
��11�֣�.AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ͼת����ϵ��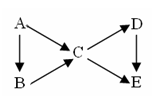
��1������ͼ��B��C��Ϊ�����D��E��Ϊ�Σ���A�����ǣ����ţ�__________��
��Na ��N2 ��C ��S
��2�����������ʵ���ɫ��Ӧ��Ϊ��ɫ������C��D��E��ˮ��Һ���Լ��ԣ��ҵ�Ũ��ʱ����C��D��E��B��������������B�к��еĻ�ѧ������Ϊ__________________��
Aת����C�����ӷ���ʽΪ��________________________________��
Dת����E�����ӷ���ʽΪ_______________________________________��
��3����������B��C��D��Ϊ���壬��B������ʹʪ��ĺ�ɫʯ����ֽ����
���ڹ�ҵ������B����ʱΪ�˼ӿ췴Ӧ����Ӧѡ���������______________����������߷�Ӧ���ת���ʵ�������___________��
��C��D������β���е��к��ɷ֣���NaOH��Һ���տ�������Ⱦ����Ӧ�Ļ�ѧ����ʽΪ__________________________________��
����11�֣���1���٢ۣ�2�֣�
��2�����Ӽ����Ǽ��Թ��ۼ����ۼ�������2�֣�2Na��2H2O��2Na+��2OH-��H2������1�֣�
CO32-��CO2��H2O��2HCO3? ��1�֣�
��3���ٸ��¡���ѹ����������2�֣� ��ѹ��1�֣�
��NO��NO2��2NaOH��2NaNO2��H2O��2�֣�
������1���٢�
��������A��C��2Na��O2 Na2O2,A��B��4Na��O2=2Na2O,B��C��2Na2O��O2
Na2O2,A��B��4Na��O2=2Na2O,B��C��2Na2O��O2 2Na2O2;
2Na2O2;
��A��C,������N2ֱ������NO2;
��A��C��C��O2 CO2��A��B��2C��O2="2CO" B��C��2CO��O2=2CO2;
CO2��A��B��2C��O2="2CO" B��C��2CO��O2=2CO2;
��A��C,������Sֱ������SO3;
��2�����Ӽ����Ǽ��Թ��ۼ����ۼ�������2�֣�2Na��2H2O��2Na+��2OH-��H2������1�֣�
CO32-��CO2��H2O��2HCO3? ��1�֣�
������A��Na��B��ΪNa2O2��CΪNaOH��DΪNa2CO3��E ΪNaHCO3,B��ΪNa2O2��Na�� ��O22�D֮��Ϊ���Ӽ���O22�D��O�DO��Ϊ�Ǽ��Թ��ۼ����ۼ�����Aת����C�����ӷ���ʽΪ����2Na��2H2O��2Na+��2OH-��H2����Dת����E�����ӷ���ʽΪCO32-��CO2��H2O��2HCO3?
��3���ٸ��¡���ѹ����������2�֣� ��ѹ��1�֣�
��NO��NO2��2NaOH��2NaNO2��H2O��2�֣�
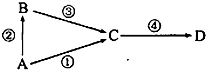
������A�DN2 B�DNH3 C�DNO D�DNO2 E�DHNO3
���ڹ�ҵ������NH3����ʱΪ�˼ӿ췴Ӧ����Ӧѡ��������Ǹ��¡���ѹ����������������߷�Ӧ���ת���ʵ������Ǹ�ѹ;��C�DNO��D�DNO2������β���е��к��ɷ֣���NaOH��Һ���տ�������Ⱦ����Ӧ�Ļ�ѧ����ʽΪNO��NO2��2NaOH��2NaNO2��H2O��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 A��B��C��D��Ϊ��ѧ�������ʣ�����AΪ���ʣ�B��C��DΪ��������Ҵ�������ת����ϵ���Իش��������⣺
A��B��C��D��Ϊ��ѧ�������ʣ�����AΪ���ʣ�B��C��DΪ��������Ҵ�������ת����ϵ���Իش��������⣺
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��14�֣�AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ת����ϵ��
��1������ͼ��B��C��Ϊ�����D��E��Ϊ�Σ���A������__________��
 ��Na ��N2 ��C ��S
��Na ��N2 ��C ��S
��2�����������ʵ���ɫ��Ӧ��Ϊ��ɫ������C��D��E��ˮ��Һ���Լ��ԣ��ҵ�Ũ��ʱ����C��D��E��B��������������B�к��еĻ�ѧ������Ϊ__________________��
Aת����C�����ӷ���ʽΪ�� ________________________________��
Dת����E�����ӷ���ʽΪ_______________________________________��
��3����������B��C��D��Ϊ���壬��B������ʹʪ��ĺ�ɫʯ����ֽ����
���ڹ�ҵ������B����ʱΪ�˼ӿ췴Ӧ����Ӧѡ���������______________����������߷�Ӧ���ת���ʵ�������___________��
��C��D������β���е��к��ɷ֣���NaOH��Һ���տ�������Ⱦ����Ӧ�Ļ�ѧ����ʽΪ__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��11�֣�.AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ͼת����ϵ��
��1������ͼ��B��C��Ϊ�����D��E��Ϊ�Σ���A�����ǣ����ţ�__________��
��Na ��N2 ��C ��S
��2�����������ʵ���ɫ��Ӧ��Ϊ��ɫ������C��D��E��ˮ��Һ���Լ��ԣ��ҵ�Ũ��ʱ����C��D��E��B��������������B�к��еĻ�ѧ������Ϊ__________________��
Aת����C�����ӷ���ʽΪ�� ________________________________��
Dת����E�����ӷ���ʽΪ_______________________________________��
��3����������B��C��D��Ϊ���壬��B������ʹʪ��ĺ�ɫʯ����ֽ����
���ڹ�ҵ������B����ʱΪ�˼ӿ췴Ӧ����Ӧѡ���������______________����������߷�Ӧ���ת���ʵ�������___________��
��C��D������β���е��к��ɷ֣���NaOH��Һ���տ�������Ⱦ����Ӧ�Ļ�ѧ����ʽΪ__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�����ʡ������һ��������黯ѧ�Ծ� ���ͣ�ѡ����
��11�֣�.AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ͼת����ϵ��

��1������ͼ��B��C��Ϊ�����D��E��Ϊ�Σ���A�����ǣ����ţ�__________��
��Na ��N2 ��C ��S
��2�����������ʵ���ɫ��Ӧ��Ϊ��ɫ������C��D��E��ˮ��Һ���Լ��ԣ��ҵ�Ũ��ʱ����C��D��E��B��������������B�к��еĻ�ѧ������Ϊ__________________��
Aת����C�����ӷ���ʽΪ�� ________________________________��
Dת����E�����ӷ���ʽΪ_______________________________________��
��3����������B��C��D��Ϊ���壬��B������ʹʪ��ĺ�ɫʯ����ֽ����
���ڹ�ҵ������B����ʱΪ�˼ӿ췴Ӧ����Ӧѡ���������______________����������߷�Ӧ���ת���ʵ�������___________��
��C��D������β���е��к��ɷ֣���NaOH��Һ���տ�������Ⱦ����Ӧ�Ļ�ѧ����ʽΪ__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ���½�ũ��ʦ����ѧ�����ڶ���ģ����ѧ�Ծ� ���ͣ������
��14�֣�AΪ���ʣ�B��C��D��EΪ��A������ͬԪ�صĻ��������֮��������ת����ϵ��
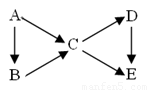
��1������ͼ��B��C��Ϊ�����D��E��Ϊ�Σ���A������__________��
��Na ��N2 ��C ��S
��2�����������ʵ���ɫ��Ӧ��Ϊ��ɫ������C��D��E��ˮ��Һ
���Լ��ԣ��ҵ�Ũ��ʱ����C��D��E��B��������������B�к��еĻ�ѧ������Ϊ__________________��
Aת����C�����ӷ���ʽΪ�� ________________________________��
Dת����E�����ӷ���ʽΪ_______________________________________��
��3����������B��C��D��Ϊ���壬��B������ʹʪ��ĺ�ɫʯ����ֽ����
���ڹ�ҵ������B����ʱΪ�˼ӿ췴Ӧ����Ӧѡ���������______________����������߷�Ӧ���ת���ʵ�������___________��
��C��D������β���е��к��ɷ֣���NaOH��Һ���տ�������Ⱦ����Ӧ�Ļ�ѧ����ʽΪ__________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com