
��14�֣�������±�ᷴӦ���Ʊ�±��������Ҫ������ʵ�����Ʊ��������װ������ͼ��ʾ���Թ�d��װ����������ˮ.��֪������ķе�Ϊ38.4oC���ܶ�Ϊ1.43g��ml-1��
���ܴ��ڵĸ���Ӧ�У����ȷ�Ӧ�����з�Ӧ��������ֻ�ɫ��������ɫ������Ũ����Ĵ�������ˮ����ϩ���ѵȡ�
��1���Ʊ����������Ҫ�õ�����ˮ���Ҵ����廯�ơ�Ũ���ᣬ����ƿ�м����⼸�����ʵ�˳���� ��
��2��С�����,����ƿ�з�������Ҫ��Ӧ�Т�NaBr + H2SO4 �� NaHSO4 + HBr
�� .��
��3����ˮ������ ��d�Թ��е����� ��
��4�������ַ�����ȡ���������еĺ���������Br2������ȥ������е���������Br2�����й�ѡ�Լ������ʺϵ���__________��
A��NaI��Һ �� B��NaOH��Һ ��C��Na2SO3��Һ D��KCl��Һ
��5����ȫƿb���Է�ֹ����,�����Լ��ʵ�����ʱ�Թ�d�Ƿ�����������д����������ʱƿb�е�����________________
��6������e��NaOH��Һ��������__________________________________.
��7���Ʊ������У������Ũ�����������ʵ���ϡ�ͣ���Ŀ����_______��
A�����ٸ�����ϩ���ѵ����� B������Br2������
C������HBr�Ļӷ� D��ˮ�Ƿ�Ӧ�Ĵ���


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
| ���� |
| �Ҵ� | ������ | ������ | 1-�嶡�� | |
| �ܶ�/g?cm-3 | 0.7893 | 1.4604 | 0.8098 | 1.2758 |
| �е�/�� | 78.5 | 38.4 | 117.2 | 101.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �Ҵ� | ������ | ������ | 1-�嶡�� | |
| �ܶ�/g?cm-3 | 0.7893 | 1.4604 | 0.8098 | 1.2758 |
| �е�/�� | 78.5 | 38.4 | 117.2 | 101.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �Ҵ� | ������ | ������ | 1-�嶡�� | |
| �ܶ�/g?cm-3 | 0.7893 | 1.4604 | 0.8098 | 1.2758 |
| �е�/�� | 78.5 | 38.4 | 117.2 | 101.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �Ҵ� | ������ | ������ | 1-�嶡�� | |
| �ܶ�/g?cm-3 | 0.7893 | 1.4604 | 0.8098 | 1.2758 |
| �е�/�� | 78.5 | 38.4 | 117.2 | 101.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
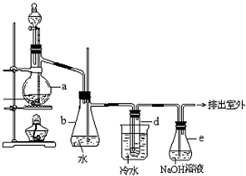 ������±�ᷴӦ���Ʊ�±��������Ҫ������ʵ�����Ʊ��������װ����ͼ��ʾ���Թ�d��װ����������ˮ����֪������ķе�Ϊ38.4��C���ܶ�Ϊ1.43g?ml-1��
������±�ᷴӦ���Ʊ�±��������Ҫ������ʵ�����Ʊ��������װ����ͼ��ʾ���Թ�d��װ����������ˮ����֪������ķе�Ϊ38.4��C���ܶ�Ϊ1.43g?ml-1���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com