
| ||


| 32m2 |
| 233m1 |
| 32m2 |
| 233m1 |
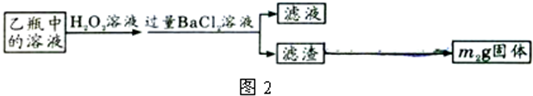
 £¬¹Ź“š°øĪŖ£ŗĖįŹ½µĪ¶Ø¹Ü£»250mLČŻĮæĘ棻
£¬¹Ź“š°øĪŖ£ŗĖįŹ½µĪ¶Ø¹Ü£»250mLČŻĮæĘ棻| 32m2 |
| 233 |
| ||
| m1 |
| 32m2 |
| 233m1 |
| 32m2 |
| 233m1 |


 µć¾¦ŠĀ½Ģ²ÄČ«Äܽā¶ĮĻµĮŠ“š°ø
µć¾¦ŠĀ½Ģ²ÄČ«Äܽā¶ĮĻµĮŠ“š°ø Š”ѧ½Ģ²ÄĶźČ«½ā¶ĮĻµĮŠ“š°ø
Š”ѧ½Ģ²ÄĶźČ«½ā¶ĮĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2007ÄźĘÕĶØøßµČѧŠ£ÕŠÉśČ«¹śĶ³Ņ»æ¼ŹŌ”¢»Æѧ(¹ć¶«¾ķ) ĢāŠĶ£ŗ038
| |||||||||||||||||||
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
(1)½«0.050 mol SO2(g)ŗĶ0.030 mol O2(g)·ÅČėČŻ»żĪŖ![]() 2SO3(g)ŌŚŅ»¶ØĢõ¼žĻĀ“ļµ½Ę½ŗā£¬²āµĆc(SO3)=0.040 mol”¤L-1”£¼ĘĖćøĆĢõ¼žĻĀSO2µÄĘ½ŗā×Ŗ»ÆĀŹ(Š“³ö¼ĘĖć¹ż³Ģ)”£
2SO3(g)ŌŚŅ»¶ØĢõ¼žĻĀ“ļµ½Ę½ŗā£¬²āµĆc(SO3)=0.040 mol”¤L-1”£¼ĘĖćøĆĢõ¼žĻĀSO2µÄĘ½ŗā×Ŗ»ÆĀŹ(Š“³ö¼ĘĖć¹ż³Ģ)”£
(2)ŅŃÖŖÉĻŹö·“Ó¦ŹĒ·ÅČČ·“Ó¦£¬µ±øĆ·“Ó¦“¦ÓŚĘ½ŗāדĢ¬Ź±£¬ŌŚĢå»ż²»±äµÄĢõ¼žĻĀ£¬ĻĀĮŠ“ėŹ©ÖŠÓŠĄūÓŚĢįøßSO2Ę½ŗā×Ŗ»ÆĀŹµÄÓŠ___________(Ģī×ÖÄø)”£
A.ÉżøßĪĀ¶Č B.½µµĶĪĀ¶Č C.Ōö“óŃ¹Ēæ D.¼õŠ”Ń¹Ēæ E.¼ÓČė“߻ƼĮ F.ŅĘ³öŃõĘų
(3)SO2Ī²ĘųÓƱ„ŗĶNa2SO3ČÜŅŗĪüŹÕæɵƵ½øüÖŲŅŖµÄ»Æ¹¤ŌĮĻ£¬·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ___________________________”£
(4)½«»ĘĢśæóµÄģŃÉÕ²śĪļFe3O4ČÜÓŚH2SO4ŗ󣬼ÓČėĢś·Ū£¬æÉÖʱøFeSO4”£ĖįČܹż³ĢÖŠŠč±£³ÖČÜŅŗ×ć¹»ĖįŠŌ£¬ĘäŌŅņŹĒ_______________________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013-2014ѧğĖÄ“ØŹ”Ć¼É½ŹŠøßČżµŚŅ»“ĪÕļ¶ĻŠŌæ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ĮņĖįŌüŹĒÓĆ»ĘĢśæóÖĘŌģĮņĖį¹ż³ĢÖŠÅųöµÄ·ĻŌü£¬Ö÷ŅŖ»Æѧ³É·ÖĪŖSiO2Ō¼45%£¬Fe2O3Ō¼40%£¬Al2O3Ō¼10%£¬MgOŌ¼5%”£ÄæĒ°ĪŅ¹śŅŃ¾ŌŚ¼¼ŹõÉĻČ”µĆĶ»ĘĘ”Ŗ”Ŗ“ÓĮņĖįŌüÖŠ·ÖĄė³öø÷Öֳɷֲ¢¼ÓŅŌĄūÓĆ”£ĘäĮ÷³ĢŗĶĻą¹ŲŹż¾ŻČēĻĀ£ŗ

²é׏ĮĻµĆÖŖ£ŗ
|
ĪļÖŹĆū³Ę |
ČܶȻż(Ksp) |
pHÖµ |
|
|
æŖŹ¼³Įµķ |
ĶźČ«³Įµķ |
||
|
Mg(OH)2 |
5.6”Į10£12 |
9.3 |
10.8 |
|
Fe(OH)3 |
2.8”Į10£16 |
2.7 |
3.7 |
|
Al(OH)3 |
1.3”Į10£33 |
3.7 |
4.7 |
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Š“³ö¹ĢĢåAµÄ»ÆѧŹ½ĪŖ ”£
£Ø2£©ČōŅŖ²ā¶ØČÜŅŗµÄpHŹĒ·ń“ļµ½3.7£¬ĻĀĮŠŹµŃéÓĆĘ·ÖŠæÉŃ”ÓƵďĒ ”£
A£®ŹÆČļŹŌŅŗ B£®¹ć·ŗpHŹŌÖ½ C£®¾«ĆÜpHŹŌÖ½ D£®pH¼Ę
£Ø3£©Ķź³ÉĻĀĮŠĄė×Ó·“Ó¦·½³ĢŹ½
¢ŁČÜŅŗDÉś³É¹ĢĢåE £» ¢ŚČÜŅŗFÉś³É¹ĢĢåG ”£
£Ø4£©ŅŖ½«¹ĢĢåC”¢¹ĢĢåEŗĶ¹ĢĢåG¶¼×Ŗ»ÆĪŖĻąÓ¦µÄĪȶØŃõ»ÆĪļ£¬Šč½ųŠŠµÄŹµŃé²Ł×÷ĪŖ ”£
£Ø5£©Čō²»æ¼ĀĒČÜŅŗĢå»żµÄ±ä»Æ£¬Ēė¼ĘĖćČÜŅŗHÖŠc£ØMg2£«£©= ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗĶ¬²½Ģā ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
»Æѧ¹¤ŅµŌŚ¹śĆń¾¼ĆÖŠÕ¼ÓŠ¼«ĘäÖŲŅŖµÄµŲĪ»£¬½ĖÕŹ”ŹĒ¹śÄŚ×īŌēµÄĮņĖįÉś²ś»łµŲÖ®Ņ»”£Ö÷ŅŖ·½³ĢŹ½ŗĶÖ÷ŅŖÉč±ø: £Ø»ĘĢśæóµÄÖ÷ŅŖ³É·ÖĪŖFeS2£© 4FeS2+11O2 = 2Fe2O3+8SO2 £Ø·ŠĢŚĀÆ£© 2SO2+O2![]() 2SO3 £Ø½Ó“„ŹŅ£© SO3+H2O=H2SO4£ØĪüŹÕĖž£©
2SO3 £Ø½Ó“„ŹŅ£© SO3+H2O=H2SO4£ØĪüŹÕĖž£©
£Ø1£©ĮņĖįÉś²śÖŠ£¬øł¾Ż»ÆŃ§Ę½ŗāŌĄķĄ“Č·¶ØµÄĢõ¼ž»ņ“ėŹ©ÓŠ £ØĢīŠ“ŠņŗÅ£©”£
A£®æóŹÆ¼ÓČė·ŠĢŚĀÆÖ®Ē°ĻČ·ŪĖé B£®½Ó“„ŹŅµÄ·“Ó¦Ź¹ÓĆV2O5×÷“߻ƼĮ
C£®½Ó“„ŹŅÖŠ²»Ź¹ÓĆŗÜøßµÄĪĀ¶Č D£®¾»»ÆŗóĀÆĘųÖŠŅŖÓŠ¹żĮæµÄæÕĘų
E£®½Ó“„ŹŅÖŠµÄŃõ»ÆŌŚ³£Ń¹ĻĀ½ųŠŠ F£®ĪüŹÕĖžÖŠÓĆ98.3%µÄÅØĮņĖįĪüŹÕSO3
£Ø2£©0.1mol/LµÄNaHSO3ČÜŅŗÖŠ£¬ÓŠ¹ŲĮ£×ÓÅضČÓɓ󵽊”µÄĖ³ŠņĪŖ£ŗ
c £ØNa+£©£¾c £ØHSO3££©£¾c £ØSO32££©£¾c £ØH2SO3£©
¢ŁøĆČÜŅŗÖŠc £ØH+£© c £ØOH££©£ØĢī”°£¾”±”¢”°£½”± »ņ”°£¼”± £©£¬ĘäĄķÓÉŹĒ£ØÓĆĄė×Ó·½³ĢŹ½±ķŹ¾£©£ŗ ”£
¢ŚĻÖĻņNaHSO3ČÜŅŗÖŠ£¬ÖšµĪ¼ÓČėÉŁĮæŗ¬ÓŠ·ÓĢŖµÄNaOHČÜŅŗ£¬æɹŪ²ģµ½µÄĻÖĻóŹĒ £¬·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com