
(8Ζ÷Θ§ΟΩΩ’2Ζ÷)Υ° «…ζΟϋ÷°‘¥Θ§Υϋ”κΈ“Ο«ΒΡ…ζΜνΟή«–œύΙΊΓΘ‘ΎΜ·―ß Β―ιΚΆΩΤ―ß―–ΨΩ÷–Θ§Υ°“≤ «“Μ÷÷≥Θ”ΟΒΡ ‘ΦΝΓΘ
(1)–¥≥ω”κH2OΖ÷Ή”ΜΞΈΣΒ»ΒγΉ”ΧεΒΡΈΔΝΘ___ ___(Χν1÷÷)ΓΘ
(2)Υ°Ζ÷Ή”‘ΎΧΊΕ®ΧθΦΰœ¬»ί“ΉΒΟΒΫ“ΜΗωHΘΪΘ§–Έ≥…Υ°Κœ«β‘≠Ή”(H3OΘΪ)ΓΘœ¬Ν–Ε‘…œ ωΙΐ≥ΧΒΡΟη ω≤ΜΚœάμΒΡ « (ΓΓΓΓ)
AΘ°―θ‘≠Ή”ΒΡ‘”Μ·άύ–ΆΖΔ…ζΝΥΗΡ±δ BΘ°ΈΔΝΘΒΡ–ΈΉ¥ΖΔ…ζΝΥΗΡ±δ
CΘ°ΈΔΝΘΒΡΜ·―ß–‘÷ ΖΔ…ζΝΥΗΡ±δ DΘ°ΈΔΝΘ÷–ΒΡΦϋΫ«ΖΔ…ζΝΥΗΡ±δ
(3)œ¬Ν– «ΡΤΓΔΒβΓΔΫπΗ’ ·ΓΔΗ…±υΓΔ¬»Μ·ΡΤΨßΧεΒΡΨßΑϊΆΦ(Έ¥Α¥Υ≥–ρ≈≈–ρ)ΓΘ”κ±υΒΡΨßΧεάύ–ΆœύΆ§ΒΡ «________(«κ”Οœύ”ΠΒΡ±ύΚ≈Χν–¥)ΓΘ
(4)‘Ύ±υΨßΧε÷–Θ§ΟΩΗωΥ°Ζ÷Ή””κœύΝΎΒΡ4ΗωΥ°Ζ÷Ή”–Έ≥…«βΦϋ(»γœ¬ΆΦΥυ Ψ)Θ§“―÷Σ±υΒΡ…ΐΜΣ»» «51 kg/molΘ§≥ΐ«βΦϋΆβΘ§Υ°Ζ÷Ή”ΦδΜΙ¥φ‘ΎΖΕΒ¬ΜΣΝΠ(11 kJ/mol)Θ§‘ρ±υΨßΧε÷–«βΦϋΒΡΓΑΦϋΡήΓ± «_____ ___kJ/molΘΜ
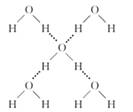
Θ®1Θ©H2S Θ®2Θ©A Θ®3Θ©BC Θ®4Θ©20
ΓΨΫβΈωΓΩΘ®1Θ©Ζ÷Ή” ΐΚΆΦέΒγΉ” ΐΖ÷±πΕΦœύΒ»ΒΡ «Β»ΒγΉ”ΧεΘ§Υυ“‘ΚΆΥ°ΜΞΈΣΒ»ΒγΉ”ΧεΒΡ «H2SΓΘ
Θ®2Θ©Υ° «V–ΆΫαΙΙΘ§ΕχH3OΘΪ «»ΐΫ«ΉΕ–ΈΘ§―θ‘≠Ή”ΜΙ «sp3‘”Μ·Θ§―ΓœνA «¥μΈσΒΡΘ§Τδ”ύΕΦ «’ΐ»ΖΒΡΘ§¥πΑΗ―ΓAΓΘ
Θ®3Θ©ΗυΨίΨßΑϊΫαΙΙΩ…÷ΣΘ§AΓΪEΖ÷±π «¬»Μ·ΡΤΓΔCO2ΓΔΒβΓΔΫπΗ’ ·ΚΆΡΤΓΘ”…”ΎΥ°–Έ≥…ΒΡΨßΧε «Ζ÷Ή”ΨßΧεΘ§Υυ“‘¥πΑΗ―ΓBCΓΘ
Θ®4Θ©ΗυΨίΫαΙΙΩ…÷ΣΘ§ΟΩΗωΥ°Ζ÷Ή”–Έ≥…2Ηω«βΦϋΘ§Υυ“‘«βΦϋΦϋΡή «Θ®51 kg/molΘ≠11 kg/molΘ©Γ¬2ΘΫ20 kg/molΓΘ


 ‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2012-2013―ßΡξΝ…Ρΰ ΓΗΏ»ΐΒΎ“Μ¥Έ‘¬ΩΦΜ·―ß ‘ΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚΧνΩ’Χβ
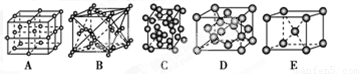
Θ®ΟΩΩ’2Ζ÷Ι≤ΦΤ8Ζ÷Θ©AΓΔBΓΔCΓΔDΥΡ÷÷Ω…»ή–‘―ΈΘ§ΥϋΟ«ΒΡ―τάκΉ”Ζ÷±π «Ba2+ΓΔAg+ΓΔNa+ΓΔCu2+ΘΜ“θάκΉ”Ζ÷±π «NO3-ΓΔSO42-ΓΔCl-ΓΔSO32-Θ®άκΉ”≤ΜΡή÷ΊΗ¥≥ωœ÷Θ©ΓΘΉω»γœ¬ Β―ιΘΚ
ΔΌΖ÷±π»ή”ΎΥ°Θ§÷Μ”–C»ή“Κ≥ άΕ…Ϊ
ΔΎ»τΑ―»ή”ΎΥ°ΒΡΥΡ÷÷»ή“ΚΖ÷±πΦ”»κ―ΈΥαΘ§B»ή“Κ≥ωœ÷≥ΝΒμΘ§D»ή“Κ÷–”–¥ΧΦΛ–‘ΤχΧεΖ≈≥ω‘ρAΈΣ »ή“ΚΘ§BΈΣ »ή“ΚΘ§CΈΣ »ή“ΚΘ§DΈΣ »ή“Κ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2011-2012―ßΡξΚΰΡœ ΓΗΏ»ΐ…œ―ßΤΎΤΎ÷–ΩΦ ‘Μ·―ß ‘Ψμ Χβ–ΆΘΚ Β―ιΧβ
Θ®8Ζ÷Θ§ΟΩΩ’2Ζ÷Θ©Α± «“Μ÷÷÷Ί“ΣΒΡΜ·ΙΛ‘≠ΝœΘ§Ρ≥―ßœΑ–ΓΉι”ϊ÷Τ»ΓΑ±Τχ≤ΔΧΫΨΩΤδ–‘÷ ΓΘ«κΜΊ¥πΘΚ
Θ®1Θ©ΗΟ–ΓΉιΆ§―ß…ηΦΤΝΥœ¬ΆΦΥυ ΨΒΡ Β―ιΉΑ÷ΟΘ®≤ΩΖ÷Φ–≥÷“«ΤςΈ¥Μ≠≥ωΘ©Θ§ΧΫΨΩΑ±ΤχΒΡΜΙ‘≠–‘≤ΔΦλ―ι≤ζΈοΓΘ
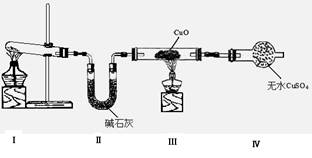
ΔΌΉΑ÷ΟΔώ÷–÷Τ»ΓΑ±ΤχΒΡΜ·―ßΖΫ≥Χ ΫΈΣ ΓΘ
ΔΎΉΑ÷ΟΔσΓΔΔτ÷–ΒΡ Β―ιœ÷œσΖ÷±π «ΘΚΚΎ…ΪCuO±δΈΣΚλ…ΪΘΜΑΉ…ΪΈόΥ°CuSO4ΖέΡ©±δΈΣάΕ…ΪΘΜΆ§ ±…ζ≥…“Μ÷÷Έό…ΪΤχΧεΘ§ΗΟΤχΧεΈόΈέ»ΨΓΘ
«κ–¥≥ωΑ±Τχ”κCuOΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ ΘΜ
ΔέΉΑ÷ΟΔρ÷–Φν ·Μ“ΒΡΉς”Ο « ΓΘ
Θ®2Θ©”–Ά§―ß»œΈΣΘΚNH3”κCuOΖ¥”Π…ζ≥…ΒΡΚλ…ΪΈο÷ ÷–Ω…ΡήΚ§”–Cu2OΓΘ“―÷ΣΘΚCu2O «“Μ÷÷Φν–‘―θΜ·ΈοΘΜ‘ΎΥα–‘»ή“Κ÷–Θ§Cu+ΒΡΈ»Ε®–‘±»Cu2+≤ν(Cu2O+2H+= Cu + Cu2+ +2H2O Θ©ΓΘ
«κΡψ…ηΦΤ“ΜΗωΦρΒΞΒΡ Β―ιΦλ―ιΗΟΚλ…ΪΈο÷ ÷– «ΖώΚ§”–Cu2OΓΘ
ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ
Θ®8Ζ÷Θ§ΟΩΩ’2Ζ÷Θ©Α± «“Μ÷÷÷Ί“ΣΒΡΜ·ΙΛ‘≠ΝœΘ§Ρ≥―ßœΑ–ΓΉι”ϊ÷Τ»ΓΑ±Τχ≤ΔΧΫΨΩΤδ–‘÷ ΓΘ«κΜΊ¥πΘΚ
Θ®1Θ©ΗΟ–ΓΉιΆ§―ß…ηΦΤΝΥœ¬ΆΦΥυ ΨΒΡ Β―ιΉΑ÷ΟΘ®≤ΩΖ÷Φ–≥÷“«ΤςΈ¥Μ≠≥ωΘ©Θ§ΧΫΨΩΑ±ΤχΒΡΜΙ‘≠–‘≤ΔΦλ―ι≤ζΈοΓΘ
ΔΌΉΑ÷ΟΔώ÷–÷Τ»ΓΑ±ΤχΒΡΜ·―ßΖΫ≥Χ ΫΈΣ ΓΘ
ΔΎΉΑ÷ΟΔσΓΔΔτ÷–ΒΡ Β―ιœ÷œσΖ÷±π «ΘΚΚΎ…ΪCuO±δΈΣΚλ…ΪΘΜΑΉ…ΪΈόΥ°CuSO4ΖέΡ©±δΈΣάΕ…ΪΘΜΆ§ ±…ζ≥…“Μ÷÷Έό…ΪΤχΧεΘ§ΗΟΤχΧεΈόΈέ»ΨΓΘ
«κ–¥≥ωΑ±Τχ”κCuOΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ ΘΜ
ΔέΉΑ÷ΟΔρ÷–Φν ·Μ“ΒΡΉς”Ο « ΓΘ
Θ®2Θ©”–Ά§―ß»œΈΣΘΚNH3”κCuOΖ¥”Π…ζ≥…ΒΡΚλ…ΪΈο÷ ÷–Ω…ΡήΚ§”–Cu2OΓΘ“―÷ΣΘΚCu2O «“Μ÷÷Φν–‘―θΜ·ΈοΘΜ‘ΎΥα–‘»ή“Κ÷–Θ§Cu+ΒΡΈ»Ε®–‘±»Cu2+≤ν(Cu2O+2H+= Cu + Cu2+ +2H2OΘ©ΓΘ
«κΡψ…ηΦΤ“ΜΗωΦρΒΞΒΡ Β―ιΦλ―ιΗΟΚλ…ΪΈο÷ ÷– «ΖώΚ§”–Cu2OΓΘ
ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2012ΫλΚΰΡœ ΓθΖΝξΥΡ÷–ΗΏ»ΐ…œ―ßΤΎΤΎ÷–ΩΦ ‘Μ·―ß ‘Ψμ Χβ–ΆΘΚ Β―ιΧβ
Θ®8Ζ÷Θ§ΟΩΩ’2Ζ÷Θ©Α± «“Μ÷÷÷Ί“ΣΒΡΜ·ΙΛ‘≠ΝœΘ§Ρ≥―ßœΑ–ΓΉι”ϊ÷Τ»ΓΑ±Τχ≤ΔΧΫΨΩΤδ–‘÷ ΓΘ«κΜΊ¥πΘΚ
Θ®1Θ©ΗΟ–ΓΉιΆ§―ß…ηΦΤΝΥœ¬ΆΦΥυ ΨΒΡ Β―ιΉΑ÷ΟΘ®≤ΩΖ÷Φ–≥÷“«ΤςΈ¥Μ≠≥ωΘ©Θ§ΧΫΨΩΑ±ΤχΒΡΜΙ‘≠–‘≤ΔΦλ―ι≤ζΈοΓΘ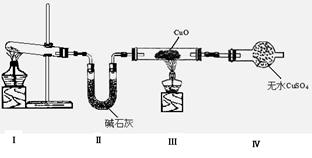
ΔΌΉΑ÷ΟΔώ÷–÷Τ»ΓΑ±Τχ ΒΡΜ·―ßΖΫ≥Χ ΫΈΣ ΓΘ
ΒΡΜ·―ßΖΫ≥Χ ΫΈΣ ΓΘ
ΔΎΉΑ÷ΟΔσΓΔΔτ÷–ΒΡ Β―ιœ÷œσΖ÷±π «ΘΚΚΎ…ΪCuO±δΈΣΚλ…ΪΘΜΑΉ…ΪΈό Υ°CuSO4ΖέΡ©±δΈΣάΕ…ΪΘΜΆ§ ±…ζ≥…“Μ÷÷Έό…ΪΤχΧεΘ§ΗΟΤχΧεΈόΈέ»ΨΓΘ
Υ°CuSO4ΖέΡ©±δΈΣάΕ…ΪΘΜΆ§ ±…ζ≥…“Μ÷÷Έό…ΪΤχΧεΘ§ΗΟΤχΧεΈόΈέ»ΨΓΘ
«κ–¥≥ωΑ±Τχ”κCuOΖ¥ ”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ
”ΠΒΡΜ·―ßΖΫ≥Χ Ϋ  ΘΜ
ΘΜ
ΔέΉΑ÷ΟΔρ÷–Φν ·Μ“ΒΡΉς”Ο « ΓΘ
Θ®2Θ©”–Ά§―ß»œΈΣΘΚNH3”κCuOΖ¥”Π…ζ≥…ΒΡΚλ…ΪΈο÷ ÷–Ω…ΡήΚ§”–Cu2OΓΘ“―÷ΣΘΚCu2O «“Μ÷÷Φν–‘―θΜ·ΈοΘΜ‘ΎΥα–‘»ή“Κ÷–Θ§Cu+ΒΡΈ»Ε®–‘±»Cu2+≤ν(Cu2O+2H+=" Cu" + Cu2+ +2H2OΘ©ΓΘ
«κΡψ…ηΦΤ“ΜΗωΦρΒΞΒΡ Β―ιΦλ―ιΗΟΚλ…ΪΈο÷ ÷– «ΖώΚ§”–Cu2OΓΘ 
ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΑΌΕ»÷¬–≈ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com