
| A��ͭ�� | B����ˮ | C������ͭ | D��������ͭ |
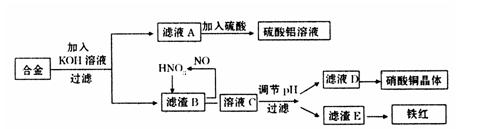
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
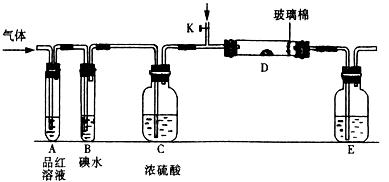
 ��SO2��Cl2��װ��A�й۲쵽�������Ƿ���ͬ�� (���ͬ������ͬ��)����װ��D��װ��
��SO2��Cl2��װ��A�й۲쵽�������Ƿ���ͬ�� (���ͬ������ͬ��)����װ��D��װ�� ��V2O5(����)��ͨ��SO2ʱ����Kͨ������O2�Ļ�ѧ��Ӧ����ʽΪ ��
��V2O5(����)��ͨ��SO2ʱ����Kͨ������O2�Ļ�ѧ��Ӧ����ʽΪ �� �� C���������
�� C��������� Һ D��������Һ
Һ D��������Һ l2�뺬X����Һ��Ӧ�����ӷ���ʽ ��
l2�뺬X����Һ��Ӧ�����ӷ���ʽ ��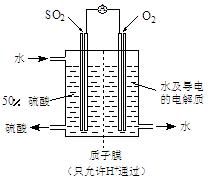

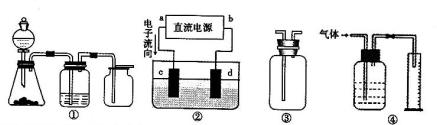
 ��aΪ���⣬dΪ����
��aΪ���⣬dΪ���� 2��NH3��Cl2,��HCl��NO2��
2��NH3��Cl2,��HCl��NO2�� ��װ�â������ڲ����������
��װ�â������ڲ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 Fe2O3+SO2��+SO3��+14H2O������ͼ��װ�ÿ���������������Ӧ�����е���������ش��������⣺
Fe2O3+SO2��+SO3��+14H2O������ͼ��װ�ÿ���������������Ӧ�����е���������ش��������⣺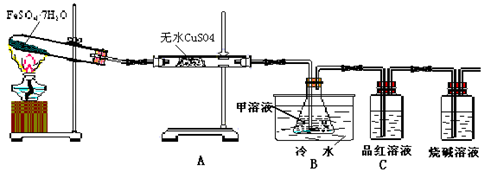
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
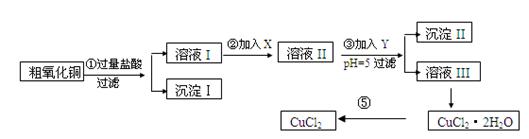
| ���� | ��ʼ����ʱpH | ��ȫ����ʱpH |
| Fe(OH)3 | 2��7 | 3��7 |
| Fe(OH)2 | 7��6 | 9��6 |
| Cu(OH)2 | 5��2 | 6��4 |
| ������ | ����pH������ | ||
| A | ˫��ˮ | D | ��ˮ |
| B | ������� | E | ��ʽ̼��ͭ |
| C | ��ˮ | F | ����ͭ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| �ζ� ���� | ������Һ����� /mL | ����Һ�����/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 0.20 | 20.20 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
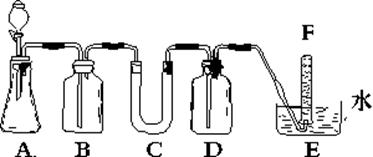
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
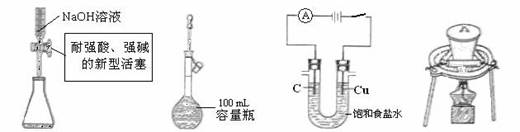
| A����ȡ15.00 mL | B�����ݡ������������� |
| C�������Cl2��H2 | D����������ʯ��ʯ. |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��B�е������в������� | B��B�е������� �γ�һ��ˮ�� �γ�һ��ˮ�� |
| C������Ƭ���ڱ�� | D��п����ʴ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������ƽ���������зֱ�ŵ�������ֽ����ȡ2.0g NaOH���� |
| B����NaOH�������ձ����ܽ��Ѹ��С��ת����250mL����ƿ�� |
| C������ʱ��С�ļ�ˮ�����˿̶��ߣ���ʱѸ���ý�ͷ�ι�����һЩ |
| D����Һ������ϣ���������ת�������������Լ�ƿ�����ϱ�ǩ��ϴ������ƿ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com