
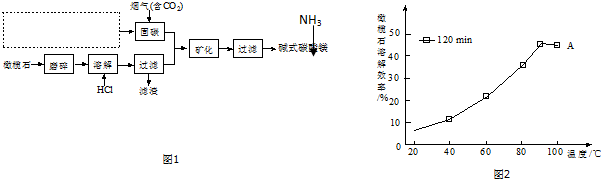
 ��
������ ������ͼ����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH��aq��+CO2 ��g��=NaHCO3 ��aq������ҵ�����ȵ�ⱥ��ʳ��ˮ�����ռȱ���ռ����ȡ���̣��Ȼ�����Һ���õ�����������������������Һ����ѧ��Ӧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2������������������Һ��̼������̼�����Σ����ʯ����Ҫ�ɷ�ΪMg2SiO4�����Կ���MgO��SiO2��ɣ������ʯĥ����ٽ����ܽ��Ŀ��������Ӵ�������ӿ��ܽ����ʣ�ĥ����������ܽ⣬Mg2SiO4+4HCl�T2MgCl2+SiO2+2H2O�����˵õ���Һ�Ȼ�þ�������������裬��Ͽ�3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+���õ���ʽ̼��þ��
��1��������ͼ����̼ʱ���������ƣ���ҵ�����ȵ�ⱥ��ʳ��ˮ����ռ
��2�����ʯ����Ҫ�ɷ�ΪMg2SiO4�����Կ���MgO��SiO2��ɣ����������������Ӧ��
��3����ӦΪþ���Ӻ�̼��������ӷ�Ӧ���ɼ�ʽ̼��þ��
��4�������ܺͶ�����̼֮�䷴Ӧ�����������̶�������̼���ش�
��5�������¶ȶԻ�ѧ��Ӧƽ���ƶ���Ӱ��֪ʶ���ش�
��6�������ӵļ����������ữ����������
��� �⣺��1����������ͼ����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH��aq��+CO2 ��g��=NaHCO3 ��aq����ȱ���ռ����ȡ���̣���ҵ�����ȵ�ⱥ��ʳ��ˮ�����ռ��ѧ��Ӧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2��������ͼΪ ��
��
�ʴ�Ϊ�� ��
��
��2�����ʯ����Ҫ�ɷ�ΪMg2SiO4�����Կ���MgO��SiO2��ɣ������ʯĥ����ٽ����ܽ��Ŀ��������Ӵ�������ӿ��ܽ����ʣ�ĥ����������ܽ⣬Mg2SiO4+4HCl�T2MgCl2+SiO2+2H2O�����˵õ���Һ�Ȼ�þ�������������裬
�ʴ�Ϊ��SiO2��
��3�����ʯ�������ܽ⣬���˵õ���Һ�Ȼ�þ�����̼��IJ����Ͽ�������ӦΪ��3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+���õ���ʽ̼��þ��
�ʴ�Ϊ��3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+��
��4�������������У�ֻ�У�NH4��2CO3���ԺͶ�����̼֮�䷴Ӧ����̼����泥�����������̼�����Լ���
�ʴ�Ϊ��c��
��5��1 ͼ����ʾ�����Լ����߱仯֪����20min���ܽ�ﵽƽ�⣬���÷�Ӧ�Ƿ��ȣ����£�ƽ�������ƶ������ܽ�Ч�ʽ��ͣ�
�ʴ�Ϊ��120min���ܽ�ﵽƽ�⣬����Ӧ���ȣ�����ƽ�������ƶ����ܽ�Ч�ʽ��ͣ�
��6���жϲ�Ʒϴ��ֻ��Ҫ����ϴ��Һ�в����������Ӽ��ɣ������ӵļ����������ữ���������������ǣ�ȡ�������һ�ε�ϴ��Һ���������ữ����������Һ������������������ϴ����
�ʴ�Ϊ��ȡ�������һ�ε�ϴ��Һ���������ữ����������Һ������������������ϴ����
���� ������һ����ѧ��������ϵĹ��������⣬�����ڿ��Ե��ȵ㣬ע��֪ʶ��Ǩ�ƺ����Ӧ���ǽ���Ĺؼ�����Ŀ�Ѷ��еȣ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Zn��Fe | B�� | Zn��Cu | C�� | Fe��Cu | D�� | Zn��Fe��Cu |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | b��a��c | B�� | a��c��b | C�� | c��a��b | D�� | c��b��a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
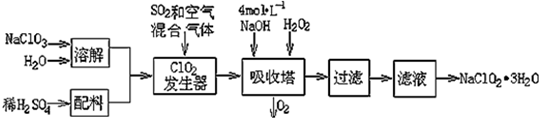
| ���� | HClO2 | HF | HCN | H2S |
| Ka | 1��10-2 | 6.3��10-4 | 4.9��10-10 | K1=9.1��10-8 K2=1.1��10-12 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
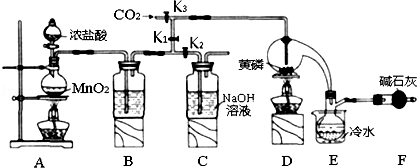
| ���� | �۵�/�� | �е�/�� |
| PCl3 | -112 | 75.5 |
| POCl3 | 2 | 105.3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ����ȩ��
����ȩ��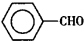 ���������ᣨ
���������ᣨ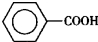 ��������Ҫ�Ļ���ԭ�ϣ������üױ���
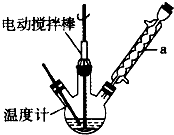
��������Ҫ�Ļ���ԭ�ϣ������üױ���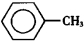 ��Ϊԭ���������±��г����й����ʵIJ����������ʣ���ش�
��Ϊԭ���������±��г����й����ʵIJ����������ʣ���ش�| ���� | ��״ | �۵㣨�棩 | �е㣨�棩 | ����ܶ� ��ˮ=1g/cm3 | �ܽ��� | |
| ˮ | �Ҵ� | |||||
| �ױ� | ��ɫҺ����ȼ�ӷ� | -95 | 110.6 | 0.8660 | ���� | ���� |
| ����ȩ | ��ɫҺ�� | -26 | 179 | 1.0440 | �� | ���� |
| ������ | ��ɫƬ״����״���� | 122.1 | 249 | 1.2659 | �� | ���� |
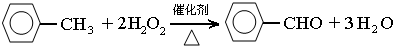 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
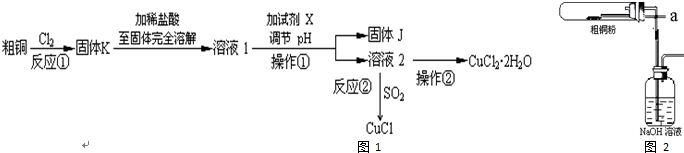
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����


�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com