
I������ռ������Ҫ�Ļ���ԭ�ϡ�
��1��������ͼ��ʾװ�ÿɼ��֤��������̼���ռ���Һ�����˷�Ӧ����A��B���ӣ���ֹˮ�У�����ͷ�ι��е�Һ�強����ƿ����ʱ��ʵ��������___________________��
�������������䣬��A��C���ӣ��ɹ۲쵽��������__________________________��
��2����NaOH��Һ��ͨ��һ����CO2���ᾧ��õ���ɫ���壬�ð�ɫ�������ɿ����ǣ�
A��NaOH��Na2CO3��B����������������C������������������D����������������
��3�����ʵ��ȷ����2���а�ɫ�����д���A���е������ӣ�
| ʵ����� | ʵ������ | ���� |
| ��ȡ������ɫ�������Թ��У�������ˮ�ܽ⣬�ټ�����BaCl2��Һ | | |
| �� | | |

I����1��ˮ�ص����ɹ��ƿ������ƿ ���ƿ�еij����ܿ������ݲ���
��2��Na2CO3 Na2CO3��NaHCO3 NaHCO3
��3��ʵ����� ʵ������ ���� ��1�� ������ɫ���� ��CO32�� ��2�����ˣ�ȡ������Һ���Թ��У��μӷ�̪ ��Һ��� ��OH��
II����1�������ɵ�CO2ȫ������C�У�ʹ֮��ȫ��Ba(OH)2��Һ����
��2��Ba2++2OH-+CO2=BaCO3��+H2O ��3��C D ��4��25%
��5��B��ˮ�������Ȼ�������Ƚ���Cװ���� ��6����ֹ�����е�CO2����C
�������������I����1��CO2+2NaOH=Na2CO3+H2O���ڶ��߷�����Ӧ����Aװ�õ�����ѹǿ��С��Bװ���е�ˮ�ڴ���ѹ�������¾������ܽ���Aװ�á��������������䣬��A��C���ӣ��ɹ۲쵽�������ǹ��ƿ�еij����ܿ�ð���ݡ���2����NaOH��Һ��ͨ��һ����CO2�����ܷ����ķ�ӦΪ��2NaOH+CO2= Na2CO3+H2O��NaOH+CO2= NaHCO3�����Խᾧ��õ���ɫ���壬�ð�ɫ�������ɿ����ǣ�A��NaOH��Na2CO3��B��Na2CO3��C��Na2CO3��NaHCO3��D��NaHCO3����3����ȡ������ɫ�������Թ��У�������ˮ�ܽ⣬�ټ�����BaCl2��Һ��������ɫ����֤������CO32�����ӡ��ڹ��ˣ�ȡ������Һ���Թ��У��μӷ�̪����Һ���֤������OH����
II����1��ʵ����������������ͨ������������ó��˿ɽ���B��C�еķ�Ӧ���⣬���а����ɵ�CO2ȫ������C�У�ʹ֮��ȫ��Ba(OH)2��Һ���ա���2��C�з�Ӧ����BaCO3�����ӷ���ʽ��Ba2++2OH-+ CO2= BaCO3��+H2O����3��A���ڼ�������֮ǰ��Ӧ�ž�װ���ڵ�CO2������Լ��ٿ����еĶ�����̼�Բ���������Ӱ�죬��Сʵ�������ʵ���ȷ�ȡ�����B���μ�����˹�������Ƿ�Ӧ�����Ķ�����̼���屻����������Һ������գ���߷�Ӧ��ȷ�ȡ�����C����A��B֮������ʢ��Ũ�����ϴ��װ�ã�����ˮ�ֶԶ�����̼�����IJⶨ��Ȼ��Ӱ�졣��ȷ��D������B��C֮������ʢ�б���̼��������Һ��ϴ��װ�ã����ɷ�Ӧ��ӷ�������HCl���������䷴Ӧ����������̼���壬ʹ�ⶨ���ƫ����ȷ����ѡ��Ϊ��C D����4���ɷ���ʽCaCO3+2HCl=CaCl2+ CO2��+H2O; CO2+ Ba(OH)2= BaCO3��+H2O���ɵù�ϵʽCaCO3-- BaCO3����100g CaCO3��197g BaCO3.��BaCO3����Ϊ3.94 g�����Ժ��е�CaCO3������Ϊ2.0g.��Ʒ��̼��Ƶ���������Ϊ(2.0g��8.0g)��100%=25%.��5��B��ˮ�������Ȼ�������Ƚ���Cװ����.��6��װ����U�ι�D�еļ�ʯ�ҵ������Ƿ�ֹ�����е�CO2����C ,�Է�ֹӰ��ʵ��ⶨ�����ȷ�ԡ�
���㣺���������̼�Ļ�ѧ���ʼ���ʵ����������ʳɷֵIJⶨ�����ʵij�ȥ�������ܲ���ʵ�����ķ�����֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ѧ��ȤС���������ʵ�鷽�����ⶨij�Ѳ��ֱ��ʵ�С�մ���Ʒ��Na2CO3������������
����һ����ȡһ��������Ʒ�����������м��������غ���ȴ������ʣ��������������㡣
(1)�����з�����Ӧ�Ļ�ѧ����ʽΪ��________��
(2)ʵ���У�����������ص�Ŀ����________��
����������ȡһ��������Ʒ������С�ձ��У�������ˮ�ܽ⣻��С�ձ��м�������Ba(OH)2��Һ�����ˣ�ϴ�ӡ���������������������������㡣(��֪��Ba2����OH����HCO3��=BaCO3����H2O)
(1)���˲����У������ձ���©���⣬��Ҫ�õ��IJ�������Ϊ________��
(2)ʵ�����жϳ����Ƿ���ȫ�ķ�����________��
��������������ͼ��ʾװ�ý���ʵ�飺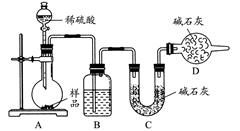
(1)Dװ�õ�������________����Һ©����________(��ܡ����ܡ�)���������ϡ�������ʵ�顣
(2)ʵ��ǰ��ȡ17.90 g��Ʒ��ʵ�����Cװ������8.80 g������Ʒ��Na2CO3����������Ϊ________��
(3)���ݴ�ʵ���õ����ݣ��ⶨ���������Ϊʵ��װ�û�����һ������ȱ�ݣ���ȱ����________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��֪Fe2O3��H2��Ӧ�����¶Ȳ�ͬ��������Fe3O4���ɡ�ij��ѧ��ȤС������H2��ԭFe2O3��ʵ���У��ô����������ɵĺ�ɫ��ĩX��Ϊ̽��X����ɣ����ǽ���������ʵ�飺
(1)��ͬѧ��Ϊ��ɫ��ĩX�ܱ��������������X����������ͬѧ��ͬ�����Ľ��ۣ�ԭ����___________________________��
(2)��ͬѧ�Ƚ�������ɫ��ĩX����װ����������ͭ��Һ���ձ��У������岿���ܽ⣬�м�������ɫ�������������ˣ�Ȼ���������м������ᣬ�ٵμӼ���KSCN��Һ����Һ���ֺ�ɫ��ͨ�������������ͬѧ�ó�X�������Fe��Fe3O4��
�ٵμ�KSCN��Һ��Ŀ����_____________________��
��������ĩXֱ�Ӽ��������У��ټ�KSCN��Һ����Һ�����ֺ�ɫ�����ֺ�ɫ��ԭ����(�����ӷ���ʽ��ʾ) _____________��
(3)��ͬѧ��ʵ�鷽����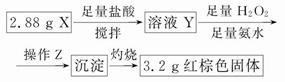
�ٲ���Z��__________________��
��ͨ���������ݣ��ó�2.88 g��ɫ��ĩX�и��ɷֵ����ʵ���Ϊ____________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����NaHCO3��Na2CO3��xH2O�Ļ���Ϊ�˲ⶨxֵ��ijͬѧ������ͼ��ʾ��װ�ý���ʵ��(CaCl2����ʯ�Ҿ�����)��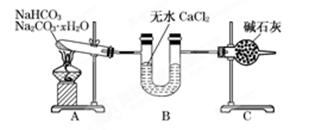
(1)Aװ�õ�����:________________________________________________________________��
(2)Bװ�õ�������:_______________________________________________________________��
(3)Cװ�õ�������:_________________________________________________________________��
(4)����װ��A���Թ���װ��NaHCO3��Na2CO3��xH2O�Ļ����3.7 g���þƾ��Ƽ��ȵ���Ӧ��ȫ����ʱB������1.89g��C������0.22 g����x��ֵΪ______��
(5)��װ�û����Ǻ����ƣ�����ʹ�ⶨ���ƫС��Ӧ��θĽ�________��Ϊʲô��___________________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�̷���FeSO4��7H2O��������ȱ����ƶѪҩƷ����Ҫ�ɷ֡���������������м�����������������������ʣ�Ϊԭ�����������̷���һ�ַ�����
��֪�������±���H2S��Һ��pHԼΪ3.9��SnS������ȫʱ��Һ��pHΪ1.6��FeS��ʼ����ʱ��Һ��pHΪ3.0��������ȫʱ��pHΪ5.5��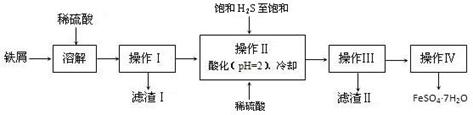
��1�������Ƶõ��̷��������Ƿ���Fe3+��ʵ������� ��
��2������II�У�ͨ�����������͵�Ŀ���� ������Һ���������ữ��pH=2��Ŀ���� ��
��3������IV��˳������Ϊ�� ����ȴ�ᾧ�� ��
��4���ⶨ�̷���Ʒ��Fe2+ �����ķ����ǣ�
a����ȡ2.8500g�̷���Ʒ���ܽ⣬��250mL����ƿ�ж��ݣ�
b����ȡ25.00mL������Һ����ƿ�У�
c���������ữ��0.01000mol?L-1KMnO4��Һ�ζ����յ㣬����KMnO4��Һ�����ƽ��ֵΪ20.00mL��
����֪KMnO4������Һ��Fe2+ ��Ӧʱ����ԭΪMn2+����д���÷�Ӧ�����ӷ���ʽ�� ��
�ڼ���������Ʒ��FeSO4?7H2O����������Ϊ ����С����ʾ��������λС������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�о�С��������ͼװ��̽��SO2��Fe(NO3)3��Һ�ķ�Ӧ����֪��1��0 mol��L��1��Fe(NO3)3��Һ��pH��1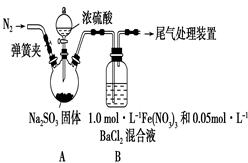
��ش�
��1��װ��A�з�Ӧ�Ļ�ѧ����ʽ��______________________________��
��2��Ϊ�ų�������ʵ��ĸ��ţ��μ�Ũ����֮ǰӦ���еIJ�����_______________________________��
��3��װ��B�в����˰�ɫ��������ɷ���________��
��4������B�в�����ɫ������ԭ��
����1��SO2��Fe3��������������NO3������Ӧ��
����2��SO2��Fe3����Ӧ��
����3��������������SO2��NO3����Ӧ��
�ٰ�����2��װ��B�з�Ӧ�����ӷ���ʽ��_________��֤���ò�����ȷӦ��һ��ȷ��������________��
�ڰ�����3��ֻ�轫װ��B�е�Fe(NO3)3��Һ�滻Ϊ�������������Һ������ͬ�����½���ʵ�顣Ӧѡ����Լ���________(����ĸ)��
a��0��1 mol��L��1ϡ����
b��1��5 mol��L��1Fe(NO3)2��Һ
c��6��0 mol��L��1 NaNO3��0��2 mol��L��1����������ϵ���Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
FeCl3���ִ���ҵ������Ӧ�ù㷺��ij��ѧ�о���ѧϰС��ģ�ҵ�����Ʊ���ˮFeCl3�����ø���ƷFeCl3��Һ�����ж���H2S��
I�����������ϵ�֪����ˮFeCl3�ڿ������׳��⣬����������������������Ʊ���ˮ FeCl3��ʵ�鷽����װ��ʾ��ͼ�����ȼ��г�װ����ȥ���������������£�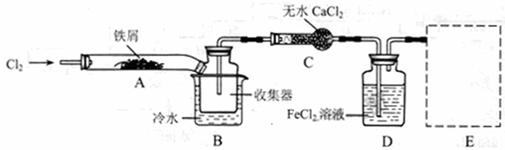
�ټ���װ�õ������ԣ�
��ͨ������Cl2���Ͼ�װ���еĿ�����
���þƾ�������м�·���������Ӧ��ɣ�
�ܡ���
����ϵ��ȴ��ֹͣͨ��Cl2�����ø����H2�Ͼ�Cl2�����ռ����ܷ⡣
��ش��������⣺
��1��װ��A�з�Ӧ�Ļ�ѧ����ʽΪ ��
��2���ڢ۲����Ⱥ����ɵ���״FeCl3�ֽ����ռ��������������ڷ�Ӧ��A�Ҷˡ�Ҫʹ������FeCl3�����ռ������ڢܲ������� ��
��3�����������У�Ϊ��ֹFeCl3��������ȡ�Ĵ�ʩ�У������ţ� ��
��4�����û��װ��C����ƣ��ᵼ�� ��
��5�������ӷ���ʽ��ʾ���߿�E��������װ�ú��Լ������ã� ��
��6����װ��D�еĸ���ƷFeCl3��Һ����H2S���õ�������
��д����Ӧ�����ӷ���ʽ�� ��
��Ӧ�������ռ��������ù�����ȫ����ϡ���ᣬС��ͬѧ��������Һ���������ӵijɷ������ֹ۵㣺��ֻ��Fe3+����ֻ��Fe2+���� ��
Ϊ̽����Һ����ɣ�ʵ�����£�
| ʵ�鲽�� | ʵ������ | ʵ����ۼ���Ӧ���ӷ���ʽ |
| ��ȡ����������Һ���Թ��У���������KSCN��Һ�� | | ˵��������ڲ�����������ٻ�۳����������ӷ���ʽ�� �� |
| ����ȡ����������Һ���Թ��У������������� KMnO4��Һ�� | ��Һ�Ϻ�ɫ��ȥ | ˵���� �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ڿ�����һ�㶼�ױ���������Ħ����[(NH4)2SO4��FeSO4��6H2O]��һ���������Ҫ�ȶ���������ʱ�ֽ��ױ���������ʵ���ҵ��Ʊ�ԭ��Ϊ:FeSO4+(NH4)2SO4+6H2O=(NH4)2SO4��FeSO4��6H2O��
��ͼΪ��ȡĦ���εļ�Ҫ���̣�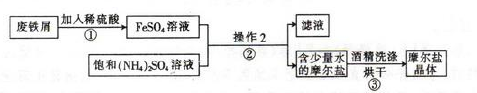
��ش���������:
��1��������з�Ӧ����Ӧ��������ҡ�λ���裬Ŀ���� ��
��2��������еIJ����Ǽ��������� �� ��Ϊʲô���ܼ�������? ��
��3��Ħ������NH4+��Fe2+��SO42-�ļ���:
�ټ�ͬѧ���������ʪ��� ��ֽ��ϡ����� ��Һ���������������ӡ�
����ͬѧ����������е�Fe2+��������KSCN��Һ�� ��Ԥ�ڵ�����ͽ����� ��
��ʵ�ʲ����У���ͬѧ����KSCN��Һʱ��������Һ���dz��ɫ�����������Լ��ķ�������˻��ɡ���ͬѧ������ϸ˼����Ϊ����ͬѧ�ķ����ǿ��еģ�������Ϊ ��
��4����ͬѧ���ⶨĦ������Fe2+�ĺ���������ȡ��4. 0gĦ������Ʒ������ˮ������������ϡ���ᣬ��0.20mol/L��KMnO4��Һ�ζ�������KMnO4��Һ10.00mL
�ٱ�ʵ���ָʾ���� (����ĸ)��
| A����̪ | B��ʯ�� | C������ | D������Ҫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʵ����ģ�ҵ����̼���ƣ�һ���¶��£���һ��������NaCl��Һ��ͨ�백���ﵽ���ͺ��ٲ���ͨ��CO2��һ��ʱ����ֳ��������˵õ�NaHCO3���塣
��1���ù��̵Ļ�ѧ����ʽ�� ��
��2������NaHCO3�õ�������Ʒ��ʵ���ҿɽ��д˲�����װ���� ��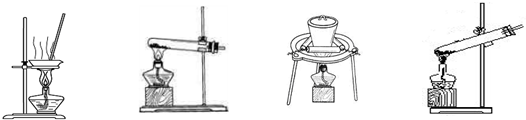
A B C D
��3���õ��Ĵ�����Ʒ��������NaHCO3��NaCl�����ʵ����֤��Ʒ�и�����֡��Լ���������ѡ�ã��Թܡ���ͷ�ιܡ�����װ�á�Ba(NO3)2��Һ��NaOH��Һ��AgNO3��Һ������ʯ��ˮ��ϡ���ᡢϡ���ᡢϡ���ᡣ
| ����һ��ȡ������Ʒ���Թ��У�����������ˮ�����ܽ⡣���Թ��м��� ���۲� | ������Һ�в���������ɫ������ ���ۣ� �� |
| �������������һ����Һ���ˣ�ȡ��Һ���Թ���B�У����� �� ���۲� | ���� �� ���ۣ���Ʒ�к���NaHCO3�� |
| �������������������Һ���ˣ�ȡ��Һ���Թ�C�У� ���۲� | ���� �� ���ۣ� �� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com