

| n |
| v |
| n |
| v |


 ½šŌæ³×ŹŌ¾ķĻµĮŠ“š°ø
½šŌæ³×ŹŌ¾ķĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŹµŃéŹŅÓūÅäÖĘ90mLÅضČĪŖ1.00mol”¤L-1µÄNa2CO3ČÜŅŗ£ŗ
£Ø1£©ÅäÖĘŹ±µÄŹµŃé²½ÖčĪŖ£ŗ¼ĘĖć”¢³ĘĮ攢Čܽā”¢×ŖŅĘ£ØĻ“µÓ£©”¢ ”ų ”¢Ņ”ŌČ£»
£Ø2£©±¾ŹµŃéÖŠ±ŲŠėÓƵ½µÄŅĒĘ÷ÓŠĶŠÅĢĢģĘ½”¢Ņ©³×”¢²£Į§°ō”¢ÉÕ±”¢ĮæĶ²”¢½ŗĶ·µĪ¹Ü”¢
”ų (×¢Ć÷ĖłŠčŅĒĘ÷µÄ¹ęøń)”£
£Ø3£©Ä³Ķ¬Ń§Óū³ĘĮæNa2CO3µÄÖŹĮ棬ĖūĻČÓĆĶŠÅĢĢģĘ½³ĘĮæÉÕ±µÄÖŹĮ棬ĢģĘ½Ę½ŗāŗóµÄדĢ¬ČēĶ¼”£ÉÕ±µÄŹµ¼ŹÖŹĮæĪŖ ”ų g£¬ŅŖĶź³É±¾ŹµŃéøĆĶ¬Ń§Ó¦³Ę³ö ”ų g Na2CO3”£

£Ø4£©Ź¹ÓĆČŻĮæĘæĒ°±ŲŠė½ųŠŠµÄŅ»²½²Ł×÷ŹĒ ”ų ”£
£Ø5£©Čō³öĻÖČēĻĀĒéæö£¬¶ŌĖłÅäČÜŅŗÅØ¶Č½«ÓŠŗĪÓ°Ļģ£ØĢī”°Ę«øß”±”¢”°Ę«µĶ”±»ņ”°ĪŽÓ°Ļģ”±£©
¢ŁĆ»ÓŠĻ“µÓÉÕ±ŗĶ²£Į§°ō ”ų £»¢ŚČŻĮæĘæ²»øÉŌļ£¬ŗ¬ÓŠÉŁĮæÕōĮóĖ® ”ų £»
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010-2011ѧğÕć½Ź”Äž²ØŹŠøßŅ»ÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗŹµŃéĢā
ŹµŃéŹŅÓūÅäÖĘ90mLÅضČĪŖ1.00mol”¤L-1µÄNa2CO3ČÜŅŗ£ŗ
£Ø1£©ÅäÖĘŹ±µÄŹµŃé²½ÖčĪŖ£ŗ¼ĘĖć”¢³ĘĮ攢Čܽā”¢×ŖŅĘ£ØĻ“µÓ£©”¢ ”ų ”¢Ņ”ŌČ£»
£Ø2£©±¾ŹµŃéÖŠ±ŲŠėÓƵ½µÄŅĒĘ÷ÓŠĶŠÅĢĢģĘ½”¢Ņ©³×”¢²£Į§°ō”¢ÉÕ±”¢ĮæĶ²”¢½ŗĶ·µĪ¹Ü”¢
”ų (×¢Ć÷ĖłŠčŅĒĘ÷µÄ¹ęøń)”£
£Ø3£©Ä³Ķ¬Ń§Óū³ĘĮæNa2CO3µÄÖŹĮ棬ĖūĻČÓĆĶŠÅĢĢģĘ½³ĘĮæÉÕ±µÄÖŹĮ棬ĢģĘ½Ę½ŗāŗóµÄדĢ¬ČēĶ¼”£ÉÕ±µÄŹµ¼ŹÖŹĮæĪŖ ”ų g£¬ŅŖĶź³É±¾ŹµŃéøĆĶ¬Ń§Ó¦³Ę³ö ”ų g Na2CO3”£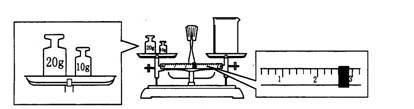
£Ø4£©Ź¹ÓĆČŻĮæĘæĒ°±ŲŠė½ųŠŠµÄŅ»²½²Ł×÷ŹĒ ”ų ”£
£Ø5£©Čō³öĻÖČēĻĀĒéæö£¬¶ŌĖłÅäČÜŅŗÅØ¶Č½«ÓŠŗĪÓ°Ļģ£ØĢī”°Ę«øß”±”¢”°Ę«µĶ”±»ņ”°ĪŽÓ°Ļģ”±£©
¢ŁĆ»ÓŠĻ“µÓÉÕ±ŗĶ²£Į§°ō ”ų £»¢ŚČŻĮæĘæ²»øÉŌļ£¬ŗ¬ÓŠÉŁĮæÕōĮóĖ® ”ų £»
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģÕć½Ź”Äž²ØŹŠøßŅ»ÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗŹµŃéĢā
ŹµŃéŹŅÓūÅäÖĘ90mLÅضČĪŖ1.00mol”¤L-1µÄNa2CO3ČÜŅŗ£ŗ
£Ø1£©ÅäÖĘŹ±µÄŹµŃé²½ÖčĪŖ£ŗ¼ĘĖć”¢³ĘĮ攢Čܽā”¢×ŖŅĘ£ØĻ“µÓ£©”¢ ”ų ”¢Ņ”ŌČ£»
£Ø2£©±¾ŹµŃéÖŠ±ŲŠėÓƵ½µÄŅĒĘ÷ÓŠĶŠÅĢĢģĘ½”¢Ņ©³×”¢²£Į§°ō”¢ÉÕ±”¢ĮæĶ²”¢½ŗĶ·µĪ¹Ü”¢
”ų (×¢Ć÷ĖłŠčŅĒĘ÷µÄ¹ęøń)”£
£Ø3£©Ä³Ķ¬Ń§Óū³ĘĮæNa2CO3µÄÖŹĮ棬ĖūĻČÓĆĶŠÅĢĢģĘ½³ĘĮæÉÕ±µÄÖŹĮ棬ĢģĘ½Ę½ŗāŗóµÄדĢ¬ČēĶ¼”£ÉÕ±µÄŹµ¼ŹÖŹĮæĪŖ ”ų g£¬ŅŖĶź³É±¾ŹµŃéøĆĶ¬Ń§Ó¦³Ę³ö ”ų g Na2CO3”£

£Ø4£©Ź¹ÓĆČŻĮæĘæĒ°±ŲŠė½ųŠŠµÄŅ»²½²Ł×÷ŹĒ ”ų ”£
£Ø5£©Čō³öĻÖČēĻĀĒéæö£¬¶ŌĖłÅäČÜŅŗÅØ¶Č½«ÓŠŗĪÓ°Ļģ£ØĢī”°Ę«øß”±”¢”°Ę«µĶ”±»ņ”°ĪŽÓ°Ļģ”±£©
¢ŁĆ»ÓŠĻ“µÓÉÕ±ŗĶ²£Į§°ō ”ų £»¢ŚČŻĮæĘæ²»øÉŌļ£¬ŗ¬ÓŠÉŁĮæÕōĮóĖ® ”ų £»
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com