
ЁОЬтФПЁПЃЈ1ЃЉПЦбЇМвПЩвдЭЈЙ§__________ЗЈЗЂЯжЬЋбєДцдкгаДѓСПЕФЬњдЊЫиЃЌаДГіЛљЬЌFeдзгЕФМлЕчзгХХВМЭМ__________________ЁЃДгНсЙЙЩЯЗжЮіFe3+БШFe2+ЮШЖЈЕФдвђЃК________________________ЁЃ
ЃЈ2ЃЉSCN-ГЃгУРДМьВтFe3+ЕФДцдкЃЌSCN-жааФдзгЕФдгЛЏЗНЪНЮЊ_________ЃЌШ§жждЊЫиЕчИКадгЩДѓЕНаЁЕФЫГађЮЊ__________________ЁЃ
ЃЈ3ЃЉЩщЛЏяивдЕкШ§ДњАыЕМЬхжјГЦЃЌШлЕуЮЊ1230ЁцЃЌОпгаПеМфЭјзДНсЙЙЁЃвбжЊЕЊЛЏХ№гыЩщЛЏяиЪєгкЭЌжжОЇЬхРраЭЁЃдђСНжжОЇЬхШлЕуНЯИпЕФЪЧ_________(ЬюЛЏбЇЪН)ЃЌЦфРэгЩЪЧ________________________ЁЃ
ЃЈ4ЃЉC60ЗжзггжУћзуЧђЯЉЃЌЫќЪЧгЩ60ИіЬМдзгзщГЩЕФЧђзДЗжзгЃЌгвЭМЮЊC60НсЙЙЭМЁЃвЛИіC60ЗжзгжаСљдЊЛЗЕФЪ§ФПЮЊ____________ЃЌдкC60ОЇАћжаC60ЗжзгЕФХфЮЛЪ§ЮЊ___________ЁЃвбжЊC60ЗжзгЕФЧђЬх АыОЖЮЊa nmЃЌЧѓC60ОЇЬхЕФІб=_________g/cm3ЁЃ(гУNAБэЪОАЂЗќМгЕТТоГЃЪ§ЃЌаДГіБэДяЪН)
ЁОД№АИЁП дзгЙтЦз ![]() Fe3+МлЕчзгХХВМЪНЮЊ3d5ЃЌЮЊАыТњНсЙЙЃЌЖјFe2+МлЕчзгХХВМЪНЮЊ3d6 sp N>S>C BN СНжжОЇЬхОљЮЊдзгОЇЬхЃЌN гыB дзгАыОЖНЯаЁЃЌМќФмНЯДѓЃЌШлЗаЕуИќИп 20 12
Fe3+МлЕчзгХХВМЪНЮЊ3d5ЃЌЮЊАыТњНсЙЙЃЌЖјFe2+МлЕчзгХХВМЪНЮЊ3d6 sp N>S>C BN СНжжОЇЬхОљЮЊдзгОЇЬхЃЌN гыB дзгАыОЖНЯаЁЃЌМќФмНЯДѓЃЌШлЗаЕуИќИп 20 12 ![]() ЁС1021
ЁС1021
ЁОНтЮіЁПЪдЬтЗжЮіЃКБОЬтПМВщдзгЙтЦзЁЂМлЕчзгХХВМЭМЕФЪщаДЁЂдгЛЏЗНЪНЕФХаЖЯЁЂЕчИКадДѓаЁЕФБШНЯЁЂОЇЬхШлЕуИпЕЭЕФБШНЯЁЂОЇАћЕФЗжЮіКЭМЦЫуЁЃ
ЃЈ1ЃЉМјЖЈдЊЫиГЃгУдзгЙтЦзЃЌПЦбЇМвПЩвдЭЈЙ§дзгЙтЦзЗЈЗЂЯжЬЋбєДцдкДѓСПЕФЬњдЊЫиЁЃFeдзгКЫЭтга26ИіЕчзгЃЌИљОнЙЙдьдРэЃЌЛљЬЌFeдзгЕФКЫЭтЕчзгХХВМЪНЮЊ[Ar]3d64s2ЃЌЛљЬЌFeдзгЕФМлЕчзгХХВМЪНЮЊ3d64s2ЃЌЛљЬЌFeдзгЕФМлЕчзгХХВМЭМЮЊЃК![]() ЁЃFe3+ЕФМлЕчзгХХВМЪНЮЊ3d5ЃЌЮЊАыТњНсЙЙНЯЮШЖЈЃЌFe2+ЕФМлЕчзгХХВМЪНЮЊ3d6ЃЌЫљвдFe3+БШFe2+ЮШЖЈЁЃ
ЁЃFe3+ЕФМлЕчзгХХВМЪНЮЊ3d5ЃЌЮЊАыТњНсЙЙНЯЮШЖЈЃЌFe2+ЕФМлЕчзгХХВМЪНЮЊ3d6ЃЌЫљвдFe3+БШFe2+ЮШЖЈЁЃ
ЃЈ2ЃЉSCN-гыCO2ЛЅЮЊЕШЕчзгЬхЃЌCO2жажааФдзгЕФдгЛЏЗНЪНЮЊspдгЛЏЃЌдђSCN-жажааФдзгЕФдгЛЏЗНЪНвВЮЊspдгЛЏЁЃИљОнЕчИКаддНДѓдЊЫиЕФЗЧН№ЪєаддНЧПЃЌЗЧН№ЪєадЃКN![]() S
S![]() CЃЌSЁЂCЁЂNШ§жждЊЫиЕчИКадгЩДѓЕНаЁЕФЫГађЪЧЃКN
CЃЌSЁЂCЁЂNШ§жждЊЫиЕчИКадгЩДѓЕНаЁЕФЫГађЪЧЃКN![]() S
S![]() CЁЃ
CЁЃ
ЃЈ3ЃЉЩщЛЏяиШлЕуНЯИпЃЌОпгаПеМфЭјзДНсЙЙЃЌЩщЛЏяиЪєгкдзгОЇЬхЃЛЕЊЛЏХ№гыЩщЛЏяиЪєгкЭЌжжОЇЬхРраЭЃЌЕЊЛЏХ№вВЪєгкдзгОЇЬхЃЌЕЊЁЂХ№ЕФдзгАыОЖаЁгкЩщЁЂяиЕФдзгАыОЖЃЌЕЊЛЏХ№жаЙВМлМќЕФМќФмДѓгкЩщЛЏяижаЙВМлМќЕФМќФмЃЌСНжжОЇЬхжаШлЕуНЯИпЕФЕЊЛЏХ№ЃЌЕЊЛЏХ№ЕФЛЏбЇЪНЮЊBNЁЃ
ЃЈ4ЃЉИљОнЭМЪОЃЌУПИіЬМдзгаЮГЩ3ИіЬМЬММќЃЈ2ИіЬМЬМЕЅМќКЭ1ИіЬМЬМЫЋМќЃЉЃЌУПИіЮхдЊЛЗжмЮЇга5ИіСљдЊЛЗЃЌУПИіСљдЊЛЗжмЮЇга3ИіЮхдЊЛЗКЭ3ИіСљдЊЛЗЃЌЮхдЊЛЗгыСљдЊЛЗЕФИіЪ§БШЮЊ3:5ЃЌЩшЮхдЊЛЗЁЂСљдЊЛЗЗжБ№гаxЁЂyИіЃЌдђxЃКy=3:5ЃЌ5x+6y=3![]() 60ЃЌНтЕУx=12ЃЌy=20ЃЌвЛИіC60ЗжзгжаСљдЊЛЗЕФЪ§ФПЮЊ20ЁЃдкC60ОЇАћжаC60ЗжзгВЩШЁЗжзгУмЖбЛ§ЃЌ1ИіC60ЗжзгжмЮЇЕШОрРызюНќЕФC60Зжзгга12ИіЃЌC60ЗжзгЕФХфЮЛЪ§ЮЊ12ЁЃC60ОЇАћЮЊУцаФСЂЗНзюУмЖбЛ§ЃЌгУЁАОљЬЏЗЈЁБЃЌ1ИіОЇАћжаКЌC60ЃК8
60ЃЌНтЕУx=12ЃЌy=20ЃЌвЛИіC60ЗжзгжаСљдЊЛЗЕФЪ§ФПЮЊ20ЁЃдкC60ОЇАћжаC60ЗжзгВЩШЁЗжзгУмЖбЛ§ЃЌ1ИіC60ЗжзгжмЮЇЕШОрРызюНќЕФC60Зжзгга12ИіЃЌC60ЗжзгЕФХфЮЛЪ§ЮЊ12ЁЃC60ОЇАћЮЊУцаФСЂЗНзюУмЖбЛ§ЃЌгУЁАОљЬЏЗЈЁБЃЌ1ИіОЇАћжаКЌC60ЃК8![]() +6
+6![]() =4ИіЃЌC60ЗжзгЕФЧђЬхАыОЖЮЊanmЃЌОЇАћЕФБпГЄЮЊ2
=4ИіЃЌC60ЗжзгЕФЧђЬхАыОЖЮЊanmЃЌОЇАћЕФБпГЄЮЊ2![]() anmЃЌ1ИіОЇАћЕФЬхЛ§ЮЊЃЈ2
anmЃЌ1ИіОЇАћЕФЬхЛ§ЮЊЃЈ2![]() anmЃЉ3=16
anmЃЉ3=16![]() a3
a3![]() 10-21cm3ЃЌ1molОЇЬхЕФЬхЛ§ЮЊ16
10-21cm3ЃЌ1molОЇЬхЕФЬхЛ§ЮЊ16![]() a3
a3![]() 10-21cm3
10-21cm3![]() 4
4![]() NA=4
NA=4![]() a3NA
a3NA![]() 10-21cm3ЃЌ1molC60ОЇЬхЕФжЪСПЮЊ720gЃЌC60ОЇЬхЕФІб=720g
10-21cm3ЃЌ1molC60ОЇЬхЕФжЪСПЮЊ720gЃЌC60ОЇЬхЕФІб=720g![]() ЃЈ4
ЃЈ4![]() a3NA
a3NA![]() 10-21cm3ЃЉ=
10-21cm3ЃЉ=![]() 1021g/cm3ЁЃ
1021g/cm3ЁЃ


 ЛюСІЪдОэЯЕСаД№АИ
ЛюСІЪдОэЯЕСаД№АИ ПЮПЮгХФмСІХргХ100ЗжЯЕСаД№АИ
ПЮПЮгХФмСІХргХ100ЗжЯЕСаД№АИ
| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁП40ЁцЪБЃЌдкАБ-ЫЎЬхЯЕжаВЛЖЯЭЈШыCO2ЃЌИїжжРызгБфЛЏЧїЪЦШчЭМЫљЪОЁЃЯТСаЫЕЗЈВЛе§ШЗЕФЪЧ
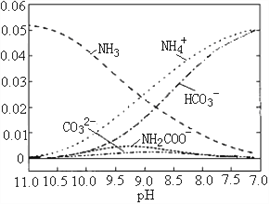
A. ЕБ8.5<pH<10.5ЪБЃЌЛсгажаМфВњЮяЃЈNH2COOЁЅЃЉЩњГЩ
B. дкpHЃН9.5ЪБЃЌШмвКжаДцдкЙиЯЕЃКc(NH4+)ЃОc(HCO3ЁЅ)ЃОc(NH2COOЁЅ)ЃОc(CO32ЁЅ)
C. дкpHЃН9.0ЪБЃЌШмвКжаДцдкЙиЯЕЃКc(NH4+)ЃЋc(H+)ЃН2c(CO32ЁЅ)ЃЋc(HCO3ЁЅ)ЃЋc(NH2COOЁЅ)ЃЋc(OHЁЅ)
D. ЫцзХCO2ЕФЭЈШыЃЌ![]() ВЛЖЯМѕаЁ
ВЛЖЯМѕаЁ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаа№Ъіе§ШЗЕФЪЧ
A. УїЗЏКЭЦЏАзЗлГЃгУгкздРДЫЎЕФОЛЛЏКЭЯћЖОЃЌСНепдРэЯрЭЌ
B. ГЃЮТЯТЃЌЭЌХЈЖШЕФNa2SгыNaHSШмвКЯрБШЃЌNa2SШмвКЕФpHаЁ
C. ЕШЮяжЪЕФСПХЈЖШЕФNH4ClШмвККЭNH4HSO4ШмвКЃЌКѓепЕФc(NH4ЃЋ)Дѓ
D. FeCl3гыKSCNЗДгІДяЕНЦНКтЪБЃЌМгШыKClШмвКЃЌдђШмвКбеЩЋБфЩю
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯжгаШ§зщЪЕбщЃКЂйГ§ШЅЛьдкжВЮягЭжаЕФЫЎЃЛЂкЖдЕтЫЎжаЕФЕтНјааХЈЫѕЃЛЂлЖд30%ЕФОЦОЋШмвКжаЕФОЦОЋНјааЬсДПЁЃвдЩЯЪЕбщВЩгУЕФе§ШЗЗНЗЈвРДЮЪЧЃЈ ЃЉ
A.ЗжвКЁЂнЭШЁЁЂеєСѓB.нЭШЁЁЂеєСѓЁЂЗжвК
C.ЗжвКЁЂеєСѓЁЂнЭШЁD.еєСѓЁЂнЭШЁЁЂЗжвК
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаЮяжЪжаМШФмЕМЕчЃЌгжЪєгкЧПЕчНтжЪЕФвЛзщЮяжЪЪЧЃЈ ЃЉ
A.ЪЏФЋЁЂДзЫсШмвКЁЂЪГбЮОЇЬх
B.ШлШкзДЬЌЕФKOHЁЂШлШкзДЬЌЕФNaCl
C.ЯЁH2SO4ЁЂNaOHШмвКЁЂДПHNO3
D.вКАБЁЂЪЏЛвЫЎЁЂЫЎвј
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЃЈ1ЃЉБъПіЯТ5.6LЕФCO2жЪСПЮЊ _____g,ЦфжаКЌга______ИіЗжзгЃЌКЌга_____ИідзгЃЛ
ЃЈ2ЃЉжЪСПОљЮЊm gЕФ HClЁЂNH3ЁЂCO2ЁЂO2ЫФжжЦјЬхЃЌЫљКЌЗжзгЪ§ФПзюЩйЕФЪЧ_________ЃЌЬхЛ§зюДѓЕФЪЧ_____ЃЌУмЖШзюаЁЕФЪЧ_______ЃЈдкЯрЭЌЮТЖШКЭбЙЧПЬѕМўЯТЃЉЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаааЮЊВЛЗћКЯЪЕбщАВШЋвЊЧѓЕФЪЧЃЈ ЃЉ
A.ЯЈУ№ОЦОЋЕЦЪБЃЌгУЕЦУБИЧУ№
B.ЕуШМЧтЦјЧАЃЌЯШНјаабщДПВйзї
C.ХфжЦЯЁСђЫсЪБЃЌНЋХЈСђЫсЛКТ§ЕЙШыЫЎжаВЂВЛЖЯНСАш
D.ДѓСПТШЦјаЙТЉЪБЃЌбИЫйРыПЊЯжГЁВЂОЁСПЭљЕЭДІШЅ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПБНМзЫсЙуЗКгІгУгкжЦвЉКЭЛЏЙЄаавЕЃЌФГЭЌбЇГЂЪдгУМзБНЕФбѕЛЏЗДгІжЦБИБНМзЫсЁЃ
ЂёЃЎжЦБИБНМзЫсКЭЛиЪеЮДЗДгІЕФМзБН
ЗДгІдРэЃК
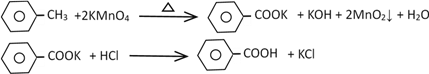
Ек1ВНЃКвЛЖЈСПЕФМзБНКЭKMnO4ШмвКдк100ЁцЗДгІвЛЖЮЪБМфКѓЭЃжЙЗДгІЃЛ
Ек2ВНЃКЙ§ТЫЗДгІЛьКЯЮяЃЌЕУЕНТЫвКЃЛОЙ§ВйзїЂёЃЌЕУЕНгаЛњВуКЭЫЎВуЃЛ
Ек3ВНЃКЯђгаЛњВужаМгЮоЫЎNa2SO4ЃЌЙ§ТЫЃЌОЙ§ВйзїЂђЃЌЕУЕНЮоЩЋвКЬхAЃЛ
Ек4ВНЃКЯђЫЎВуМгШыХЈбЮЫсЫсЛЏЃЌеєЗЂХЈЫѕЃЌРфШДЃЌЙ§ТЫЃЌЕУЕНАзЩЋЙЬЬхBЃЛ
вбжЊЃК
ЯрЖдЗжзгжЪСП | ШлЕу | ЗаЕу | УмЖШ | ШмНтЖШ | |
МзБН | 92 | Ѓ95Ёц | 110.8Ёц | 0.8669gЁЄmLЃ1 | МЋЮЂШмгкЫЎ |
БНМзЫс | 122 | 122.4Ёц | 249Ёц | 1.2659 gЁЄmLЃ1 | 0.3g (25ЁцЪБ) 6.9g (95ЁцЪБ) |
ЃЈ1ЃЉВйзїЂёЫљгУЕФВЃСЇвЧЦїгаЩеБКЭ______________ЃЌВйзїЂђЮЊ________________ЁЃ
ЃЈ2ЃЉЕк3ВНжаМгШыЮоЫЎNa2SO4ЕФФПЕФЪЧ_____________________ЃЌЮоЩЋвКЬхAЪЧ_______ЁЃ
ЂђЃЎЬсДПДжБНМзЫс
ИУЭЌбЇРћгУжиНсОЇЕФЗНЗЈЖдЕУЕНЕФBНјааЬсДПЁЃжиНсОЇЕФЙ§ГЬЃКМгШШШмНтЁњГУШШГщТЫЁњРфШДНсОЇЁњГщТЫЁњЯДЕгЁњИЩдяЁњДПБНМзЫсЁЃЃЈзЂЃКГщТЫзАжУШчЭМЫљЪОЃЌжївЊвЧЦїгаAВМЪЯТЉЖЗЃЌBГщТЫЦПЃЌГщЦјБУЕШЃЉ
ЃЈ3ЃЉАзЩЋЙЬЬхBжаЕФдгжЪЪЧ____________ЁЃ
ЃЈ4ЃЉГУШШГщТЫЕУЕНЕФТЫвКЗХжУРфШДПЩвдНсОЇГіДПОЛЕФБНМзЫсОЇЬхЃЌЮЊСЫЕУЕНИќЖрЕФБНМзЫсЃЌЪЧВЛЪЧЮТЖШдНЕЭдНКУВЂЫЕУїРэгЩ______ЃЈЬюЁАЪЧЁБЛђЁАВЛЪЧЁБЃЉЃЌРэгЩ__________________________________________________________ЁЃ
ЃЈ5ЃЉЪЙгУГщТЫзАжУБШЦеЭЈЙ§ТЫзАжУЕФгХЕуЪЧ_______________________________________ЁЃ
ЃЈ6ЃЉЪЕбщжаШЁМзБН10.0 mLЃЌжЦЕУБНМзЫс8.0gЃЌБОДЮЪЕбщжаБНМзЫсЕФВњТЪЮЊ____________ЃЈНсЙћБЃСєЕНаЁЪ§ЕуКѓвЛЮЛЃЉЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЂёЃЎеЦЮевЧЦїУћГЦЁЂзщзАМАЪЙгУЗНЗЈЪЧжабЇЛЏбЇЪЕбщЕФЛљДЁЃЌШчЭМЮЊеєСѓЪЕбщзАжУЁЃ
ЃЈ1ЃЉаДГіЯТСавЧЦїЕФУћГЦЃКaЃЎ__________ЃЛ bЃЎ___________ЃЛ
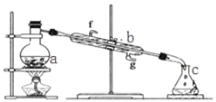
ЃЈ2ЃЉЪЕбщЙ§ГЬжаЃЌашвЊЭЈРфЫЎЃЌЭМжаЕФНјЫЎЗНЯђЪЧ_______НјЃЈЬюЭМжазжФИЃЉЃЛ
ЃЈ3ЃЉШєРћгУзАжУЗжРыввЫсЃЈЗаЕу118ЁцЃЉКЭввЫсввѕЅЃЈЗаЕу77ЁцЃЉЕФЛьКЯЮяЃЌЛЙШБЩйЕФвЧЦїЪЧ_______ЃЛ
IIЃЎЯжгУNaOHЙЬЬхХфжЦ0.1 mol/L NaOHШмвК480mLЃЌОнДЫЛиД№ЯТСаЮЪЬтЃК
ЃЈ4ЃЉХфжЦЧтбѕЛЏФЦШмвКашвЊгУЕНЕФВЃСЇвЧЦїгаЩеБЁЂВЃСЇАєЁЂНКЭЗЕЮЙмКЭ______ЃЛ
ЃЈ5ЃЉЪЕбщЪБашвЊГЦСПЧтбѕЛЏФЦ_______gЃЛ
ЃЈ6ЃЉХфжЦ0.1 mol/L NaOHШмвКЕФЪЕбщжаЃЌШчЙћГіЯжвдЯТВйзїЃЌЛсЕМжТХфжЦШмвКЕФХЈЖШЦЋДѓЕФга_______ЃЈЬюаДзжФИЃЉЁЃ
AЃЎГЦСПЧтбѕЛЏФЦЙЬЬхЪБэРТыЗХЗДСЫ BЃЎЮДЯДЕгШмНтNaOHЕФЩеБ
CЃЎNaOHдкЩеБжаШмНтКѓЃЌЮДРфШДОЭСЂМДзЊвЦЕНШнСПЦПжа
DЃЎШнСПЦПЮДИЩдяМДгУРДХфжЦШмвК EЃЎЖЈШнЪБИЉЪгПЬЖШЯп
FЃЎЖЈШнКѓШћЩЯЦПШћЗДИДвЁдШЃЌОВжУКѓЃЌвКУцВЛЕНПЬЖШЯпЃЌдйМгЫЎжСПЬЖШЯп
ВщПДД№АИКЭНтЮі>>
АйЖШжТаХ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com