

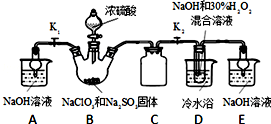
���� ��ҵ�ϳ����ú����ˮ����Na2S2O3•5H2O������װ��A�Ʊ��������ѷ�Һ©���е�Ũ���������ƿ�����������з�Ӧ�õ������������壬��������ͨ��Bװ�ã�װ��B������֮һ�ǹ۲�SO2���������ʣ� ʵ����ΪʹSO2����������ƿC����Ҫ���ƿ��Ƶμ���������ʣ�ͨ��װ��C������Ӧ��ƿC�з�����Ӧ���£�
Na2S��aq��+H2O��l��+SO2��g���TNa2SO3��aq��+H2S��aq�� ����
2H2S��aq��+SO2��g���T3S��s��+2H2O��l�� ����
S��s��+Na2SO3��aq��$\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq�� ����
��Ӧ��ֹ����ƿC�е���Һ������Ũ����������Na2S2O3•5H2O���õ���������ƣ�װ��D�Ƿ�ֹ�����İ�ȫƿ��װ��E������ʣ�����壬��ֹ��Ⱦ������
��1��Һ���߶ȱ��ֲ��䣬˵�����������ã�D�����Ϊ�̵��ܣ�Ϊ��ȫƿ����ֹ������װ��E������β����SO2��H2S�����ã�
��2��������Ŀ����3����Ӧ���ɵó���Ӧ��ϵ��2Na2S��2H2S��3S��3 Na2SO3��2Na2S��Ӧʱͬʱ����2Na2SO3������Ҫ1Na2SO3��
��3���۲�SO2���������ʣ�����ǿ����ȡ����ķ�Ӧ�����з���S��g��+Na2SO3��aq��$\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq������Ӧ�ﵽ�յ���S��ȫ�ܽ⣬�ձ�����ƿ��Ҫ����ʱ����ʹ��ʯ�������������Թܿ�ֱ�Ӽ��ȣ�
��4���������ᣬNa2S2O3��Ӧ����S���������Ծ��ú�ȡ�ϲ���Һ���μ�BaCl2��Һ��������Һ���Ƿ�����������ӣ�
��� �⣺��1��������װ��ɺر����˻�������װ��B�еij���©����ע��Һ�����γ�һ��Һ������Һ���߶ȱ��ֲ��䣬�����������ã�D�����Ϊ�̵��ܣ�Ϊ��ȫƿ����ֹ������װ��E������β����SO2��H2S�����ã���ѡ��NaOH��Һ��
�ʴ�Ϊ��Һ���߶ȱ��ֲ��䣻��ֹ������NaOH��
��2��������Ŀ����3����Ӧ���ɵó���Ӧ��ϵ��2Na2S��2H2S��3S��3 Na2SO3��2Na2S��Ӧʱͬʱ����2Na2SO3������Ҫ1Na2SO3��������ƿC��Na2S��Na2SO3���ʵ���֮��Ϊ2��1��
�ʴ�Ϊ��2��1��
��3���۲�SO2���������ʣ�����ǿ����ȡ����ķ�Ӧ��a�����ɶ�������bd�����ʾ����������Ӧ��ֻ��c�б���NaHSO3��Һ�ʺ���ȡ���������з���S��g��+Na2SO3��aq��$\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq������Ӧ�ﵽ�յ���S��ȫ�ܽ⣬�ɹ۲쵽��Һ����壨�������ʧ�����ձ�����ƿ��Ҫ����ʱ����ʹ��ʯ������
�ʴ�Ϊ��c�����Ƶμ�������ٶȣ���Һ����壨�������ʧ����ad��
��4������Ʒ���Ƿ����Na2SO4��ʵ�鷽��Ϊ��ȡ������Ʒ��������ϡ���ᡢ���á�ȡ�ϲ���Һ������ˣ�ȡ��Һ�����μ�BaCl2��Һ�������ֳ�����˵������Na2SO4���ʣ�
�ʴ�Ϊ��ȡ������Ʒ��������ϡ���ᡢ���á�ȡ�ϲ���Һ������ˣ�ȡ��Һ�����μ�BaCl2��Һ�������ֳ�����˵������Na2SO4���ʣ�
���� ���⿼��ʵ�鷽���ķ��������ۣ��漰�����Լ��顢���Ӽ��顢�Բ����ķ������۵ȣ�����ʵ�����������֪ʶ�ۺ�Ӧ�������Ŀ��飬���������ܷ�Ӧ�ķ�������Ŀ�Ѷ��еȣ�


 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �����Ƽ��������ȡ���չ����о�Ӧ���������ܽ�ȵIJ��� | |
| B�� | ʹ�ÿ�������Դ���ó��ٽ������̼����л��ܼ���ע��ԭ�ӵľ����ԡ����õ��ܺ��������յȶ�����ɫ��ѧ������ | |
| C�� | ʯ�ͷ���ú����������ˮɹ�Ρ���ȥ���ۡ���������ȡ�����͵ȹ��̶��������仯 | |
| D�� | �Ͻ�������ָ�ۺ����ֻ��������ϵĽ���Ԫ�ز��γɾ��г������Եij������ϣ�����һ�ѣ�Nb-Ti���Ͻ������۵�һ��ȴ���Ҫ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
 ��ͼ��ʾ����A�г���1mol X��1mol Y����B�г���2mol X��2mol Y����ʼʱV��A��=V��B��=a L������ͬ�¶Ⱥ��д����������£��������и��Է������з�Ӧ��X��g��+Y��g��?2Z��g��+W��g������H��0���ﵽƽ��ʱ��V��A��=1.1a L��
��ͼ��ʾ����A�г���1mol X��1mol Y����B�г���2mol X��2mol Y����ʼʱV��A��=V��B��=a L������ͬ�¶Ⱥ��д����������£��������и��Է������з�Ӧ��X��g��+Y��g��?2Z��g��+W��g������H��0���ﵽƽ��ʱ��V��A��=1.1a L��| A�� | ��Ӧ��ʼʱ��B�����л�ѧ��Ӧ���ʱ�A�����п� | |
| B�� | A������X��ת����Ϊ20%���ұ�B������X��ת����С | |
| C�� | ��Kһ��ʱ�䣬����ƽ��ʱ��A�����Ϊ2.3a L����ͨ��������������ƣ� | |
| D�� | ��K����ƽ��ʱ������B�������¶ȣ�A��������������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��״���£�1molH2O�����ԼΪ22.4L | |
| B�� | 1mol/L��CaCl2��Һ�к�Cl-����ĿΪ2 NA | |
| C�� | ���³�ѹ�£�17g NH3����ԭ����ĿΪ3NA | |
| D�� | ��״���£�2.24 LCl2���ɵ���ˮ�к���0.1NA��Cl2���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
 ������ṹ��ʽΪ����ʽ�����磺CH3CH=CHCH2Cl�ļ���ʽ��дΪ
������ṹ��ʽΪ����ʽ�����磺CH3CH=CHCH2Cl�ļ���ʽ��дΪ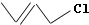 ������������ѧ֪ʶ�жϣ��ϳ�̼��άR�ĵ������п��ܵ��ǣ�������
������������ѧ֪ʶ�жϣ��ϳ�̼��άR�ĵ������п��ܵ��ǣ�������| A�� | CH2=CH2��N2 | B�� | CH3-C��C-NH2 | C�� | HC��C-C��N | D�� | CH2=CH-CH=NH |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��״���£�����NA����ԭ�ӵĺ��������ԼΪ11.2L | |
| B�� | ���³�ѹ�£�64g SO2���е�ԭ����Ϊ3NA | |
| C�� | ���³�ѹ�£�11.2L Cl2���еķ�����Ϊ0.5NA | |
| D�� | ��״���£�11.2L H2O���еķ�����Ϊ0.5NA |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com