
�Ը������������Ĺ�ҵ��ҺΪԭ�������������Ĺ�������(���ֲ�����������)��
��.�ӷ�Һ���ᴿ���ᾧ��FeSO47H2O��
��.��FeSO47H2O���Ƴ���Һ��
��.FeSO4��Һ���Թ�����NH4HCO3��Һ��ϣ��õ���FeCO3����Һ��
��.����Һ���ˣ���90����ˮϴ�ӳ����� �����õ�FeCO3���塣
�����õ�FeCO3���塣
��.����FeCO3���õ�Fe2O3���塣
��֪��NH4HCO3����ˮ�зֽ⡣
(1)���У�����������м��ȥ��Һ�е�Fe3+���÷�Ӧ�����ӷ���ʽ��_________________��
(2)���У����һ�������ᡣ���û�ѧƽ��ԭ���������������_____________��
(3)���У�����FeCO3�����ӷ���ʽ��_____________����FeCO3��Һ��ʱ�䱩¶�ڿ����У����в��ֹ�������Ϊ���ɫ���ñ仯�Ļ�ѧ����ʽ��_____________��
(4)���У�ͨ������SO42-���жϳ����Ƿ�ϴ�Ӹɾ�������SO42-�IJ�����_____________��
(5)��֪����FeCO3�Ļ�ѧ����ʽ��4FeCO3+O2 2Fe2O3+4CO2��������464.0kg��FeCO3,�õ�316.8kg��Ʒ������Ʒ������ֻ��FeO����ò�Ʒ��Fe2O3��������_________kg��(Ħ������/gmol-1:FeCO3116Fe2O3160FeO72)
2Fe2O3+4CO2��������464.0kg��FeCO3,�õ�316.8kg��Ʒ������Ʒ������ֻ��FeO����ò�Ʒ��Fe2O3��������_________kg��(Ħ������/gmol-1:FeCO3116Fe2O3160FeO72)
 ѧ���������ν��Ͼ���ѧ������ϵ�д�
ѧ���������ν��Ͼ���ѧ������ϵ�д� Happy holiday���ּ��������ҵ�㶫���������ϵ�д�
Happy holiday���ּ��������ҵ�㶫���������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2017�����ʡТ���и����ϵ�һ��ͳ����ѧ�Ծ��������棩 ���ͣ�ʵ����
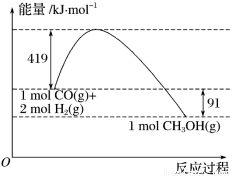
�״�����Ҫ�Ĺ�ҵԭ�ϡ�ú������������ú̿��ȡˮú���Ӷ��ϳɼ״���
CO(g)+2H2(g) CH3OH(g)��
CH3OH(g)��
��֪�ٳ�ѹ�·�Ӧ�������仯��ͼ��ʾ��
��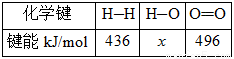
��CO(g)��1/2O2(g)===CO2(g) ��H=��280kJ/mol
H2(g)��1/2O2(g)===H2O(l) ��H=��284kJ/mol
H2O(l)===H2O(g) ��H=+ 44kJ/mol
��ش��������⣺
(1)��д����ʾ��̬�״�ȼ���ȵ��Ȼ�ѧ����ʽ_________________________��
(2)H��O���ļ���xΪ_________kJ/mol��
(3)�״�����ֽ�ΪCO��H2��������ķ�Ӧ�Ļ��Ϊ_________kJ/mol��
(4)��________(����¡����¡�)�����������CO��H2�Ʊ��״��ķ�Ӧ�Է����С�
(5)ij��ȤС��ͬѧ���ü״�ȼ�ϵ��̽���縡ѡ��������ˮ��һ�ַ�ʽ��������ˮ��pH��5.0��6.0֮�䣬ͨ���������Fe(OH )3������Fe(OH)3���������ԣ�������������������������о���ˮ�����á�װ������ͼ��ʾ��
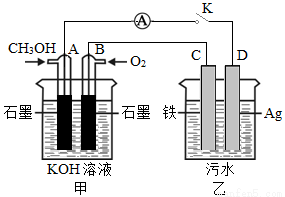
��д���׳ص�A���缫��Ӧʽ��______________________��
�����ҳ�ʵ��ʱ��ˮ������Ũ�Ƚ�С�����������ϲ��ˮЧ�����ã���ʱӦ����ˮ�м���������_______��
A��H2SO4
B��BaSO4
C��Na2SO4
D��NaOH
E��CH3CH2OH
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꽭��ʡ�����и߶���ѧ��ѧҵģ������ѧ�Ծ��������棩 ���ͣ������
�Ų�����������������ͷ�չ����Ҫ���ʻ�����
�� ����ˮ�����Ҫԭ��������� (������)��
�� ����������̥����Ҫԭ�ϣ���Ȼ��ͨ����ʩ������ǿ�Ⱥ͵��ԣ�������� �ṹ��(ѡ����͡������͡�)��
�����彡����ʳƷ��ϵ���С�
�� ��Щѧ����Сƫʳ��������ˮ�����߲ˣ����Ӫ��ȱ������������������Ҫ��������ȡ (ѡ�֬��������ά���ء������ʡ�)��������ġ�
�� ������Ӧ��������ȡ��������Ԫ�أ����� Ԫ�غ������ͣ��������״�ټ�����
�� ���ࡢ��֬�������ʶ�����������Ӫ�����ʡ����е����ʱ������������ø��������ˮ��Ϊ________ (д����)��
�����彡����ҩ��ȹ�ϵ���У�
���˵Ĵ���θҺ��һ����ɫҺ�壬�����ԣ�pH��0.9��1.5֮�䣬����H+��Ũ�ȱ�ѪҺ�и����ش��������⣺��θҺ�а�����������ʳ��ȶ��ֹ��ܣ�θ�����Ҫ�ɷ���
����θ���������ͻỼθ�����֢���������ʲ�����������θ������ҩ����
A��С�մ� B���������� C���������� D��̼��þ
��θ��ƽ����Ҫ�ɷ�������������������ˮ�����ó־ã�д����ҩ���к�θ������ӷ���ʽ ��
�� ���������в��˽�����ʹ��ҩ���� (ѡ���ù�ء���˾ƥ�֡�)��
�Ƚྻ��ȫ�����滷�����ǽ����ı��ϡ�
��ijũ����������ֺ���Ϊ�������ˮ���ڽ��ر�ˮȡ�ؼҺ��ȼ����� �� �ٹ��ˣ�ȥ��������ˮ�еĽϴ���������˺�ʹ��Ư�۽���ɱ����������ԭ�����û�ѧ����ʽ��ʾΪ ��
������������������Һ��Ӧ���Ƶ�Ư�ۡ���д����ҵ��Ư�۷�Ӧ�Ļ�ѧ����ʽ�� ��
��ͨ��������ר�������䣬�����ϱ�����ͼ��ʾ����ͼͼ���ʾ ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�꽭��ʡ�����и߶���ѧ��ѧҵģ������ѧ�Ծ��������棩 ���ͣ�ѡ����
���г������ʵ������뻯ѧʽ��Ӧ��ȷ����
A���ռNaOH B��С�մ�Na2SO4
C����ʯ�ҡ�CaCl2 D��������Al2(SO4)3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ���������ݸ�����һ12���¿���ѧ���������棩 ���ͣ�������
��֪������H2���ܶ���0.893gL��1��ij�ձ��м���50.0mLˮ��������һС������ƣ���Ӧ��ȫ����ȴ����������Һ��������Ϊ52.2g������
(1)������Ƶ�������
(2) ����H2�ڳ����µ������
����H2�ڳ����µ������
(3)������Һ��NaOH������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ���������ݸ�����һ12���¿���ѧ���������棩 ���ͣ��ƶ���
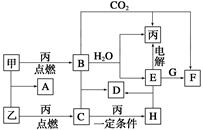
�ס��ҡ���Ϊ�������ʡ�A��B��C��D��E��F��G��H��Ϊ��ѧ��ѧ�г����Ļ��������B��G����ɫ��Ӧ��Ϊ��ɫ��C��ʹƷ����Һ��ɫ����һ�������£��������ת����ϵ��ͼ��ʾ��
��ش��������⣺
(1)�û�ѧʽ��ʾ����Ϊ__________��HΪ__________��
(2)A�ĵ���ʽΪ________________________________��
(3)���E��ˮ��Һʱ��E��������________________________��
(4)д��B��C����D�Ļ�ѧ����ʽ��_____________________________��
д��E��G����F�����ӷ���ʽ��__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ���������ݸ�����һ12���¿���ѧ���������棩 ���ͣ�ѡ����
��Na2CO3��NaAlO2�Ļ����Һ����μ���150mL1molL��1HCl��Һ�������Һ�е�ij�����������ʵ����ı仯��ͼ��ʾ��������˵������ȷ����
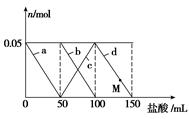
A��a���߱�ʾ�����ӷ���ʽΪ��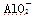 ��H����H2O===Al(OH)3��
��H����H2O===Al(OH)3��
B��b��c���߱�ʾ�����ӷ�Ӧ����ͬ��
C��ԭ�����Һ�е�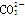 ��
��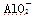 �����ʵ���֮��Ϊ1��2
�����ʵ���֮��Ϊ1��2
D��M��ʱ����Һ�г���������Ϊ3.9g
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ�����ʡ�����и�������һ12���¿���ѧ�Ծ��������棩 ���ͣ�������
��amolNH3��O2��ɵĻ���������ܱշ�Ӧ���У���һ�������·�����ѧ��Ӧ�����������O2�����ʵ���Ϊx������HNO3�����ʵ���Ϊy����ȷ��x�ڲ�ͬȡֵ��Χʱy=f��x���ĺ�������ʽ��������������ͼ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016-2017ѧ����������������и߶�����ĩ��ѧ���������棩 ���ͣ�ѡ����
��֪25 ��ʱ��CaSO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ������������100 mL��CaSO4������Һ�У�����200 mL 0.03 mol��L ��1 ��Na2SO4��Һ����Դ˹��̵�����������ȷ����(���Ի�Ϲ����е�����仯)
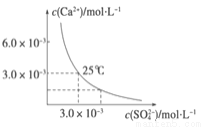
A����Һ������CaSO4������������Һ��c(SO42��)��ԭ����
B ��Һ������CaSO4��������Һ��c(Ca2�� )��c(SO 42��)����С
C����Һ��������������Һ��c(Ca2�� )��c(SO42��)����С
D����Һ��������������������Һ��c(SO42��)��ԭ����
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com