 c£®NaClO d£®NaHCO3
pHÓÉŠ”µ½“óÅÅĮŠµÄĖ³ŠņŹĒ_______________(ÓƱąŗÅĢīŠ“)”£
(2)³£ĪĀĻĀ£¬0.1mol”¤L£1CH3COOHČÜŅŗ¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠ£¬ĻĀĮŠ±ķ“ļŹ½µÄŹż¾Ż±ä“óµÄŹĒ ”£
A£®c(H£«) B£®c(H£«)/c(CH3COOH) C£®c(H£«)”¤c(OH£)
D£®c(OH£)/c(H£«) E£®c(H£«)”¤c(CH3COO£)/c(CH3COOH)
(3)Ģå»ż¾łĪŖ100mL pH£½2µÄCH3COOHÓėŅ»ŌŖĖįHX£¬¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠpHÓėČÜŅŗĢå»żµÄ¹ŲĻµČēĶ¼ĖłŹ¾£¬ŌņHXµÄµēĄėĘ½ŗā³£Źż_____(Ģī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±)CH3COOHµÄµēĄėĘ½ŗā³£Źż£¬ĄķÓÉŹĒ_____________________”£

(4)25”ꏱ£¬CH3COOHÓėCH3COONaµÄ»ģŗĻČÜŅŗ£¬Čō²āµĆpH£½6£¬ŌņČÜŅŗÖŠ
c(CH3COO£)£c(Na£«)£½ mol”¤L£1(Ģī¾«Č·Öµ)”£
(5)±ź×¼×“æöĻĀ£¬½«1.12L CO2ĶØČė100mL 1mol”¤L£1µÄNaOHČÜŅŗÖŠ£¬ÓĆČÜŅŗÖŠĪ¢Į£µÄÅØ¶Č·ūŗÅĶź³ÉĻĀĮŠµČŹ½£ŗ
¢Łc (OH£)£½2c(H2CO3)£« £»
¢Śc(H£«)£«c(Na£«)£½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010-2011ѧğĢģ½ņŹŠøßČżµŚ¶ž“ĪÄ£Äāæ¼ŹŌ£ØĄķ×Ū£©»Æѧ²æ·Ö ĢāŠĶ£ŗĢīæÕĢā
£Ø14·Ö£©ŅŃÖŖ25”ꏱ²æ·ÖČõµē½āÖŹµÄµēĄėĘ½ŗā³£ŹżŹż¾ŻČēĻĀ±ķ£ŗ

»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ĪļÖŹµÄĮæÅØ¶Č¾łĪŖ0.1mol”¤L-1µÄĖÄÖÖČÜŅŗ£ŗa.CH3COOH b.Na2CO3 c.NaClO d.NaHCO3£»pHÓÉŠ”µ½“óµÄÅÅĮŠĖ³ŠņŹĒ £ØÓĆ×ÖÄø±ķŹ¾£©
£Ø2£©³£ĪĀĻĀ£¬0.1mol”¤L-1µÄCH3COOHČÜŅŗ¼ÓĻ”ŹĶ¹ż³ĢÖŠ£¬ĻĀĮŠ±ķ“ļŹ½µÄŹż¾Ż±ä“óµÄŹĒ
A£®c(H+) B£®c(H+)/c(CH3COOH)
C£®c(H+)”¤c(OH-) D£®c(OH-)/c(H+)
£Ø3£©Ģå»ż¾łĪŖ100mL pH=2µÄCH3COOHÓėŅ»ŌŖĖįHX£¬¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠpHÓėČÜŅŗĢå»żµÄ¹ŲĻµČēĶ¼ĖłŹ¾

ŌņHXµÄµēĄėĘ½ŗā³£Źż CH3COOHµÄµēĄėĘ½ŗā³£Źż£ØĢī“óÓŚ”¢Š”ÓŚ»ņµČÓŚ£©ĄķÓÉŹĒ
£Ø4£©25”ꏱ£¬CH3COOHÓėCH3COONaµÄ»ģŗĻČÜŅŗ£¬Čō²āµĆpH=6£¬ŌņČÜŅŗÖŠc(CH3COO-)”Ŗc(Na+)= mol”¤L-1£ØĢī¾«Č·Öµ£©£¬c(CH3COO-)/c(CH3COOH)=
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com