
ʹ���Ȼ�����ԭ�Ͽ��Եõ����ֲ�Ʒ��
(1)ʵ�����ö��Ե缫���100 mL 0.1 mol��L��1 NaCl��Һ���������������õ�112 mL����(��״��)����������Һ�����ʵ���Ũ����______________(���Է�Ӧǰ����Һ����ı仯)��
(2)ijѧ��������һ�ּ��û���������Һ����������ʯī���缫��ⱥ���Ȼ�����Һ��ͨ��ʱ��ΪʹCl2����ȫ���գ��Ƶ��н�ǿɱ������������Һ���������ͼ��װ��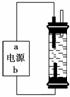 ���Ե�Դ�缫���ƺ�����Һ����Ҫ�ɷ��ж���ȷ����________(����ĸ���)��
���Ե�Դ�缫���ƺ�����Һ����Ҫ�ɷ��ж���ȷ����________(����ĸ���)��
A��a������b������NaClO��NaCl
B��a������b������NaClO��NaCl
C��a������b������HClO��NaCl
D��a������b������HClO��NaCl
(3)ʵ�����к�������������Һ���ռӦ�Ƶð�ɫ������Fe(OH)2������ijͬѧ������ͼװ�ã�ֻ����һ���缫��ͨ����ⷨ��ȡ�ϴ�����Fe(OH)2�������ҽϳ�ʱ�䲻��ɫ����ͬѧ���ϵĵ�������__________(��Ԫ�ط��ű�ʾ)���ܵķ�Ӧʽ��___________��
�𰸡�(1)0.1 mol��L��1��(2)B
(3)Fe��Fe��2H2O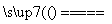 Fe(OH)2����H2��
Fe(OH)2����H2��
������(1)n(H2)��n(Cl2)��0.005 mol���μӷ�Ӧ��n(NaCl)��0.01 mol��˵��NaClǡ�õ����ϣ���Һ��ΪNaOH��Һ��n(NaOH)��n(NaCl)��0.01 mol��c(NaOH)��0.1 mol��L��1��
(2)��ⱥ���Ȼ�����Һ���ܷ�Ӧ��2NaCl��2H2O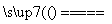 2NaOH��H2����Cl2������ĿҪ��ʹCl2����ȫ���գ��������������²��ĵ缫�ϲ������²��ĵ缫���������Ƴ�bΪ��Դ����������һ��Ϊ����a�����ɵ�Cl2��������������NaOH��Ӧ��Cl2��2NaOH===NaCl��NaClO��H2O��������Һ����Ҫ�ɷ���NaClO��NaCl��
2NaOH��H2����Cl2������ĿҪ��ʹCl2����ȫ���գ��������������²��ĵ缫�ϲ������²��ĵ缫���������Ƴ�bΪ��Դ����������һ��Ϊ����a�����ɵ�Cl2��������������NaOH��Ӧ��Cl2��2NaOH===NaCl��NaClO��H2O��������Һ����Ҫ�ɷ���NaClO��NaCl��
(3)��ĿҪ��ͨ����ⷨ��ȡFe(OH)2���������Һ��NaCl��Һ������ͨ���缫��Ӧ����Fe2����OH�������ݵ缫������Ӧ���ص㣬ѡ������������������������Ӧ����Fe2������Һ�е�������Na����H���������ƶ������ݷŵ�˳������H���ŵ����������H����Դˮ�ĵ��룬��Ӧʽ2H2O��2e��===H2����2OH�����ڵ糡�����£�����������Fe2��������������OH���ڶ����ƶ��Ĺ������������������ֽⷴӦ����Fe(OH)2��������������Ӧʽ��������Ӧʽ���ӵõ��ܵķ�Ӧʽ��Fe��2H2O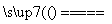 Fe(OH)2����H2����
Fe(OH)2����H2����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�������ʵķ����м��ЦҼ����Цм�����( )
��HCl ��H2O ��N2 ��H2O2 ��C2H4 ��C2H2
A���٢ڢ� B���ۢܢݢ� C���٢ۢ� D���ۢݢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���Ȼ�����(S2Cl2)�ǹ㷺������ҵ����������ӽṹ��ͼ��ʾ�������£�S2Cl2��ˮ��ˮ�⣬��������ʹƷ����ɫ�����塣����˵���������
A. S2Cl2Ϊ���ۻ�����
B. S2Cl2ֻ���м��Թ��ۼ�
C. S2Cl2��ˮ��ӦʱS��S����S��Cl��������
D. S2Cl2������S��S����S��Cl����ͨ�����õ��Ӷ��γɵ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ȼ�շ��Dzⶨ�л������ﻯѧʽ��һ����Ҫ������ȡ0.1molij����ȫȼ�գ�ȼ�ղ�������ͨ������ͼ��ʾ��װ�ã�ʵ������Ƶü�װ������5.4g����װ������26.4g��
��1����5�֣�������ķ���ʽ
��2����3�֣�����������ѧ��Ӧʹ��ˮ������KMnO4��Һ��ɫ��
д������ȴ�����ܵĽṹ��ʽ
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(1)������Ϊ��������H2SO4��Һ�е�⣬���ı����γ�����Ĥ�������缫��ӦʽΪ________________________________________________________________________��
(2)��Al������������ʯī��������NaHCO3��Һ�����Һ���е�⣬����������R��R���ȷֽ����ɻ�����Q��д����������R�ĵ缫��Ӧʽ��________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ͼ��ʾ��װ���У���ֱͨ����5 minʱ��ͭ�缫��������2.16 g���Իش��������⡣
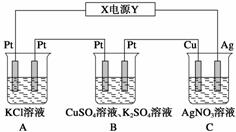
(1)��Դ��X�缫Ϊֱ����Դ��________����
(2)pH�仯��A��________��B��________��C��________(���������С�����䡱)��
(3)ͨ��5 minʱ��B�й��ռ�224 mL(��״����)���壬��Һ���Ϊ200 mL����ͨ��ǰCuSO4��Һ�����ʵ���Ũ��Ϊ________(����ǰ����Һ����ޱ仯)��
(4)��A��KCl��������Һ�����Ҳ��200 mL��������Һ��pHΪ__________(����ǰ����Һ����ޱ仯)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������ͼװ�ý���ʵ�飬��ʼʱ��a��b����Һ����ƽ���ܷ�ã�����һ��ʱ�䡣����˵������ȷ���� (����)
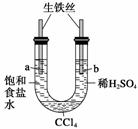
A��a�ܷ���������ʴ��b�ܷ������ⸯʴ
B��һ��ʱ���a��Һ�����b��Һ��
C��a����Һ��pH����b����Һ��pH��С
D��a��b����������ͬ�ĵ缫��Ӧʽ��Fe��2e��===Fe2��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ö��Ե缫���������Һ����������������������������Ϊ1��1���� (����)
A��NaCl��Һ B��CuCl2��Һ
C��CuSO4��Һ D��KOH��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������Ҫ���л�����ԭ�ϣ�������ϩֱ��ˮ�Ϸ�����ˮ�Ϸ��������ش��������⣺
��1�����ˮ�Ϸ���ָ�Ƚ���ϩ��Ũ���ᷴӦ��������������(C2H5OSO3H)����ˮ�������Ҵ���д����Ӧ�ķ�Ӧ�Ļ�ѧ����ʽ
��2����֪��
�״���ˮ��Ӧ��2CH3OH(g)��CH3OCH3(g)��H2O(g)��H1����23.9KJ·mol��1
�״���ϩ����Ӧ��2CH3OH(g)��C2H4 (g)��2H2O(g) ��H2����29.1KJ·mol��1
�Ҵ��칹����Ӧ��CH3CH2OH(g)��CH3OCH3(g)) ��H3��+50.7KJ·mol��1
����ϩ����ֱ��ˮ�Ϸ�ӦC2H4 (g)��H2O(g)��C2H5OH(g)����H�� KJ·mol��1
����ˮ�Ϸ���ȣ�����ֱ��ˮ�Ϸ����ŵ��ǣ� ��
��3����ͼΪ����ֱ��ˮ�Ϸ�����ϩ��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ(����n(H2O)︰n(C2H4)=1︰1)
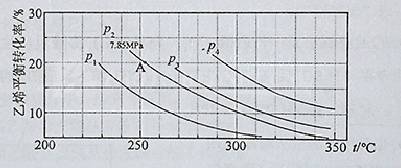
����ʽ������ϩˮ�����Ҵ���Ӧ��ͼ��A���ƽ�ⳣ��K�� (��ƽ���ѹ����ƽ��Ũ�ȼ��㣬��ѹ����ѹ�����ʵ�������)
��ͼ��ѹǿP1��P2��P3��P4�Ĵ�С˳��Ϊ�� �������ǣ�
������ֱ��ˮ�Ϸ������õĹ�������Ϊ������/������Ϊ��������Ӧ�¶�290 �棬ѹǿ6��9MPa��n(H2O)︰n(C2H4)=0��6︰1����ϩ��ת����Ϊ5℅����Ҫ��һ�������ϩ��ת���ʣ����˿����ʵ��ı䷴Ӧ�¶Ⱥ�ѹǿ�⣬�����Բ�ȡ�Ĵ�ʩ�У� �� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com