

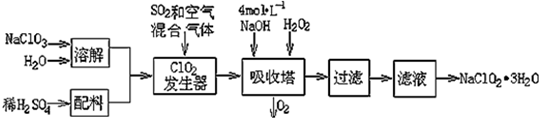
���� �ϳ�1-������ʵ���漰��ѧ����ʽ����д����Ӧ������ѡ�����ʵ����ʡ�ʵ��Ļ���������֪ʶ��
��1���Ʊ�����ѡ��п����ϡ����Ʊ���ϩѡ��2-������Ũ���
��2�������װ���У�a�����ñ��ַ�Һ©������ƿ�ڵ���ѹ��ȣ��Ա�֤��Һ©���ڵ�Һ����˳��������ƿ�У�b��Ҫ����ȫƿ�����ã��Է�ֹ������cΪ��ȥCO�е��������壬ѡ��NaOH��Һ��dΪ��ȥCO�е�H2O���Լ�ѡ��Ũ����������װ���Ʊ�H2������Ҫ�ƾ��ƣ�
��3�������ϩ������SO2��CO2��ˮ������ɵĻ��������ɷ�ʱ��Ӧ����ѡ����ˮCuSO4����ˮ������Ȼ���â�Ʒ����Һ����SO2�����âٱ���Na2SO3��Һ��ȥSO2��Ȼ���â�ʯ��ˮ����CO2���â�����KMnO4��Һ�����ϩ��
��4������ϳ�����ȩ�ķ�ӦΪ���������С�ķ��ȷ�Ӧ��Ϊ����Ӧ���ʺ����ԭ������ת���ʣ�
��5������NaHSO3��Һ�γɳ�����Ȼ��ͨ�����˼��ɳ�ȥ��1-���������ѵķе����ܴ���˿���������������뿪��
��� �⣺��1���������û��ý���п���������������ͨ���û���Ӧ�Ʊ������������������Ũ������п��Ӧ���ܲ�������������ʽΪZn+2HCl=ZnCl2+H2����2-����ͨ����ȥ��Ӧ�������ϩ������ʽΪ����CH3��2CHOH $\stackrel{����}{��}$CH2=CHCH3��+H2O��
�ʴ�Ϊ��Zn+2HCl=ZnCl2+H2������CH3��2CHOH $\stackrel{����}{��}$CH2=CHCH3��+H2O��
��2��������Ũ�����������ͨ��������ˮ������CO�����ڼ����ӷ���������CO�б�Ȼ����м��ᣬ�������ռ�֮ǰ��Ҫ��ȥ���ᣬ��������NaOH��Һ���ռ��ᣮ����Ϊ����������ˮ�����Ա����ֹҺ�嵹������b�������Ƿ�ֹ���������ͨ��Ũ�������CO��Ϊ��ʹ������������˳���Ĵӷ���װ�����ų����ͱ��豣��ѹǿһ�£����a�������DZ��ֺ�ѹ����������װ���Ʊ��������Ͳ�����Ҫ���ȣ����Դ�ʱ����װ���еIJ������������Ƿ�Һ©����������ƿ�������ܶ�С�ڿ����ģ����Ҫ�ռ��������������ֻ���������ſ�����������������ˮ���ռ���
�ʴ�Ϊ����ѹ���������� NaOH��Һ��ŨH2SO4��
��3�������ϩ����������KMnO4��Һ������SO2����������KMnO4��Һ��ɫ��Ʒ����Һ��ʯ��ˮ������CO2����ʯ��ˮ������ˮ����������ˮCuSO4�������ڼ���������������迼���Լ���ѡ���˳��ֻҪͨ����Һ���ͻ����ˮ����������ȼ���ˮ������Ȼ�����SO2���ڼ���֮���ȥSO2����SO2�����ñ���Na2SO3��Һ��������CO2�ͱ�ϩ�����˳��Ϊ�ܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ���
�ʴ�Ϊ���ܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ���
��4�����ڷ�Ӧ��һ�������С�Ŀ��淴Ӧ�����Բ��ø�ѹ������������Ӧ���ʺ����ԭ������ת���ʣ�����Ӧ�Ƿ��ȷ�Ӧ����Ȼ�������������ԭ������ת���ʣ�������������Ӧ���ʣ����Ҫ�����ʵ����¶ȣ������������ԭ������ת���ʣ�������������Ӧ���ʣ����̵���ƽ������Ҫ��ʱ�䣬����ȷ��ѡ����b��
�ʴ�Ϊ��b��
��5����Ʒ�к�������ȩ��������������Ϣ���ñ���NaHSO3��Һ�γɳ�����Ȼ��ͨ�����˼��ɳ�ȥ�����ڱ���NaHSO3��Һ�ǹ����ģ����Լ������ѵ�Ŀ������ȡ��Һ�е�1-��������Ϊ1-���������ѵķе����ܴ���˿���������������뿪��
�ʴ�Ϊ������NaHSO3��Һ�����ˣ���ȡ������
���� ���⿼���л���ϳɷ�������ƣ���Ŀ�ѶȽϴ��ۺ��Խ�ǿ������ʱע��������ʵķ��롢�ᴿ�������������ʵ����ʵ���ͬ�ǽ�����Ĺؼ���


 �Űٷֿ�ʱ����ϵ�д�
�Űٷֿ�ʱ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ���� | HClO2 | HF | HCN | H2S |
| Ka | 1��10-2 | 6.3��10-4 | 4.9��10-10 | K1=9.1��10-8 K2=1.1��10-12 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
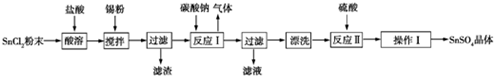
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����

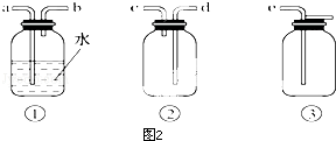
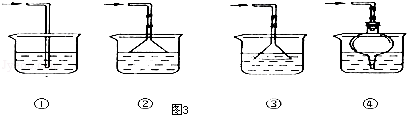
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 25�棬1.01��105Pa��64 g SO2�к��е�ԭ����Ϊ3NA | |
| B�� | ���³�ѹ�£�1.06 g Na2CO3���е�Na+����Ϊ0.02NA | |
| C�� | ���³�ѹ�£�32 g O2��O3�Ļ����������ԭ����Ϊ2NA | |
| D�� | ����NA����ԭ�ӵ������ڱ�״���µ����Ϊ22.4 L |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| ѡ�� | �ı������ | ���� |
| A | ���� | CO32-��ˮ��ƽ�������ƶ� |
| B | ����AlCl3���� | ������������ |
| C | ����100mLH2O | ��Һ��c��H+����c��OH-������С |
| D | ��������CH3COONa���� | ��Һ��n��CO32-������ |
| A�� | A | B�� | B | C�� | C | D�� | D |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
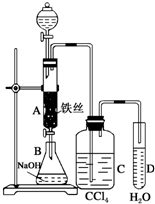 ij��ѧ����С������ͼ��ʾ��װ����ȡ�屽�������Һ©���м��뱽��Һ�壬�ٽ����Һ�������뷴Ӧ��A��A�¶˻����رգ��У�
ij��ѧ����С������ͼ��ʾ��װ����ȡ�屽�������Һ©���м��뱽��Һ�壬�ٽ����Һ�������뷴Ӧ��A��A�¶˻����رգ��У� ��
���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com