
Š“³öĻĀĮŠø÷ÓŠ»śĪļµÄ½į¹¹¼ņŹ½£ŗ
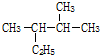
£Ø1£©µ±0.2molĢžAŌŚŃõĘųÖŠĶźČ«Č¼ÉÕŹ±Éś³ÉCO2ŗĶH2Oø÷1.2mol£¬“߻ƼÓĒāŗóÉś³É2£¬2-¶ž¼×»ł¶”Ķ飬ŌņAµÄ½į¹¹Ź½ĪŖ__________________”£
£Ø2£©Ä³Č²ĢžŗĶH2³ä·Ö¼Ó³ÉÉś³É2£¬5-¶ž¼×»łŅŃĶ飬øĆČ²ĢžµÄ½į¹¹¼ņŹ½ĪŖ______________”£
£Ø3£©Ä³Ģž1molÓė2mol HClĶźČ«¼Ó³É£¬Éś³ÉµÄĀČ“śĶéĢž×ī¶ą»¹æÉŅŌÓė4molĀČĘų·“Ó¦£¬ŌņøĆĢžµÄ½į¹¹¼ņŹ½ĪŖ________________”£
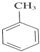
£Ø4£©Ä³ĶéĢžAÕōĘųµÄĆܶȏĒĻąĶ¬×“æöĻĀĒāĘųĆܶȵÄ64±¶£¬¾²ā¶ØµĆÖŖA·Ö×ÓÖŠ¹²ŗ¬6øö¼×»ł”£ČōA²»æÉÄÜŹĒĻ©ĢžÓėĒāĘų¼Ó³ÉµÄ²śĪļ£¬AµÄ½į¹¹¼ņŹ½ĪŖ_____________”£

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ


| ³żŌÓŹŌ¼Į | ·½·Ø | |
| C2H6 £ØC2H2£© | ||
| C6H6£ØC6H5OH£© |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ


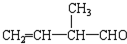 ŗ¬Ńõ¹ŁÄÜĶŵÄĆū³ĘŹĒ
ŗ¬Ńõ¹ŁÄÜĶŵÄĆū³ĘŹĒ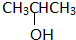
 +3HNO3
+3HNO3
| ||
| ¼ÓČČ |
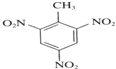 +3H2O£®
+3H2O£® +3HNO3
+3HNO3
| ||
| ¼ÓČČ |
 +3H2O£®
+3H2O£®| ¼ÓČČ |
| ¼ÓČČ |
| “¼ |
| ¼ÓČČ |
| “¼ |
| ¼ÓČČ |
| ¼ÓČČ |
| ¼ÓČČ |


²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
(10·Ö)Š“³öĻĀĮŠø÷ÓŠ»śĪļµÄ½į¹¹¼ņŹ½£ŗ
£Ø1£©µ±0.2 molĢžAŌŚŃõĘųÖŠĶźČ«Č¼ÉÕŹ±Éś³ÉCO2ŗĶH2Oø÷1.2 mol£¬“߻ƼÓĒāŗóÉś³É2£¬2-¶ž¼×»ł¶”Ķ飬ŌņAµÄ½į¹¹Ź½ĪŖ___________”£
£Ø2£©·Ö×ÓŹ½ĪŖC6H12µÄijĻ©ĢžµÄĖłÓŠµÄĢ¼Ō×Ó¶¼ŌŚĶ¬Ņ»Ę½ĆęÉĻ£¬ŌņøĆĻ©ĢžµÄ½į¹¹¼ņŹ½ĪŖ______________________”£
£Ø3£©Ä³ĶéĢžAÕōĘųĆܶȏĒĻąĶ¬×“æöĻĀĒāĘųĆܶȵÄ64±¶£¬¾²ā¶ØÖŖA·Ö×ÓÖŠ¹²ŗ¬6øö¼×»ł”£
¢ŁČōA²»æÉÄÜŹĒĻ©ĢžÓėĒāĘų¼Ó³ÉµÄ²śĪļ£¬AµÄ½į¹¹¼ņŹ½ĪŖ______________________”£
¢ŚČōAŹĒČ²ĢžÓėĒāĘų¼Ó³ÉµÄ²śĪļ£¬AµÄ½į¹¹¼ņŹ½ĪŖ______________________”£
£Ø4£©Ä³ŗ¬ŃõÓŠ»ś»ÆŗĻĪļ£¬ĖüµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ88.0£¬ŗ¬CµÄÖŹĮæ·ÖŹżĪŖ68.2%£¬ŗ¬HµÄÖŹĮæ·ÖŹżĪŖ13.6%£¬¾ŗģĶā¹āĘײā¶Øŗ¬ÓŠŅ»øöōĒ»ł£¬ŗĖ“Ź²ÕńĒāĘ×ĻŌŹ¾øĆ·Ö×ÓÖŠÓŠ3øö¼×»ł£¬ĒŅÓŠČżÖÖ²»Ķ¬»·¾³µÄĒāŌ×Ó£¬ĒėŠ“³öĘä½į¹¹¼ņŹ½_____________________.
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģ¹óÖŻŹ”ø߶žĻĀѧʌµŚŅ»“ĪŌĀæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
(10·Ö)Š“³öĻĀĮŠø÷ÓŠ»śĪļµÄ½į¹¹¼ņŹ½£ŗ
£Ø1£©µ±0.2 molĢžAŌŚŃõĘųÖŠĶźČ«Č¼ÉÕŹ±Éś³ÉCO2ŗĶH2Oø÷1.2 mol£¬“߻ƼÓĒāŗóÉś³É2£¬2-¶ž¼×»ł¶”Ķ飬ŌņAµÄ½į¹¹Ź½ĪŖ___________”£
£Ø2£©·Ö×ÓŹ½ĪŖC6H12µÄijĻ©ĢžµÄĖłÓŠµÄĢ¼Ō×Ó¶¼ŌŚĶ¬Ņ»Ę½ĆęÉĻ£¬ŌņøĆĻ©ĢžµÄ½į¹¹¼ņŹ½ĪŖ______________________”£
£Ø3£©Ä³ĶéĢžAÕōĘųĆܶȏĒĻąĶ¬×“æöĻĀĒāĘųĆܶȵÄ64±¶£¬¾²ā¶ØÖŖA·Ö×ÓÖŠ¹²ŗ¬6øö¼×»ł”£
¢ŁČōA²»æÉÄÜŹĒĻ©ĢžÓėĒāĘų¼Ó³ÉµÄ²śĪļ£¬AµÄ½į¹¹¼ņŹ½ĪŖ______________________”£
¢ŚČōAŹĒČ²ĢžÓėĒāĘų¼Ó³ÉµÄ²śĪļ£¬AµÄ½į¹¹¼ņŹ½ĪŖ______________________”£
£Ø4£©Ä³ŗ¬ŃõÓŠ»ś»ÆŗĻĪļ£¬ĖüµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ88.0£¬ŗ¬CµÄÖŹĮæ·ÖŹżĪŖ68.2%£¬ŗ¬HµÄÖŹĮæ·ÖŹżĪŖ13.6%£¬¾ŗģĶā¹āĘײā¶Øŗ¬ÓŠŅ»øöōĒ»ł£¬ŗĖ“Ź²ÕńĒāĘ×ĻŌŹ¾øĆ·Ö×ÓÖŠÓŠ3øö¼×»ł£¬ĒŅÓŠČżÖÖ²»Ķ¬»·¾³µÄĒāŌ×Ó£¬ĒėŠ“³öĘä½į¹¹¼ņŹ½_____________________.
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com