
����Ŀ��ijѧϰС������ͼ��ʾװ�òⶨ��þ�Ͻ������������������������ԭ��������
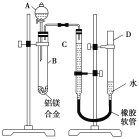
(1)A���Լ�Ϊ_________________��
(2)ʵ��ǰ���Ƚ���þ�Ͻ���ϡ���н���Ƭ�̣���Ŀ����__________________________��
(3)��������ԣ���ҩƷ��ˮװ��������У����Ӻ�װ�ú�����еIJ������У��ټ�¼C��Һ��λ�ã��ڽ�B��ʣ�������ˣ�ϴ�ӣ�������أ��۴�B�в���������������ָ������º�¼C��Һ��λ�ã�����A��B�еμ������Լ�������������˳����__________________ (�����)����¼C��Һ��λ��ʱ����ƽ���⣬��Ӧ_________________��
(4)B�з�����Ӧ�Ļ�ѧ����ʽΪ___________________________________________��
(5)��ʵ������þ�Ͻ������Ϊa g������������Ϊb mL(�ѻ���Ϊ��״��)��B��ʣ����������Ϊc g�����������ԭ������Ϊ_____________________��
���𰸡�NaOH��Һ ��ȥ��þ�Ͻ���������Ĥ �٢ܢۢ� ʹD��C��Һ����ƽ 2Al+2NaOH+2H2O��2NaAlO2+3H2�� ![]()
��������
������ǿ�Ӧ��þ���ܣ�������һ�������ʿɴﵽʵ��Ŀ�ġ��ⶨ�Ͻ����������������ɳ�ȡһ�����Ͻ���Ʒ������������������������������ԭ���������ɲ���Ħ������=��������/�������ʵ�������������������������������ʵ�����
(1)A���Լ�Ϊǿ����Һ��������NaOH��Һ�ȡ�
(2)����þ���ǻ��ý������ڿ����б����γ���������Ĥ��ʵ��ǰ������þ�Ͻ���ϡ���н���Ƭ�̣�����Ҫ��ȥ���������Ĥ��
(3)ʵ������Ҫ�����Ʒ��������������Ӧ����������������ڼ�������ԡ�ҩƷ��ˮװ�����������Ӻ�װ�ú�����¼C��Һ��λ�ã�����A��B�еμ������Լ�������B�в���������������ָ������º�¼C��Һ��λ�ã�����B��ʣ�������ˣ�ϴ�ӣ�������أ�������˳�����٢ܢۢ�����¼C��Һ��λ��ʱ��Ӧ����D�ܸ߶�ʹD��C��Һ����ƽ����ƽ�Ӷ�����
(4)�Թ�B�ڣ��Ͻ��е�����NaOH��Һ��Ӧ�Ļ�ѧ����ʽΪ2Al+2NaOH+2H2O��2NaAlO2+3H2����
(5)��Al�����ԭ������Ϊx����
2Al ~ 3H2
2x g 3��22.4 L
(a-c) g b��10-3 L
��x=![]() ��
��


 ������ҵ��ͬ����ϰ��ϵ�д�
������ҵ��ͬ����ϰ��ϵ�д� С��ſ�ʱ��ҵϵ�д�
С��ſ�ʱ��ҵϵ�д� һ������ϵ�д�
һ������ϵ�д� �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ���ҴӺ�������ȡ���������ͼ��
![]()
���в���װ�ò�����Ҫ�����
A.  B.
B.  C.
C.  D.
D. 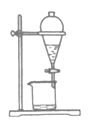
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����һ��������ܱ������У��������»�ѧ��Ӧ��N2(g)+3H2(g)![]() 2NH3(g) ��H��0���仯ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±�������������⡣
2NH3(g) ��H��0���仯ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±�������������⡣
t/�� | 25 | 125 | 225 | �� |
K | 4.1��105 | K1 | K2 | �� |
��1���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK=________��K1______K2����������������������������������ѹǿʹƽ��������Ӧ�����ƶ�����ƽ�ⳣ��_________������������������������
��2���жϸ÷�Ӧ�ﵽ��ѧƽ��״̬��������____________������ţ���
A��2��H2������= 3��NH3���棩 B�����������ܶȱ��ֲ���
C��������ѹǿ���ֲ��� D��N2���������ʵ���H2����������
E�������������ƽ����Է�����������ʱ����仯 F������������ɫ���ֲ���
��3������ͬ����N2��H2�ֱ�ͨ�뵽���Ϊ2L�ĺ����ܱ������У�����������Ӧ�õ������������ݣ�
ʵ���� | �¶ȣ��棩 | ��ʼ����mol�� | ƽ������mol�� | �ﵽƽ������ʱ�䣨min�� | |
N2 | H2 | NH3 | |||
1 | 650 | 2 | 4 | 0.9 | 9 |
2 | 900 | 1 | 2 | 0.3 | 0.01 |
ʵ��1������(NH3)��ʾ�ķ�Ӧ����Ϊ______��ʵ��2�����ʱ�ʵ��1���ԭ����____________��
��4��������Ʋ��ø��������ӵ������SCY�մɣ��ܴ���H+����ʵ���˳�ѹ�¼��ܺϳɰ����ܷ����ʵ��װ�ã���ͼ�������������ĵ缫��ӦΪ_____________��
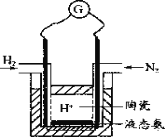
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��A��һ���¶����зֽⷴӦA (s) = B(s) + C(g) + 4D(g)����������ɵ������������ͬ��ѹ�£���ͬ���������10�����ҵ������ɵ������ڱ���µ����Ϊ22.4Lʱ������B������Ϊ30.4g��A��Ħ������Ϊ( )
A.120.4g/molB.50.4g/molC.182.4g/molD.252g/mol
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���������������ܰ뵼�����Ҫԭ�ϡ���ҵ�ϳ���п��ұ���ķ����л����ء���֪ijп������Ҫ��Zn��Si��Pb��Fe��Ga����������øÿ������صĹ����������£�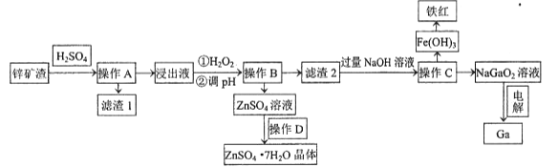
��֪��������Ԫ�����ڱ���λ�ڵ������ڵڢ�A�壬��ѧ�����������ơ�
��lg2=0.3 lg3=0.48��
�۲������ʵ�Ksp���±���ʾ��
���� | Zn(OH)2 | Ga(OH)3 | Fe(OH)2 | Fe(OH)3 |
Ksp | 1.6��10��17 | 2.7��10��31 | 8��10��16 | 2.8��10��39 |
(1)Ϊ�����������ʣ����ʵ���������Ũ���⣬Ӧ��ȡ�Ĵ�ʩ��__________________��д��������������1����Ҫ�ɷ�������Ǧ��_________________(д��ѧʽ)��
(2)����H2O2��Ŀ����(�����ӷ���ʽ��ʾ)______________________ ��
(3)���������£�������Һ�и������ӵ�Ũ�Ⱦ�Ϊ0.01mo/L������Һ��ij������Ũ��С��1��10-5mol/Lʱ����Ϊ����������ȫ��ȥ����pHӦ���ڵķ�ΧΪ___________________��
(4)����D������_______________���ˡ�ϴ�ӡ����
(5)��ⷨ�Ʊ������ء��ö��Ե缫���NaGaO2��Һ�����Ƶý����أ�д�������缫��Ӧʽ________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����A��B��C��D��E��F��G��H��I��ȫ���ɶ�����Ԫ����ɵľ��ֳ������ʡ���֪��������ֻ��B��C��FΪ���ʣ��ҳ��³�ѹ�£�BΪ��̬��C��FΪ��̬���ڻ������н�A��EΪ��̬�����ڳ����£�DΪ����ɫ���壻��H����ɫ��Ӧ�ʻ�ɫ�����ǵ�ת����ϵ��Ӧ����δע����ͼ��ʾ��
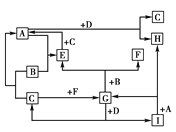
�Իش�(1)д���������ʵĻ�ѧʽ��E________��F________��H________��I________��
(2)д��A��D��Ӧ�Ļ�ѧ����ʽ��_______________��
(3)д��B��G��Ӧ�Ļ�ѧ����ʽ��______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����100 mL FeBr2��Һ��ͨ���״����2.24 L Cl2����Һ����![]() ��Br���������ɵ���Br2����ԭFeBr2��Һ��FeBr2�����ʵ���Ũ��Ϊ(����)
��Br���������ɵ���Br2����ԭFeBr2��Һ��FeBr2�����ʵ���Ũ��Ϊ(����)
A.4 mol L-1B.![]() mol L-1C.
mol L-1C.![]() mol L-1D.
mol L-1D.![]() mol L-1
mol L-1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������BN�������ж�����ṹ�������൪������ͨ�����ڵ��ȶ��࣬��ʯī���ƣ����в�״�ṹ�������������������ܵ��硣�����൪�����dz�Ӳ���ϣ����������ĥ�ԡ����ǵľ���ṹ��ͼ��ʾ�����������־����˵������ȷ����
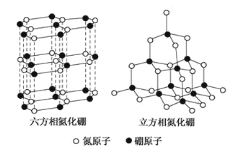
A. �����൪������λ��B��N
B. �����൪������������С�������ʵ������۵��
C. ���ֵ������е���ԭ�Ӷ��Dz���sp2�ӻ�
D. �����൪��������ṹ��ʯī����ȴ�����磬ԭ����û�п��������ƶ��ĵ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��1.52 gͭþ�Ͻ���ȫ�ܽ���50 mL�ܶ�Ϊ1.40 g/mL����������Ϊ63%��Ũ�����У��õ�NO2��N2O4�Ļ������1120 mL(��״��)����Ӧ�����Һ�м���1.0 mol/L NaOH��Һ������������ȫ������ʱ���õ�2.54 g����������˵������ȷ���ǣ� ��
A.�úϽ���ͭ��þ�����ʵ���֮����2:1
B.��Ũ������HNO3�����ʵ���Ũ����14.0 mol/L
C.�õ�2.54 g����ʱ������NaOH��Һ�������600 mL
D.NO2��N2O4�Ļ�������У�NO2�����������80%
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com