
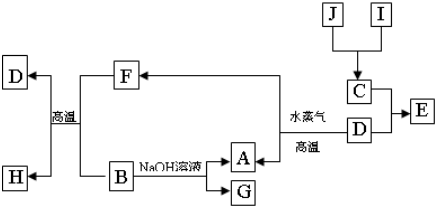
A��B��C��D�����ֳ������ʣ����ӦԪ�ص�ԭ������������������B��D���ڳ��������������Ϊ���������J��һ�ֺ�ɫ���壬I��Ũ��Һ���л�ԭ�ԣ�A��J����������֮�������µ�ת����ϵ�����ַ�Ӧ����ʡ�ԣ���
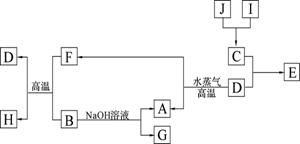
��1��BԪ�غ�CԪ�صļ����Ӱ뾶��С��ϵ�� �������ӷ��ű�ʾ���� _��
��2������Ԫ����C�γɵĻ�����NC3����ˮ�в���ʹ��ɫʯ����ֽ���������壬д���÷�Ӧ�Ļ�ѧ����ʽ ��
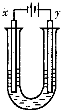
��3����E�ı�����Һ�����ˮ���γ���Һ�壬�ٽ���Һ��װ��U�ܣ�����U�ܵ����˲���缫����ֱͨ���磬�������˿ɹ۲쵽��������______________ ___��
��4��������J�����ữ��H2O2����Һ�У�J�ܽ���������+2�����ӣ��÷�Ӧ�����ӷ���ʽ��__________________________________________________ _��
��5������0��1 mol G����Һ�еμ�5 mol/L ��������Һ���õ�����3��9 g �������������������Ϊ______________________________��mL����
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
| ||
| ||
| ||
 Al��OH��3+OH-
Al��OH��3+OH- Al��OH��3+OH-
Al��OH��3+OH-�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��b��c�γɻ�����ĵ���ʽΪ
��b��c�γɻ�����ĵ���ʽΪ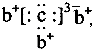 ���бȽ�����ȷ���ǣ�������
���бȽ�����ȷ���ǣ�������| A��ԭ�Ӱ뾶��a��c��d��b | B����ۺ����������c��d��a | C��ԭ��������a��d��b��c | D�����ʵ�������a��b��d��c |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com