
£Ø13·Ö£©¶ąōĒ»łĮ¬ŌŚĶ¬Ņ»øöĢ¼Ō×ÓÉĻŅ××Ō¶ÆŹ§Ė®Éś³ÉČ©»ņĶŖ
£Ø1£©øł¾ŻÉĻŹöŌĄķ£¬ĒėŠ“³ö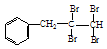 ÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ
£»
ÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ
£»
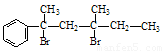 ÓėNaOHµÄ“¼ČÜŅŗ¹²ČČæÉŅŌÉś³É ÖÖĪČ¶ØµÄ¶žĻ©Ģž”£
ÓėNaOHµÄ“¼ČÜŅŗ¹²ČČæÉŅŌÉś³É ÖÖĪČ¶ØµÄ¶žĻ©Ģž”£
£Ø2£©Č©ĶŖŌŚĖ®ÖŠæÉÉś³ÉĖ®ŗĻĪļ£ŗ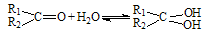 £» ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹”£Čō
£» ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹”£Čō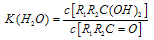 ĪŖČ©ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£¬ĘäÖµŌ½“óŌņ¶ŌÓ¦µÄČ©ĶŖĖ®ŗĻĪļŌ½ĪČ¶Ø”£ĻĀ±ķŹĒ²æ·ÖČ©”¢ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£ŗ
ĪŖČ©ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£¬ĘäÖµŌ½“óŌņ¶ŌÓ¦µÄČ©ĶŖĖ®ŗĻĪļŌ½ĪČ¶Ø”£ĻĀ±ķŹĒ²æ·ÖČ©”¢ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£ŗ
|
»ÆŗĻĪļ |
|
|
|
|
|
K(H2O) |
2”Į103 |
1.3 |
0.71 |
8.3”Į10-3 |
|
»ÆŗĻĪļ |
|
|
|
|
|
K(H2O) |
2”Į10-3 |
2.9 |
10 |
ŗÜ“ó |
øł¾ŻŅŌÉĻ±ķÖŠŹż¾Ż·ÖĪö£¬Ēė×ܽį³öĮ½Ģõ½įĀŪ£ŗ
¢Ł ”£
¢Ś ”£
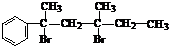
£Ø3£©¹¤ŅµÉĻÉś²ś±½¼×Č©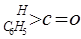 ÓŠŅŌĻĀĮ½ÖÖ·½·Ø£ŗ
ÓŠŅŌĻĀĮ½ÖÖ·½·Ø£ŗ
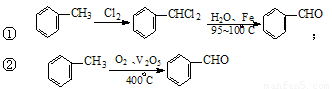
Óė·½·Ø¢ŁĻą±Č£¬·½·Ø¢ŚµÄÓŵćŹĒ £¬
ȱµćŹĒ ”£
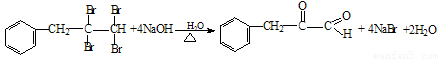
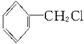
£Ø4£©±½ėźŹĒÓŠ»śČ¾ĮĻµÄÖ÷ŅŖŌĮĻ£¬ÓÉČ©£Ø»ņĶŖ£©Óė±½ėĀÉś³É±½ėźµÄ·“Ó¦ĪŖ£ŗ

·“Ó¦¢ŁµÄŌ×ÓĄūÓĆĀŹĪŖ100%£¬ŌņÖŠ¼ä²śĪļAµÄ½į¹¹¼ņŹ½ĪŖ £»
·“Ó¦¢ŚµÄ·“Ó¦ĄąŠĶŹōÓŚ ”£
£Ø5£©ÓɶŌ±½¶ž¼×Č©µÄŃõ»Æ²śĪļ”°¶Ō±½¶ž¼×Ėį”±Óė”°ŅŅ¶ž“¼”±Ėõ¾Ū£¬Éś³ÉµÄ¾Ūõ„ĻĖĪ¬£ØµÓĀŚ£©
µÄ½į¹¹¼ņŹ½ĪŖ ”£
£Ø13·Ö£©
£Ø1£©£ØŠ“ĪŖÓėĖ®·“Ó¦”¢NaOHŠ“ŌŚ¼żĶ·ÉĻ”¢²śĪļĪŖHBrŅ²æÉŅŌ£©£»£Ø2·Ö£© 5ÖÖ£Ø1·Ö£©
£Ø2£©ŅŌĻĀ£Ø¼°ĘäĖü£©ŗĻĄķ±ķŹö¾łøų·Ö£ŗ£Ø2·Ö£¬ĆææÕ1·Ö£©
a.ōŹ»łĢ¼ČēĮ¬½ÓµÄ¼×»łŌ½¶ą£¬Ļą¶ŌÓ¦µÄČ©ĶŖĖ®ŗĻĪļŌ½²»ĪČ¶Ø£»
b.ōŹ»łĢ¼ÉĻĮ¬½ÓĢž»łŹ±£¬Ģž»łµÄĢå»żŌ½“ó£¬Ė®ŗĻĪļŌ½²»ĪČ¶Ø£»
c. ōŹ»łĢ¼ĻąĮŚµÄĢ¼ÉĻĮ¬½ÓĮĖĀ±Ō×ÓŹ¹Ė®ŗĻĪļ±äµĆøü¼ÓĪČ¶Ø£»
d. ōŹ»łĢ¼ĻąĮŚµÄĢ¼ÉĻĮ¬½ÓĮĖĀ±Ō×ÓŌ½¶ą”¢Ā±Ō×Ó·Ē½šŹōŠŌŌ½Ē棬ĘäĖ®ŗĻĪļŌ½ĪČ¶Ø£Ø»ņōŹ»łĻąĮ¬µÄĢ¼Ō×ÓĮ¬ÉĻĀ±ĖŲŗó£¬øĆĢ¼Ō×ÓĮōĻĀµÄĒāŌ×ÓŌ½¶ą£¬ĘäĖ®ŗĻĪļŌ½²»ĪČ¶Ø£©
£Ø3£©·½·Ø¢ŚĪ“Ź¹ÓĆĘäĖüÓŠ¶¾ŌĮĻ£¬Éś³ÉĪļ³ż²śĘ·ĶāĮķŅ»ÖÖŹĒĪŽ¶¾ĪŽĪŪČ¾µÄĖ®£Ø1·Ö£© ·½·Ø¢Ś±Č·½·Ø¢ŁŠčŅŖµÄĪĀ¶Čøߣ¬Ļą¶ŌŗÄÄܽĻ¶ą£Ø1·Ö£©
£Ø4£©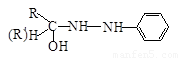 £Ø2·Ö£© ĻūČ„·“Ó¦£Ø1·Ö£©
£Ø2·Ö£© ĻūČ„·“Ó¦£Ø1·Ö£©
£Ø5£©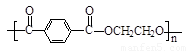 »ņ
»ņ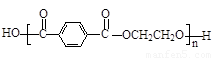 £Ø2·Ö£©
£Ø2·Ö£©
”¾½āĪö”æ



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
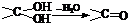 £®
£®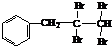 ÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ
ÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ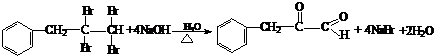
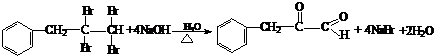
 ÓėNaOHµÄŅŅ“¼ČÜŅŗ¹²ČČ£¬æÉŅŌÉś³É
ÓėNaOHµÄŅŅ“¼ČÜŅŗ¹²ČČ£¬æÉŅŌÉś³É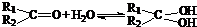 £®ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹£®ČōK(H2O)=
£®ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹£®ČōK(H2O)=| c[R1R2C(OH)2] |
| c[R1R2C=O] |
| »ÆŗĻĪļ |
|
|
|
| ||||||||
| K£ØH2O£© | 2”Į103 | 1.3 | 0.71 | 8.3”Į10-3 | ||||||||
| »ÆŗĻĪļ |
|
|
|
| ||||||||
| K£ØH2O£© | 2”Į10-3 | 2.9 | 10 | ŗÜ“ó |
H C6H5 |
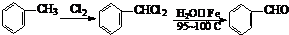 £»¢Ś
£»¢Ś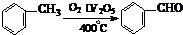
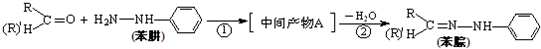
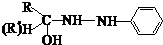
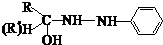
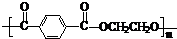 »ņ
»ņ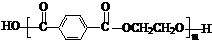
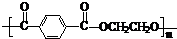 »ņ
»ņ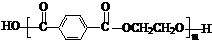
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĶ¼ŹĒ9øö»ÆŗĻĪļµÄ×Ŗ±ä¹ŲĻµ£ŗ
(1)»ÆŗĻĪļ¢ŁŹĒ___________£¬ĖüøśĀČĘų·¢Éś·“Ó¦µÄĢõ¼žŹĒ___________”£
(2)»ÆŗĻĪļ¢Żøś¢ßæÉŌŚĖįµÄ“ß»ÆĻĀČ„Ė®Éś³É»ÆŗĻĪļ¢į£¬¢įµÄ½į¹¹¼ņŹ½ŹĒ___________£¬Ćū³ĘŹĒ___________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
£Ø13·Ö£©¶ąōĒ»łĮ¬ŌŚĶ¬Ņ»øöĢ¼Ō×ÓÉĻŅ××Ō¶ÆŹ§Ė®Éś³ÉČ©»ņĶŖ
£Ø1£©øł¾ŻÉĻŹöŌĄķ£¬ĒėŠ“³öÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ £»
ÓėNaOHµÄ“¼ČÜŅŗ¹²ČČæÉŅŌÉś³É ÖÖĪČ¶ØµÄ¶žĻ©Ģž”£
£Ø2£©Č©ĶŖŌŚĖ®ÖŠæÉÉś³ÉĖ®ŗĻĪļ£ŗ£» ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹”£Čō
ĪŖČ©ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£¬ĘäÖµŌ½“óŌņ¶ŌÓ¦µÄČ©ĶŖĖ®ŗĻĪļŌ½ĪČ¶Ø”£ĻĀ±ķŹĒ²æ·ÖČ©”¢ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£ŗ
| »ÆŗĻĪļ |
|
|
|
|
| K(H2O) | 2”Į103 | 1.3 | 0.71 | 8.3”Į10-3 |
| »ÆŗĻĪļ |
|
|
|
|
| K(H2O) | 2”Į10-3 | 2.9 | 10 | ŗÜ“ó |
øł¾ŻŅŌÉĻ±ķÖŠŹż¾Ż·ÖĪö£¬Ēė×ܽį³öĮ½Ģõ½įĀŪ£ŗ
¢Ł ”£
¢Ś ”£
£Ø3£©¹¤ŅµÉĻÉś²ś±½¼×Č©![]() ÓŠŅŌĻĀĮ½ÖÖ·½·Ø£ŗ
ÓŠŅŌĻĀĮ½ÖÖ·½·Ø£ŗ
Óė·½·Ø¢ŁĻą±Č£¬·½·Ø¢ŚµÄÓŵćŹĒ £¬
ȱµćŹĒ ”£
£Ø4£©±½ėźŹĒÓŠ»śČ¾ĮĻµÄÖ÷ŅŖŌĮĻ£¬ÓÉČ©£Ø»ņĶŖ£©Óė±½ėĀÉś³É±½ėźµÄ·“Ó¦ĪŖ£ŗ
·“Ó¦¢ŁµÄŌ×ÓĄūÓĆĀŹĪŖ100%£¬ŌņÖŠ¼ä²śĪļAµÄ½į¹¹¼ņŹ½ĪŖ £»
·“Ó¦¢ŚµÄ·“Ó¦ĄąŠĶŹōÓŚ ”£
£Ø5£©ÓɶŌ±½¶ž¼×Č©µÄŃõ»Æ²śĪļ”°¶Ō±½¶ž¼×Ėį”±Óė”°ŅŅ¶ž“¼”±Ėõ¾Ū£¬Éś³ÉµÄ¾Ūõ„ĻĖĪ¬£ØµÓĀŚ£©
µÄ½į¹¹¼ņŹ½ĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010”Ŗ2011ѧğĮÉÄžŹ”ÉņŃōŹŠ¶«±±Óż²Å֊ѧø߶žĻĀѧʌŌĀæ¼ŹŌ»Æѧ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£Ø13·Ö£©¶ąōĒ»łĮ¬ŌŚĶ¬Ņ»øöĢ¼Ō×ÓÉĻŅ××Ō¶ÆŹ§Ė®Éś³ÉČ©»ņĶŖ
£Ø1£©øł¾ŻÉĻŹöŌĄķ£¬ĒėŠ“³ö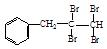 ÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ £»
ÓėNaOHµÄĖ®ČÜŅŗ¹²ČȵĻÆѧ·½³ĢŹ½ĪŖ£ŗ £»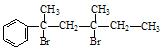 ÓėNaOHµÄ“¼ČÜŅŗ¹²ČČæÉŅŌÉś³É ÖÖĪČ¶ØµÄ¶žĻ©Ģž”£
ÓėNaOHµÄ“¼ČÜŅŗ¹²ČČæÉŅŌÉś³É ÖÖĪČ¶ØµÄ¶žĻ©Ģž”£
£Ø2£©Č©ĶŖŌŚĖ®ÖŠæÉÉś³ÉĖ®ŗĻĪļ£ŗ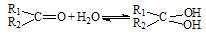 £»ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹”£Čō
£»ÕāŹĒŅ»øöæÉÄę·“Ó¦£¬Ę½ŗāדĢ¬£ØĘ½ŗāµÄĪ»ÖĆ£©¾ö¶ØÓŚČ©ĶŖµÄ½į¹¹”£Čō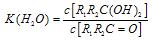 ĪŖČ©ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£¬ĘäÖµŌ½“óŌņ¶ŌÓ¦µÄČ©ĶŖĖ®ŗĻĪļŌ½ĪČ¶Ø”£ĻĀ±ķŹĒ²æ·ÖČ©”¢ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£ŗ
ĪŖČ©ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£¬ĘäÖµŌ½“óŌņ¶ŌÓ¦µÄČ©ĶŖĖ®ŗĻĪļŌ½ĪČ¶Ø”£ĻĀ±ķŹĒ²æ·ÖČ©”¢ĶŖµÄĖ®ŗĻĘ½ŗā³£Źż£ŗ
| »ÆŗĻĪļ |   | 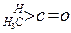 | 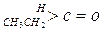 | 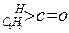 |
| K(H2O) | 2”Į103 | 1.3 | 0.71 | 8.3”Į10-3 |
| »ÆŗĻĪļ | 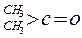 | 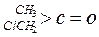 | 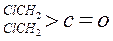 | 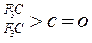 |
| K(H2O) | 2”Į10-3 | 2.9 | 10 | ŗÜ“ó |
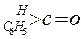 ÓŠŅŌĻĀĮ½ÖÖ·½·Ø£ŗ
ÓŠŅŌĻĀĮ½ÖÖ·½·Ø£ŗ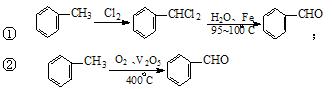

²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com