
������һ�����͵���ɫ��Դ������һ����Ҫ�Ļ���ԭ�ϡ�
��1������ȼ����ֵ�ߡ�ʵ���ã��ڳ��³�ѹ�£�1 g H2��ȫȼ������Һ̬ˮ���ų�142.9 kJ��������H2ȼ���ȵĻ�ѧ����ʽΪ ��
��2�������Ǻϳɰ�����Ҫԭ�ϣ��ϳɰ���Ӧ���Ȼ�����ʽ���£�N2(g)��3H2(g) 2NH3(g)����H����92.4 kJ/mol
2NH3(g)����H����92.4 kJ/mol
�ٵ��ϳɰ���Ӧ�ﵽƽ��ı�ijһ�������(���ı�N2��H2��NH3����)����Ӧ������ʱ��Ĺ�ϵ����ͼ��ʾ��ͼ��t1ʱ����ƽ���ƶ������������� ���б�ʾƽ��������NH3�ĺ�����ߵ�һ��ʱ���� 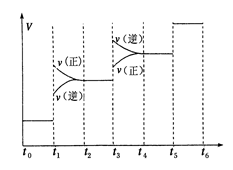
���¶�ΪT ��ʱ����2 a mol H2��a mol N2����0.5 L�ܱ������У���ַ�Ӧ����N2��ת����Ϊ50%����Ӧ��ƽ�ⳣ��Ϊ
��1�� H2(g)�� 1/2O2(g)��H2O(l)����H����285.8 kJ/mol
��2��������ѹǿ��t2��t3����
���������������1��1g���������ʵ�����0.5mol��ȼ��0.5mol�����ų�142.9kJ����������ȼ��1mol�����ų�285.8kJ����������������ȼ���Ȼ�ѧ��Ӧ����ʽΪ��H2��g��+1/2O2��g��=H2O��1����H=-285.8kJ/mol��
��2��N2��g��+3H2��g��?2NH3��g������H=-92.4kJ/mol����Ӧ�Ƿ��ȷ�Ӧ����Ӧǰ�����������С�������ݷ�Ӧ������ʱ��Ĺ�ϵͼ�������t1ʱ���淴Ӧ���ʶ�����ƽ�������ƶ�������ƽ���ƶ�����������������ѹǿ����ʾƽ��������NH3�ĺ�����ߵ�һ��ʱ����t2-t3��
�� N2��g��+3H2��g��?2NH2��g��
��ʼ 2amol/L 4amol/L 0
��Ӧ amol/L 3amol/L 2amol/L
ƽ�� amol/L amol/L 2amol/L
���Ի�ѧƽ�ⳣ��K=(2amol/L)2/(amol/L)?(amol/L)3=4/a2��L/mol��2��
���㣺�Ȼ�ѧ����ʽ����ѧƽ�ⳣ���ĺ��壻��ѧƽ��ĵ�������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ѧ��һֱ�����ڡ��˹��̵����ķ����о���
(1)�ϳɰ���ԭ��Ϊ��N2(g)+3H2(g) 2NH3(g)��H="-92.4" kJ/mol���÷�Ӧ�������仯��ͼ��ʾ��
2NH3(g)��H="-92.4" kJ/mol���÷�Ӧ�������仯��ͼ��ʾ��
���ڷ�Ӧ��ϵ�м����������Ӧ��������E2�ı仯�� (���������С�����䡱)��
�ڽ�0.3 mol N2��0.5 mol H2�������������ܱ������У���һ�������´ﵽƽ�⣬�������������ѹǿ��Ϊԭ����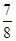 ����ʱH2��ת����Ϊ ������߸�������H2��ת���ʣ����д�ʩ���е��� (��ѡ����ĸ)��
����ʱH2��ת����Ϊ ������߸�������H2��ת���ʣ����д�ʩ���е��� (��ѡ����ĸ)��
| A���������а�ԭ�����ٳ���ԭ���� |
| B�����������ٳ���һ����H2 |
| C���ı䷴Ӧ�Ĵ��� |
| D��Һ�������������� |
 4NH3(g)+3O2(g)
4NH3(g)+3O2(g) H2O(g) ��H="+44.0" kJ/mol
H2O(g) ��H="+44.0" kJ/mol 4NH3(g)+3O2(g) ��H= kJ/mol���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK �����������������䣬����ѹǿ��Kֵ (���������С�����䡱)��
4NH3(g)+3O2(g) ��H= kJ/mol���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK �����������������䣬����ѹǿ��Kֵ (���������С�����䡱)���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ѧ��Ӧ�м������ʱ仯�����������仯���ͷŻ����������ǻ�ѧ��Ӧ�������仯����Ҫ��ʽ֮һ����֪C(ʯī)��H2(g)ȼ�յ��Ȼ�ѧ����ʽ�ֱ�Ϊ��
�� C(ʯī)+ O2(g)��CO(g)
O2(g)��CO(g) 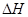 ="-111.0" KJ��mol-1
="-111.0" KJ��mol-1
�� H2(g)+  O2(g) ��H20(g)
O2(g) ��H20(g) 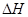 ="-242.0" kJ��mol-1
="-242.0" kJ��mol-1
�� C(ʯī)+O2(g)��CO2(g) 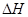 ="-394.0" kJ��mol-1
="-394.0" kJ��mol-1
�����������⣺
��1����ѧ��Ӧ���������仯�ı���ԭ���Ƿ�Ӧ�������� �Ķ��Ѻ��γɡ�����������Ӧ���� (����ȡ����ȡ�)��Ӧ��
��2�����Ȼ�ѧ����ʽ�У���Ҫ������Ӧ�P�������״̬��ԭ���� ���ڢ��У�02�Ļ�ѧ��������1/2���DZ�ʾ (����ĸ)��
a�����Ӹ��� b�����ʵ��� c����������
��3����Ӧ2H20(g)��2H2(g)+02(g)��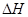 = KJ��mol-1��
= KJ��mol-1��
��4����C(���ʯ)+02(g)��C02(g)��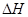 ="-395.0" kJ��mol-1�����ȶ��ԣ����ʯ (�����������������)ʯī��
="-395.0" kJ��mol-1�����ȶ��ԣ����ʯ (�����������������)ʯī��
��5����֪�γ�H20(g)�е�2 mol H-O���ܷų�926.0 kJ���������γ�1 mol 02(g)�еĹ��ۼ��ܷų�498.0 kJ�������������1 mol H2(g)�е�H-H����Ҫ������ KJ��
��6����ҵ��������һ����Ҫ;������CO(g)��H2O(g)��Ӧ����C02(g)��H2(g)����÷�Ӧ���Ȼ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������һЩ���ʵ��۷е����ݣ���ѹ����
| | �� | �� | Na2CO3 | ���ʯ | ʯī |
| �۵㣨�棩 | 63��65 | 97��8 | 851 | 3550 | 3850 |
| �е㣨�棩 | 774 | 882��9 | 1850���ֽ����CO2�� | ---- | 4250 |
 2 Na2CO3��l��+ C(s,���ʯ) ��H=��1080��9kJ/mol
2 Na2CO3��l��+ C(s,���ʯ) ��H=��1080��9kJ/mol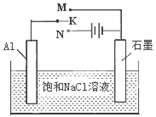
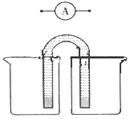
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ҵ�ķ�չ������Ϲ��ҽ��ܼ��ŵ�����Ҫ����ش��������⣺
��1����֪C(s)+H2O(g)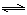 CO(g)+H2(g)����÷�Ӧ��ƽ�ⳣ������ʽΪ ��
CO(g)+H2(g)����÷�Ӧ��ƽ�ⳣ������ʽΪ ��
��2����֪��һ���¶��£�
C��s��+CO2��g�� 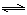 2CO��g�� ��H1
2CO��g�� ��H1
CO��g��+H2O��g�� 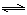 H2��g��+CO2��g�� ��H2
H2��g��+CO2��g�� ��H2
C��s��+H2O��g��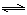 CO��g��+H2��g�� ��H3
CO��g��+H2��g�� ��H3
���H1����H2����H3֮��Ĺ�ϵ�ǣ� ��
��3��ͨ���о���ͬ�¶���ƽ�ⳣ�����Խ��ijЩʵ�����⡣��֪�������һ����̼��ˮ�������뷴Ӧʱ���ᷢ�����·�Ӧ�� CO(g)+H2O(g) 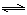 H2(g)+CO2(g)���÷�Ӧƽ�ⳣ�����¶ȵı仯�����ʾ��
H2(g)+CO2(g)���÷�Ӧƽ�ⳣ�����¶ȵı仯�����ʾ��
| �¶�/�� | 400 | 500 | 800 |
| ƽ�ⳣ��K | 9.94 | 9 | 1 |
 2NO2(g) ��H��0���¶�ΪT1��T2ʱ��ƽ����ϵ��NO2�����������ѹǿ�仯������ͼ��ʾ������˵����ȷ���� ��
2NO2(g) ��H��0���¶�ΪT1��T2ʱ��ƽ����ϵ��NO2�����������ѹǿ�仯������ͼ��ʾ������˵����ȷ���� ��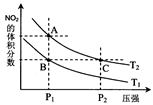

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����β���ﺬ�е�NO������������ȼ��ȼ�յĸ�����������������Ӧ���£�
N2(g)��O2(g)  2NO(g) ��H����֪�÷�Ӧ�� T ��ʱ��ƽ�ⳣ��K��9.0��
2NO(g) ��H����֪�÷�Ӧ�� T ��ʱ��ƽ�ⳣ��K��9.0��
��ش�
��1����֪��N2(g)+2O2(g)  2NO2(g) ��H1 2NO2(g)
2NO2(g) ��H1 2NO2(g)  O2+2NO(g) ��H2 ��H= ���ú���H1����H2�ı���ʽ��ʾ����
O2+2NO(g) ��H2 ��H= ���ú���H1����H2�ı���ʽ��ʾ����
��2��ij�¶��£���2 L���ܱ������г���N2��O2��1 mol��5���Ӻ�O2�����ʵ���Ϊ0.5 mol����NO�ķ�Ӧ���� ��
��3���ٶ��÷�Ӧ���ں��������½��У��������жϸ÷�Ӧ�Ѵﵽƽ�����________��
| A������1 mol N2ͬʱ����1 mol O2 |
| B����������ܶȲ��� |
| C���������ƽ����Է����������� |
| D��2v��(N2)��v��(NO) |
 2NO(g)�ġ�K-T������c(NO)-t��ͼ����ͼA������֪�÷�ӦΪ ��Ӧ������ȡ����ȡ�������ͼB��֪����a��Ӧ��������ȣ�b�ı������������ ��
2NO(g)�ġ�K-T������c(NO)-t��ͼ����ͼA������֪�÷�ӦΪ ��Ӧ������ȡ����ȡ�������ͼB��֪����a��Ӧ��������ȣ�b�ı������������ ��
 2NO(g)________________(����ڻ�ѧƽ��״̬������������Ӧ������С������淴Ӧ������С�)��ƽ��ʱ��N2�ڻ�����������ٷ���Ϊ���٣����ڴ����д�����������̣��������2λ��Ч���֣�
2NO(g)________________(����ڻ�ѧƽ��״̬������������Ӧ������С������淴Ӧ������С�)��ƽ��ʱ��N2�ڻ�����������ٷ���Ϊ���٣����ڴ����д�����������̣��������2λ��Ч���֣��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(15��)��Դ�Ŀ������������������Ŀɳ�����չϢϢ��أ�����������ú���Դ�ǰ���������ǰ���ش���⡣
I����֪��Fe2O3(s)+3C(ʯī) =2Fe(s)+3CO(g) ��H=akJ��mol-1
CO(g)+1��2O2(g)= CO2(g) ��H=bkJ��mol-1
C(ʯī)+O2(g)=CO2(g) ��H=ckJ��mol-1
��Ӧ��4Fe(s)+3O2(g)= 2Fe2O3(s)���ʱ��H= kJ��mol-1��
��1������ԭ��صĹ���ԭ�������л�ѧ��Ӧ�������Ͽ�����Ƴ�ԭ��ص��� (�����)��
A��C(s)+CO2(g)=2CO(g)
B��NaOH(aq)+HCl(aq)=NaCl(aq)+H2O(l)
C��2H2O(l)= 2H2(g)+O2(g)
D��CH4(g)+2O2(g)=CO2(g)+2H2O(l)
����KOH��ҺΪ�������Һ��������ѡ��Ӧ������Ƴ�һ��ԭ��أ���д����ԭ��صĵ缫��Ӧ��
������ ��
������ ��
��2����������(ClO2)��һ�ָ�Ч��ȫ������ˮ��������ClO2��һ�ֻ���ɫ���壬������ˮ��ʵ������NH4Cl�����ᡢNaClO2Ϊԭ���Ʊ�ClO2�������£�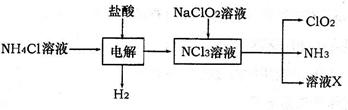
��֪���������з����ķ�ӦΪ��
NH4Cl+2HCl NCl3+3H2��������NCl3�е�Ԫ��Ϊ+3�ۡ�
NCl3+3H2��������NCl3�е�Ԫ��Ϊ+3�ۡ�
��д�����ʱ�����ĵ缫��Ӧʽ ��
���������Ϸŵ������(������)�� ��
�۳�ȥClO2�е�NH3��ѡ�õ��Լ��� (�����)
A����ʯ�� B����ʯ�� C��ŨH2SO4 D��ˮ
�������������У�ÿ����1mol ClO2�������� mol NCl3��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����ѣ�CH3OCH3����һ����Ҫ�ľ�ϸ������Ʒ������Ϊ�Ƕ�ʮһ��������DZ����ȼ��[ ��֪��CH3OCH3(g)+3O2(g)��2CO2(g)+3H2O(1) ��H����1455kJ/mol ]��ͬʱ��Ҳ������Ϊ�������������ȴ�������ҵ���Ʊ������ѵ���Ҫ���������������Σ�
�ټ״�Һ����Ũ���������»�״������ڴ�������ֱ����ˮ�ƶ����ѣ�2CH3OH CH3OCH3��H2O
CH3OCH3��H2O
�ںϳ���CO��H2ֱ�Ӻϳɶ����ѣ�3H2(g)��3CO(g) CH3OCH3(g)��CO2(g) ��H����247kJ/mol
CH3OCH3(g)��CO2(g) ��H����247kJ/mol
����Ȼ����ˮ������Ӧ�Ʊ������ѡ���CH4��H2OΪԭ���Ʊ������Ѻͼ״���ҵ�������£�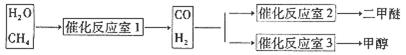
��1��д��CO(g)��H2(g)��O2(g)��Ӧ����CO2(g)��H2O(1)���Ȼ�ѧ����ʽ���������һλС���� ��
��2���ٷ������ü״�Һ����Ũ��������ֱ����ˮ�ƶ����ѣ����ܲ��ʸߣ���������̭����Ҫԭ���� ��
��3���ڷ�Ӧ��2�У�һ�������·�����Ӧ3H2(g)��3CO(g) CH3OCH3(g)��CO2(g)���ܱ������дﵽƽ���Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ�� ��
CH3OCH3(g)��CO2(g)���ܱ������дﵽƽ���Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ�� ��
A�����¸�ѹ B���Ӵ��� C������COŨ�� D�������������
��4���ڷ�Ӧ��3�У���һ���¶Ⱥ�ѹǿ�����·����˷�Ӧ��3H2(g)��CO2(g)  CH3OH(g)��H2O (g) ��H��0��Ӧ�ﵽƽ��ʱ���ı��¶ȣ�T����ѹǿ��P������Ӧ�����CH3OH�����ʵ����������仯�����ͼ��ʾ�������¶ȣ�T����ѹǿ��P���Ĺ�ϵ�ж���ȷ���� ������ţ���
CH3OH(g)��H2O (g) ��H��0��Ӧ�ﵽƽ��ʱ���ı��¶ȣ�T����ѹǿ��P������Ӧ�����CH3OH�����ʵ����������仯�����ͼ��ʾ�������¶ȣ�T����ѹǿ��P���Ĺ�ϵ�ж���ȷ���� ������ţ���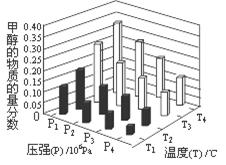
A��P3��P2 T3��T2 B��P2��P4 T4��T2
C��P1��P3 T1��T3 D��P1��P4 T2��T3
��5����Ӧ��1�з�����Ӧ��CH4(g)��H2O(g) CO(g)��3H2(g) ��H��0д��ƽ�ⳣ���ı���ʽ�� ������¶Ƚ��ͣ��÷�Ӧ��ƽ�ⳣ�� ��������䡱�����������С����
CO(g)��3H2(g) ��H��0д��ƽ�ⳣ���ı���ʽ�� ������¶Ƚ��ͣ��÷�Ӧ��ƽ�ⳣ�� ��������䡱�����������С����
��6����ͼΪ��ɫ��Դ��������ȼ�ϵ�ء��Ĺ���ԭ��ʾ��ͼ����a�缫�ķ�ӦʽΪ��________________��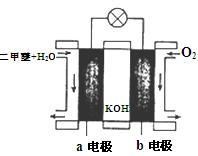
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��һ���Ҵ�ȼ�ϵ�ع���ʱ��ʾ��ͼ���ҳ��е������缫һ����ʯī�缫��һ�������缫������ʱM��N�����缫�������������٣���ش��������⣺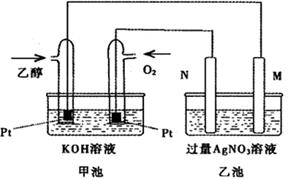
��1��M�缫�IJ����� ���缫������ �������Ҵ��IJ��缫�ĵ缫��Ӧ
ʽΪ ��д���ҳ��з����Ļ�ѧ��Ӧ�����ӷ���ʽ�� ��
��2���ڴ˹����У��ҳ���ijһ�缫����������4��32gʱ���׳�����������������Ϊ L����״���£�������ʱ�ҳ���Һ�����Ϊ400mL�����ҳ�����Һ��pHΪ ��
��3�����ڳ��³�ѹ�£�1g C2H5OHȼ������CO2��Һ̬H2Oʱ�ų�29��71kJ��������ʾ�÷�Ӧ���Ȼ�ѧ����ʽΪ ��
��4������Ҳ��һ�ֺܺõ������Դ���̲��ں��ġ���ȼ�����Ǹ�ѹ���γɵ��������ļ���ˮ������塣��������ȼ�յ��Ȼ�ѧ����ʽΪ��CH4(g)��2O2(g)��CO2(g)��2H2O(l) ��H����890��3 kJ��mol��356g����ȼ����(������ʽΪCH4��9H2O)�ͷŵļ���������ȫȼ������Һ̬ˮ���ų�������Ϊ kJ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com