
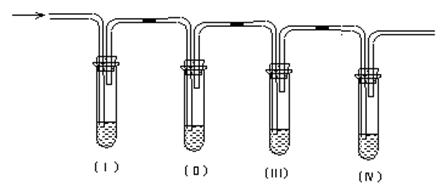
| A��Ʒ����Һ | B������������Һ | C��Ũ���� | D�����Ը��������Һ |


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
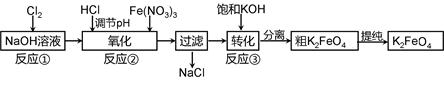
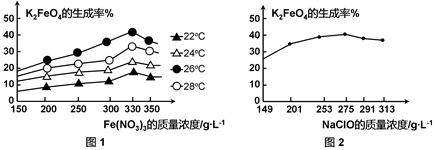
 4Fe(OH)3 + 8OH- + 3O2���ڡ��ᴿ��K2FeO4�в����ؽᾧ��ϴ�ӡ����º�ɵķ�������ϴ�Ӽ����ѡ�� ��Һ������ţ���
4Fe(OH)3 + 8OH- + 3O2���ڡ��ᴿ��K2FeO4�в����ؽᾧ��ϴ�ӡ����º�ɵķ�������ϴ�Ӽ����ѡ�� ��Һ������ţ���| A��H2O | B��CH3COONa������� | C��NH4Cl������� | D��Fe(NO3)3������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ | B����Һ | C������ | D������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
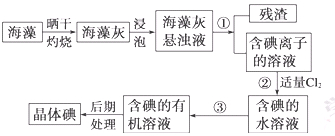
| A���ƾ� | B�����Ȼ�̼ | C������ | D������ |
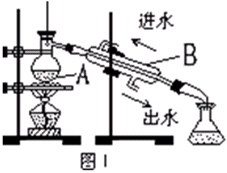
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��AgNO3��Һ | B��Ba(OH) 2��Һ | C��ϡ���� | D��NaOH��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| ѡ�� | ���ᴿ������ | ѡ�õ��Լ� | �������� |
| A | NaOH��Na2CO3�� | ���� | �� |
| B | CO2��CO�� | ���� | ���� |
| C | Fe��Zn�� | FeSO4��Һ | ���� |
| D | NaCl��Na2SO4�� | BaCl2��Һ | ��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 | 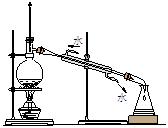 |
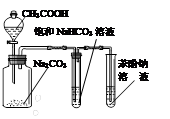 A��ʵ�����Ʊ���������ϩ A��ʵ�����Ʊ���������ϩ | B��ʯ�ͷ��� |
| | 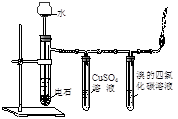 |
| C����֤���ᡢ̼�ᡢ��������ǿ�� | D��ʵ��������Ȳ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ڢݢܢ٢� | B���ܢ٢ڢݢ� |
| C���ܢڢݢ٢� | D���٢ܢڢݢ� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com