
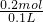 =2mol/L��
=2mol/L��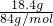 =0.219mol��NaHCO3��ȫ��Ӧ������̼Ԫ���غ㣬��֪���ɶ�����̼n��CO2��=n��NaHCO3��=0.219mol���ٶ�̼������ȫ��KHCO3����n��KHCO3��=
=0.219mol��NaHCO3��ȫ��Ӧ������̼Ԫ���غ㣬��֪���ɶ�����̼n��CO2��=n��NaHCO3��=0.219mol���ٶ�̼������ȫ��KHCO3����n��KHCO3��=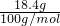 =0.184mol��KHCO3��ȫ��Ӧ������̼Ԫ���غ㣬��֪���ɶ�����̼n��CO2��=n��KHCO3��=0.184mol������ʵ�ʶ�����̼�����ʵ���Ϊ0.184mol��n��CO2����0.219mol��
=0.184mol��KHCO3��ȫ��Ӧ������̼Ԫ���غ㣬��֪���ɶ�����̼n��CO2��=n��KHCO3��=0.184mol������ʵ�ʶ�����̼�����ʵ���Ϊ0.184mol��n��CO2����0.219mol��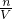 ����Ԫ�ص�Ũ�ȣ�
����Ԫ�ص�Ũ�ȣ�

 ÿ�α���ϵ�д�
ÿ�α���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
����ѡ����ר��ѵ��
2005��2��18��Ӣ��ʳƷ�����ʳ�ú��������յ���ɫ�ص�ʳƷ�����շ���֢�������߷������档�ҹ�Ҳ�ѿ�ʼ�麬�յ���ɫ�ص�ʳƷ����ν���յ���һ�š�ʵ����һ���Ϊ���յ�һ�š�����һ�ֻ��ܼ�Ⱦ�ϣ�����ѧȾ�Ϸ����Ϊ�ܼ���14��C.L.Solvent��Yellow14��������ʽΪC16H12N2O���仯ѧ�ṹ��ż����Ⱦ�ϣ����ɱ����ص�������2-����ż���Ƶá��й��յ���һ������˵����ȷ���ǣ� ��
A���յ���һ����ʳƷ��������ɫ�����ã��������������������Ӽ�
B���յ���һ�ŵ�Ħ������Ϊ248
C���յ���һ�ſ������ܽ�������͡�����Ь�͵Ȳ�Ʒ��Ⱦɫ
D��ʳƷ���Ӽ�������Σ���ϴ����Ӧȫ���ֹʹ�á�
NAΪ����ӵ�������������ȷ���� �� ��
A��80g����狀��е�ԭ����Ϊ2NA
B��1L 1mol/L��������Һ�У������Ȼ��������ΪNA
C����״���£�11.2L���Ȼ�̼����������Ϊ0.5 NA
D����ͭ����ķ�Ӧ�У�1molͭʧȥ�ĵ�����Ϊ2 NA
��pH =1����ɫ��Һ���ܴ���������������� �� ��
A��NH4+��Mg2+��SO42����Cl�� B��Ba2+��K+��OH����NO3��
C��Al3+��Cu2+��SO42����Cl�� D��Na+��Ca2+��Cl����AlO2��
����������һ���Ч���Ͷ������������ǿ
�Ҵ�ɱ���õ�ɱ��������жԹ��ȶ�������
�����Ľṹ��ʽ����ͼ��
���жԸû�����������ȷ���ǣ� ��
A�����ڷ����� B������±����
C�������������²�ˮ�� D����һ�������¿��Է����ӳɷ�Ӧ
���Խ���ӦZn+Br2=ZnBr2��Ƴ����أ�����4����Ӧ ��Br2+2e=2Br-��2Br��2e=Br2��Zn-2e=Zn2+��Zn2++2e=Zn���б�ʾ���ʱ��������Ӧ�ͷŵ�ʱ�ĸ�����Ӧ����
A �ں͢� B �ں͢� C �ۺ͢� D �ܺ͢�
NaH�ǡ������ӻ��������ˮ��Ӧ�ķ���ʽΪ��NaH��H2O��NaOH��H2������Ҳ�ܸ�Һ�����Ҵ��ȷ������Ƶķ�Ӧ���������������������й�NaH�������������( )
A ��ˮ��Ӧʱ��ˮ�������� B NaH��H���뾶��Li���뾶С
C ��Һ����Ӧʱ����NaNH2���� D ���Ҵ���Ӧʱ��NaH������
��ͼ�ǵ��CuCl2��Һ��װ�ã�����c��dΪʯī�缫���������йص��ж���ȷ���ǣ� ��
A a������b���� B a������b����
C �������У�d�缫��������
D �������У�������Ũ�Ȳ���
����ʵ������в������������Ӧ��ͼ������ϵ���
A NaHSO3��ĩ����HNO3��Һ�� B H2S����ͨ����ˮ��


C NaOH��Һ����Ba(HCO3)2��Һ�� D CO2����ͨ�����ʯ��ˮ��


��һ�ֹ����͵����������������绷�����˵�Ҫ��ClO2����ȡ��Cl2��Ϊ��������ˮ������������ҵ��ClO2����NaClO3��Na2SO3��Һ��ϲ���H2SO4�ữ��Ӧ�Ƶã������Ϸ�Ӧ��NaClO3��Na2SO3�����ʵ���֮��Ϊ�� ��

A 1�U1 B 2�U1 C 1�U2 D 2�U3
��Ȼά����P���ṹ��ͼ�������ڻ��������У�����һ��Ӫ��������������ά����P�������������
A ���Ժ���ˮ��Ӧ B �����л��ܼ���ȡ
C ���������������� D 1molά����P���Ժ�4molNaOH��Ӧ
��ͼ������֤�Ȼ�����Һ������̪���������װ����
���� �¡�
�¡�
�á� �ġ�
�ġ�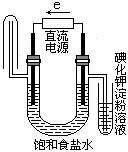
CuS��Cu2S�����������ᣬ���Ǹ������յIJ�����ͬ�����¼���CuS��Cu2S���ֺ�ɫ��ĩ�ķ�����������
������������Ʒ�ֱ��������ᣬ����������������
�¡���������Ʒ�ֱ��������ᣬ������Һ����ɫ
�á�ȡ����ͬ��������Ʒ�ֱ��ڸ��������գ�����������������
�ġ�ȡ����ͬ��������Ʒ�ֱ��ڸ��������գ���������������ɫ
�ö��Ե缫���һ��Ũ�ȵ�����ͭ��Һ��ͨ��һ��ʱ�����������Һ�м���0.1 molCu(OH)2��ǡ�ûָ������ǰ��Ũ�Ⱥ�pH�����������ת�Ƶĵ�����Ϊ�� ��
A��0.1 mol B��0.2 mol C��0.3 mol D��0.4 mol
���и����������Һ�������Լ����Լ������в���������
����������̼����������һ����̼��Ʒ����Һ���¡��ƾ������ᡢ�����ƣ�ʯ����Һ
�á��Ȼ��ơ���������̼���ƣ�ϡ���ᣩ �ġ����ᡢ���ᱵ���Ȼ��أ�̼������Һ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com