
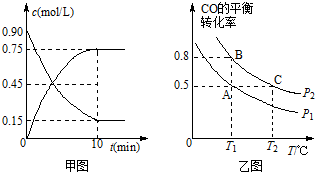
·ÖĪö £Ø1£©¢ŁSO2ÓėCaCO3Šü×ĒŅŗ·“Ó¦£¬°éĖęŃõ»Æ»¹Ō·“Ó¦£»
¢ŚSO2ÓėCa£ØOH£©2Šü×ĒŅŗ·“Ó¦£¬°éĖęŃõ»Æ»¹Ō·“Ó¦£¬Éś³ÉĮņĖįøĘ£»
£Ø2£©ÓĆŹģŹÆ»ŅµÄŠü×ĒŅŗ¶ų²»ÓĆ³ĪĒåŹÆ»ŅĖ®£¬ÓėÅضČÓŠ¹Ų£¬ÅØ¶Č“óŹ±ĪüŹÕ¶žŃõ»ÆĮņµÄŠ§¹ūŗĆ£»
£Ø3£©“ÓČ«Ēņ»·¾³±£»¤µÄ½Ē¶Č£¬¼õÉŁ¶žŃõ»ÆĮņµÄÅÅ·Å£¬¼õÉŁĖįÓźµÄ·¢Éś£®
½ā“š ½ā£ŗŹÆøą·ÖĪŖÉśŹÆøą£ØCaSO4•2H2O£©ŗĶŹģŹÆøą£Ø2CaSO4•H2O£©Į½ÖÖ£®
£Ø1£©¢ŁSO2ÓėCaCO3Šü×ĒŅŗ·“Ó¦£¬Ļąµ±ÓŚČõĖįÖĘøüČõµÄĖį£¬ŌŁŃõ»Æ£¬·¢Éś2SO2+O2+2CaCO3ØT2CaSO4+2CO2»ņ2SO2+O2+2CaCO3+4H2OØT2£ØCaSO4•2H2O£©+2CO2£¬
¹Ź“š°øĪŖ£ŗ2SO2+O2+2CaCO3+4H2OØT2£ØCaSO4•2H2O£©+2CO2£»
¢ŚSO2ÓėCa£ØOH£©2Šü×ĒŅŗ·“Ó¦£¬Ļąµ±ÓŚĖį¼īÖŠŗĶ·“Ó¦£¬ŌŁŃõ»Æ£¬·¢Éś2SO2+O2+2Ca£ØOH£©2+2H2OØT2£ØCaSO4•2H2O£©»ņ2SO2+O2+2Ca£ØOH£©2ØT2CaSO4+2H2O£¬
¹Ź“š°øĪŖ£ŗ2SO2+O2+2Ca£ØOH£©2+2H2OØT2£ØCaSO4•2H2O£©£»
£Ø2£©ÓĆŹģŹÆ»ŅµÄŠü×ĒŅŗ¶ų²»ÓĆ³ĪĒåŹÆ»ŅĖ®µÄĄķÓÉŹĒ£ŗCa£ØOH£©2Ī¢ČÜ£¬³ĪĒåµÄŹÆ»ŅĖ®ÖŠCa£ØOH£©2µÄÅØ¶ČŠ”£¬²»ĄūÓŚĪüŹÕSO2£¬
¹Ź“š°øĪŖ£ŗCa£ØOH£©2Ī¢ČÜ£¬³ĪĒåµÄŹÆ»ŅĖ®ÖŠCa£ØOH£©2µÄÅØ¶ČŠ”£¬²»ĄūÓŚĪüŹÕSO2£»
£Ø3£©ÓÉÓŚ½ØŌģøßŃĢ“ŃµÄ½į¹ū£¬µŲ±ķĆęSO2ÅضČČ“½µµĶĮĖ30%Ö®¶ą£®“ÓČ«Ēņ»·¾³±£»¤µÄ½Ē¶Čæ¼ĀĒ£¬ÕāÖÖ·½·Ø²»æÉČ”£¬ŅņĪŖSO2µÄÅÅ·Å×ÜĮæƻӊ¼õÉŁ£¬Ęä½ųŅ»²½ŠĪ³ÉµÄĖįÓźČŌ»įŌģ³É¶ŌČ«Ēņ»·¾³µÄ£¬“š£ŗ·½·Ø²»æÉČ”£¬ŅņĪŖSO2µÄÅÅ·Å×ÜĮæƻӊ¼õÉŁ£¬Ęä½ųŅ»²½ŠĪ³ÉµÄĖįÓźČŌ»įŌģ³É¶ŌČ«Ēņ»·¾³µÄ£®
µćĘĄ ±¾Ģā漲鶞Ńõ»ÆĮņµÄĪŪČ¾¼°ÖĪĄķ£¬ĪŖøßĘµæ¼µć£¬°ŃĪÕĪļÖŹµÄŠŌÖŹ”¢·¢ÉśµÄ·“Ó¦¼°»·¾³Óė±£»¤ĪŹĢāĪŖ½ā“šµÄ¹Ų¼ü£¬²ąÖŲ·ÖĪöÓėÓ¦ÓĆÄÜĮ¦µÄ漲飬ĢāÄæÄŃ¶Č²»“ó£®



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | æÉÓĆ³ĪĒåŹÆ»ŅĖ®¼ų±šNa2CO3ŗĶNaHCO3ČÜŅŗ | |
| B£® | ÅØĮņĖį¾ßÓŠĪüĖ®ŠŌ£¬æÉ×öøÉŌļ¼Į£¬µ«²»ÄÜøÉŌļNH3 | |
| C£® | Äʱ£“ęŌŚĆŗÓĶÖŠ£¬Čō³¤ĘŚ·ÅÖĆŌŚæÕĘųÖŠ£¬×īÖÕ±äĪŖNaHCO3 | |
| D£® | Ć¾×Å»šŗóæÉŅŌÓĆCO2Ą“Ćš»š |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
 Åš¼°Ęä»ÆŗĻĪļŌŚ¹¤ŅµÉĻÓŠŠķ¶ąÓĆĶ¾£®¹¤ŅµÉĻŅŌĢśÅšæóĪŖŌĮĻÖʱøÅšĖį£¬ĢśÅšæóŗ¬ÓŠMg”¢Fe”¢Ca”¢Al”¢B”¢OµČ¶ąÖÖŌŖĖŲ£¬ĖüµÄÖ÷ŅŖ³É·ÖĪŖMg2B2O5•H2OŗĶFe3O4£®
Åš¼°Ęä»ÆŗĻĪļŌŚ¹¤ŅµÉĻÓŠŠķ¶ąÓĆĶ¾£®¹¤ŅµÉĻŅŌĢśÅšæóĪŖŌĮĻÖʱøÅšĖį£¬ĢśÅšæóŗ¬ÓŠMg”¢Fe”¢Ca”¢Al”¢B”¢OµČ¶ąÖÖŌŖĖŲ£¬ĖüµÄÖ÷ŅŖ³É·ÖĪŖMg2B2O5•H2OŗĶFe3O4£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ¶¼±Č½ĻĪČ¶Ø£¬¾ĆÖĆ²»±ä»ė×Ē | B£® | ĻąĶ¬µÄŃÕÉ« | ||
| C£® | ÓŠ¶”“ļ¶ūĻÖĻó | D£® | ¼ÓŃĪĖįĻČ²śÉś³Įµķ£¬ŗó³ĮµķČܽā |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ŗŚ»šŅ©ÓÉĮņ»Ē”¢ĻõŹÆ”¢Ä¾ĢæČżÖÖĪļÖŹ°“Ņ»¶Ø±ČĄż»ģŗĻÖĘ³É | |
| B£® | ÕØÓĶĢõŹ±£¬³£½«Š”ĖÕ“ņŗĶĆ÷·Æ»ģÓĆ×ö·¢½Ķ¼Į | |
| C£® | “óĮ¦ĶĘ¹ćÓ¦ÓĆČ¼ĮĻ”°ĶŃĮņ”¢ĶŃĻõ”±¼¼Źõ£¬æɼõÉŁĮņŃõ»ÆĪļŗĶµŖŃõ»ÆĪļ¶ŌæÕĘųµÄĪŪČ¾ | |
| D£® | PM2.5æÅĮ£ŌŚ“óĘųÖŠæÉŠĪ³ÉĘųČܽŗ£¬¾ßÓŠ¶”“ļ¶ūŠ§Ó¦ |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com