
| “߻ƼĮ |
| ÅØĮņĖį”÷ |
| “߻ƼĮ |
| ÅØĮņĖį”÷ |
| “߻ƼĮ |
| “߻ƼĮ |
| ÅØĮņĖį |
| ”÷ |
| ÅØĮņĖį |
| ”÷ |



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| “߻ƼĮ |
| ÅØĮņĖį”¢”÷ |
| “߻ƼĮ |
| “߻ƼĮ |
| ÅØH2SO4 |
| ”÷ |
| ÅØH2SO4 |
| ”÷ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| “߻ƼĮ |
| ÅØĮņĖį”÷ |
| “߻ƼĮ |
| “߻ƼĮ |
| ÅØĮņĖį |
| ”÷ |
| ÅØĮņĖį |
| ”÷ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

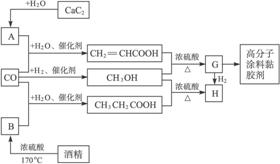
ŅŃÖŖCaC2ÓėĖ®·“Ó¦µÄ»Æѧ·½³ĢĪŖCaC2+2H2O![]() Ca(OH)2+HC”ŌCH”ü”£»Ų“šĻĀĮŠĪŹĢā£ŗ
Ca(OH)2+HC”ŌCH”ü”£»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)Š“³öGµÄ½į¹¹¼ņŹ½”£_____________________
(2)Š“³öĶ¼ÖŠÉś³ÉCH3OH(¼×“¼)µÄ»Æѧ·½³ĢŹ½”£Š“³öŌŚÅØĮņĖį×÷ÓĆĻĀÉś³ÉHµÄ»Æѧ·½³ĢŹ½”£_______________________________________________________________
(3)Öø³öÉĻŹö¹¤ŅÕµÄÓŵćŹĒŹ²Ć“£æ__________________________________________
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
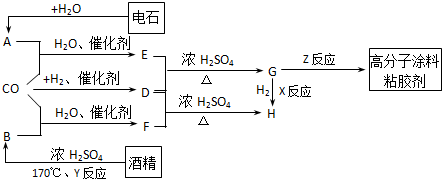
CO²»½öŹĒ¼ŅÓĆĆŗĘųµÄÖ÷ŅŖ³É·Ö£¬Ņ²ŹĒÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£ĻĀĶ¼ŹĒÓĆijŠ©¼ņµ„ÓŠ»śĪļŌŚµĶĪĀ”¢µĶŃ¹ŗĶ“߻ƼĮ“ęŌŚĻĀŗĻ³É¾ßÓŠÓÅĮ¼ŠŌÄܵÄ×°ŹĪŠŌøß·Ö×ÓĶæĮĻ”¢š¤½ŗ¼ĮµÄ»ł±¾¹ż³Ģ”£
ŅŃÖŖCaC2ÓėĖ®·“Ó¦µÄ»Æѧ·½³ĢĪŖCaC2+2H2O![]() Ca£ØOH£©2+HC”ŌCH”ü”£»Ų“šĻĀĮŠĪŹĢā£ŗ
Ca£ØOH£©2+HC”ŌCH”ü”£»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Š“³öGµÄ½į¹¹¼ņŹ½”£
£Ø2£©Š“³öĶ¼ÖŠÉś³ÉCH3OH£Ø¼×“¼£©µÄ»Æѧ·½³ĢŹ½”£Š“³öŌŚÅØĮņĖį×÷ÓĆĻĀÉś³ÉHµÄ»Æѧ·½³ĢŹ½”£
£Ø3£©Öø³öÉĻŹö¹¤ŅÕµÄÓŵćŹĒŹ²Ć“£æ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010Äź°²»ÕŹ”øßČż½Ģѧ֏Įæ¼ģ²āŹŌ¾ķ£ØČż£©ĄķæĘ×ŪŗĻ»Æѧ ĢāŠĶ£ŗĢīæÕĢā
£Ø14·Ö£©CO²»½öŹĒ¼ŅÓĆĆŗĘųµÄÖ÷ŅŖ³É·Ö£¬Ņ²ŹĒÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£ĆĄ¹ś½üÄźĄ“±Øµ¼ĮĖŅ»ÖÖµĶĪĀµĶŃ¹“߻ƹ¤ŅÕ£¬°ŃijŠ©¼ņµ„µÄÓŠ»śĪļ¾”°ōŹ»Æ”±·“Ó¦ŗóæÉŅŌ×īŗó²śÉśŅ»Ąą¾ßÓŠÓÅĮ¼ŠŌÄܵÄ×°ŹĪŠŌøß·Ö×ÓĶæĮĻ”¢Õ³ŗĻ¼ĮµČ”£ČēĻĀĶ¼ĖłŹ¾£ŗ
 |

 £Ø4£©Š“³öĻĀĮŠ×Ŗ»ÆµÄ»Æѧ·½³ĢŹ½£ŗ
£Ø4£©Š“³öĻĀĮŠ×Ŗ»ÆµÄ»Æѧ·½³ĢŹ½£ŗ²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com