
| |||||||||||||||||||

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������������ ���ͣ�022
������0��5���£������͵�Na2CO3��Һ�У��ȼ���H2O2���ȶ���(MgCl2��Na2SiO3)����ַ�Ӧ���ټ�������������˷��룬���ù�̼���ƾ��壮�������ϲ��ϣ��ش��������⣺
����(1)д��H2O2�ĵ���ʽ��__________________________________________________��
����(2)H2O2����Ϊ��ɫ����������Ҫԭ����_____________________________________��
����(3)�ȶ����ܷ�Ӧ���������ォ��̼�������Ӱ���ס��д���ȶ�����Ӧ����������Ļ�ѧ����ʽ��_________________________________________________________________��
����(4)�����������������__________________________________________________��
����(5)�������ʲ���ʹ��̼����ʧЧ����______________________________________��
����A��MnO2������������������������������������������������������������������������������������������������������������ B��NaHCO3
����C��H2S������������������������������������������������ ���� D��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�콭��������ѧ������ǰ����ѵ����ѧ���� ���ͣ������
�������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ���������ǹ������ⷨ�����������ƵĹ�������ͼ��

��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2?3H2O��
�ڴ�ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10�����°�ȫ��
��160 g/L NaOH��Һ��ָ160 gNaOH��������ˮ������Һ�����Ϊ1L��
��1��160 g/L NaOH��Һ�����ʵ���Ũ��Ϊ ��������Ҫ�������Һ������������
����Ҫ��һ�������������������� ��������������������˵������
��2���������й�����������ÿ�������������ѡ����ţ���
a����SO2������SO3����ǿ���ԣ� b��ϡ��ClO2�Է�ֹ��ը��c����NaClO3������ClO2
��3���������ڵķ�Ӧ�Ļ�ѧ����ʽΪ��������������������������������������
���������¶Ȳ��ܳ���20�棬��Ŀ�������� ��
��4���ڼ�����Һ��NaClO2�Ƚ��ȶ���������������Ӧά��NaOH�Թ������ж�NaOH��
������ļ�ʵ�鷽���� ��
��5����������Ϊ��ֹNaClO2����ԭ��NaCl�����û�ԭ���Ļ�ԭ��Ӧ���С���H2O2�⣬������ѡ��Ļ�ԭ������ ������ѡ����ţ���
a��Na2O2 b��Na2S c��FeCl2
��6������Һ�еõ�NaClO2?3H2O�־����ʵ�����������������������ѡ����ţ���
a������ b������ c������ d������ e����ȴ�ᾧ
Ҫ�õ�������NaClO2?3H2O���������еIJ��������������� ��������������ƣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�����ʡ�����и�����ѧ����ĩ�������ۻ�ѧ�Ծ��������棩 ���ͣ�ʵ����
�������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ���������ǹ������ⷨ�����������ƵĹ�������ͼ��
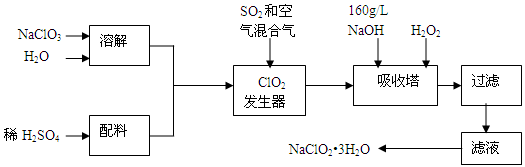
��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2•3H2O��
�ڴ�ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10�����°�ȫ��
��160 g/L NaOH��Һ��ָ160 g NaOH��������ˮ������Һ�����Ϊ1L��
��1��160 g/L NaOH��Һ�����ʵ���Ũ��Ϊ������ ��������Ҫ�������Һ����������������Ҫ��һ�������������������� ��������������������˵������
��2���������й�����������ÿ�������������ѡ����ţ���
a����SO2������SO3����ǿ���ԣ� b��ϡ��ClO2�Է�ֹ��ը��
c����NaClO3������ClO2
��3���������ڷ�Ӧ�����ӷ���ʽΪ��������������������������������������
���������¶Ȳ��ܳ���20�棬��Ŀ�������� ��
��4���ڼ�����Һ��NaClO2�Ƚ��ȶ���������������Ӧά��NaOH�Թ������ж�NaOH�Ƿ�����ļ�ʵ�鷽���� ��
��5����������Ϊ��ֹNaClO2����ԭ��NaCl�����û�ԭ���Ļ�ԭ��Ӧ���С���H2O2�⣬������ѡ��Ļ�ԭ������ ������ѡ����ţ���
a��Na2O2 b��Na2S c��FeCl2
��6������Һ�еõ�NaClO2•3H2O�־����ʵ�����������������������ѡ����ţ���
a������ b������Ũ�� c������ d������ϴ�� e����ȴ�ᾧ
Ҫ�õ�������NaClO2•3H2O���������еIJ����������� ���� ����������ƣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(08�Ϻ�������4��ģ��)�������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ���������ǹ������ⷨ�����������ƵĹ�������ͼ��

��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2•3H2O��
�ڴ�ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10�����°�ȫ��
��160 g/L NaOH��Һ��ָ160 gNaOH��������ˮ������Һ�����Ϊ1L��
��1��160 g/L NaOH��Һ�����ʵ���Ũ��Ϊ������ ��������Ҫ�������Һ������������
����Ҫ��һ�������������������� ��������������������˵������
��2���������й�����������ÿ�������������ѡ����ţ���
a����SO2������SO3����ǿ���ԣ�
b��ϡ��ClO2�Է�ֹ��ը��
c����NaClO3������ClO2
��3���������ڵķ�Ӧ�Ļ�ѧ����ʽΪ��������������������������������������
���������¶Ȳ��ܳ���20�棬��Ŀ�������� ��
��4���ڼ�����Һ��NaClO2�Ƚ��ȶ���������������Ӧά��NaOH�Թ������ж�NaOH��
������ļ�ʵ�鷽���� ��
��5����������Ϊ��ֹNaClO2����ԭ��NaCl�����û�ԭ���Ļ�ԭ��Ӧ���С���H2O2�⣬������ѡ��Ļ�ԭ������ ������ѡ����ţ���
a��Na2O2 b��Na2S c��FeCl2
��6������Һ�еõ�NaClO2•3H2O�־����ʵ�����������������������ѡ����ţ���
a������ b������ c������ d������ e����ȴ�ᾧ
Ҫ�õ�������NaClO2•3H2O���������еIJ��������������� ��������������ƣ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ���������ǹ������ⷨ�����������ƵĹ�������ͼ��


��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2??3H2O��
�ڴ�ClO2�ֽⱬը��һ����ϡ����������ϡ�͵�10�����°�ȫ��
��160 g/L NaOH��Һ��ָ160 gNaOH��������ˮ������Һ�����Ϊ1L��
��1��160 g/L NaOH��Һ�����ʵ���Ũ��Ϊ ��������Ҫ�������Һ������������
����Ҫ��һ�������������������� ��������������������˵������
��2���������й�����������ÿ�������������ѡ����ţ���
a����SO2������SO3����ǿ���ԣ� b��ϡ��ClO2�Է�ֹ��ը��c����NaClO3������ClO2
��3���������ڵķ�Ӧ�Ļ�ѧ����ʽΪ��������������������������������������
���������¶Ȳ��ܳ���20�棬��Ŀ�������� ��
��4���ڼ�����Һ��NaClO2�Ƚ��ȶ���������������Ӧά��NaOH�Թ������ж�NaOH��
������ļ�ʵ�鷽���� ��
��5����������Ϊ��ֹNaClO2����ԭ��NaCl�����û�ԭ���Ļ�ԭ��Ӧ���С���H2O2�⣬������ѡ��Ļ�ԭ������ ������ѡ����ţ���
a��Na2O2 b��Na2S c��FeCl2
��6������Һ�еõ�NaClO2??3H2O�־����ʵ�����������������������ѡ����ţ���
a������ b������ c������ d������ e����ȴ�ᾧ
Ҫ�õ�������NaClO2??3H2O���������еIJ��������������� ��������������ƣ���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com