
����þ����������θ����࣬����þ��(��Ҫ�ɷ�ΪMgCO3��������FeCO3)Ϊԭ���Ʊ��ߴ�����þ��ʵ���������£�
��1���������н��������ι��˲������ò�������IJ��������� ��
��2������H2O2����ʱ��������Ӧ�����ӷ���ʽΪ ��
��3������2�ijɷ��� (�ѧʽ)��
��4�����չ��̴������·�Ӧ��
2MgSO4��C 2MgO��2SO2����CO2��
2MgO��2SO2����CO2��
MgSO4��C MgO��SO2����CO��
MgO��SO2����CO��
MgSO4��3C MgO��S����3CO��
MgO��S����3CO��
������ͼװ�ö����ղ�����������зֲ����ջ��ռ�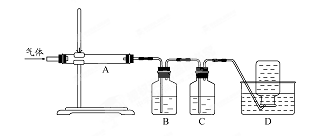
��D���ռ������������ (�ѧʽ)��
��B��ʢ�ŵ���Һ������ (����ĸ)��
a NaOH ��Һ b Na2CO3��Һ c ϡ���� d ����KMnO4��Һ
��1���ձ���©������������2��2Fe2++H2O2+2H+=2Fe3++2H2O ��3��Fe��OH��3
��4�� ��CO ��d
���������������1�����˲��������貣������Ϊ�ձ�����������©����
��2������H2O2ʱ������Ӧ2Fe2++H2O2+2H+=2Fe3++2H2O ���백ˮʱ���ɳ���Fe��OH��3
��4��CO�������������ʷ�Ӧ���Կ�����D���ռ���B��ʢ������KMnO4��Һ����ͬʱ����SO2��S��
���㣺������ԭ��Ӧ����ʵ�������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����пΪ��ɫ��ĩ��������ʪ�Ѣ��Ƥ���������ơ�������ҵ������п[����Fe����Mn����Ni��������]���������£�
��ʾ���ڱ�ʵ�������£�Ni�����ܱ�������������صĻ�ԭ������MnO2��
�ش��������⣺
��1����Ӧ���г���������������________��������Ӧ�����ӷ���ʽΪ________���ڼӸ��������Һǰ����pH�ϵͣ��Գ��ӵ�Ӱ����________��
��2����Ӧ�۵ķ�Ӧ����Ϊ________�����˵õ��������У����˹�����п���________��
��3����Ӧ���γɵij���Ҫ��ˮϴ����������Ƿ�ϴ�Ӹɾ��ķ�����______________��
��4����Ӧ���в���ijɷֿ�����ZnCO3��xZn��OH��2��ȡ�������˱�11.2 g�����պ�ɵõ���Ʒ8.1 g����x����________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ѡ������ʵ�鷽���������ʣ�����õķ��뷽����������ں����ϡ�
| A����ȡ��Һ�� | B�����ȷ� | C���ᾧ�� | D����Һ�� E�����˷� F������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ƣ�NaClO2����Ҫ�����ġ���ֽҵ��Ư����Ҳ����ʳƷ������ˮ�����ȣ��������������ֽ⡣�������Ƶ�Ϊԭ���Ʊ��������ƵĹ����������£�
��1����ߡ���Ӧl����Ӧ���ʵĴ�ʩ��________________________��д��һ�����ɣ���
��2������Ӧ2������������_____________���÷�Ӧ�Ļ�ѧ����ʽΪ__________________��
��3����ȡ����ѹ�����������á���ѹ��������ԭ����__________________��
��4���ӡ�ĸҺ���пɻ��յ���Ҫ������__________________________��
��5������ȴ�ᾧ����______________����������ƣ����ɻ�ôֲ�Ʒ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����Ǿ�Ļ�ѧ��Դ���⡣�����Ǻ�ˮ��ѧ��Դ�ۺ����õIJ�������ͼ��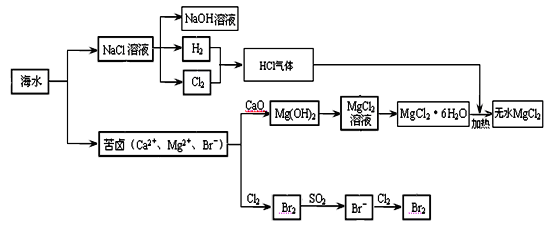
�ش�
��1���ɺ�ˮɹ�ƵĴ����к���Ca2+��Mg2+��SO42�������ӣ�Ϊ�˳�ȥ��Щ���ӣ���Ҫ���μ����Թ�����NaOH��BaCl2��__ ___�����Լ���ѧʽ����Ȼ��__ ___����������ƣ�������Һ���ټ����� �����Լ���������������Һ���õ����Ρ�
��2��ijͬѧ��ʵ����ģ���ȼҵ������ԭ����ⱥ��ʳ��ˮ���ò�����պŨ��ˮ�����������������壬���ֲ����������̡��������ɵ������� �����̵���Ҫ�ɷ��� ��
��3����ȡMgCl2�Ĺ������漰��Ӧ��MgCl2��6H2O MgCl2 + 6H2O���÷�ӦҪ��HCl�����н��У�ԭ���� ��
MgCl2 + 6H2O���÷�ӦҪ��HCl�����н��У�ԭ���� ��
��4����±��ͨ��Cl2�û���Br2����������SO2����ת��ΪBr����������Σ��Դﵽ�������Ŀ�ġ��ɺ�ˮ��������еķ�Ӧ�ɵó�Cl����SO2��Br����ԭ����ǿ������˳����_________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
̼��﮹㷺Ӧ�����մɺ�ҽҩ������,�Ԧ¡�﮻�ʯ(��Ҫ�ɷ�ΪLi2O��Al2O3��4SiO2)Ϊԭ���Ʊ�Li2CO3�Ĺ�����������: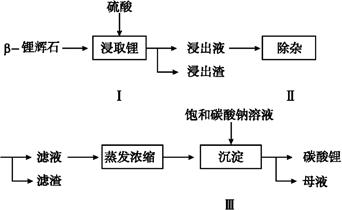
��֪:Fe3+��Al3+��Fe2+��Mg2+������������ʽ��ȫ����ʱ,��Һ��pH�ֱ�Ϊ3.2��5.2��9.7��12.4;Li2SO4��LiOH��Li2CO3��303 K�µ��ܽ�ȷֱ�Ϊ34.2 g��12.7 g��1.3 g��
��1�������ǰ,�¡�﮻�ʯҪ�����ϸ������Ŀ������������������������������������������
��2���������,�����õ���������Һ�к���Li+��SO42-��������Al3+��Fe3+��Fe2+��Mg2+��Ca2+��Na+������,���ڽ����¼���������(�ʯ��ʯ�������Ȼ��ơ���ϡ���ᡱ)�Ե�����Һ��pH��6.0~6.5,����������������,Ȼ�����õ�����Һ��
��3���������,��������H2O2��Һ��ʯ�����Na2CO3��Һ���μ������Һ��,�ɳ�ȥ�����ʽ�������������������������������������������
��4���������,���ɳ��������ӷ���ʽΪ�� ��
��5����ĸҺ�пɻ��յ���Ҫ������������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣�ij�о���ѧϰС���������ռ���������Ϣ�����ƣ��׳Ƴ����һ��ǿ��ԭ�����ڹ�ҵ����;�㷺�����Ƕ�������������Ƶķ�Ӧ����̽����
��ʵ�顿
�ڴ���������Һ�м���������Һ��������Һʼ�ճ��塣
��1�������й����Ƶ�˵����ȷ���� ��
a. ������Һ���˾��ã�Ӧ��������
b. ������Һ��ͨ��SO2�е���ɫ���ǣ���֤��SO2��������
c. �����ڿ����г��Ⲣ���Ϸų�H2S���壬˵�����ԣ�H2CO3>H2S
d. ����������Һʱ����������������
e. ��������Һ�еμ�ϡ�����г�������ζ����������
��������衿
��2����Ӧ�����Һ�п϶�����Cl����SO42����OH����Ϊ̽����Һ�п��ܻ����ڵ����������ӣ�����������ˮ�⼰������CO2��Ӱ�죩����ѧϰС��������¼��裺
����1��ֻ������S2��
����2��ֻ������ClO��
����3��
�����ʵ�鷽������֤���衿
��3����д��ʵ�鲽���Լ�Ԥ������ͽ��ۡ�
��ѡʵ���Լ���1 mol? L��1��CuSO4��Һ��1 mol? L��1NaOH��Һ�����ۣ�KI��Һ����ɫʯ����Һ����̪��Һ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ������Ӧ����Һ���Թ��У��μ� | �� ������� ������ |
| ����:2����ȡ������Ӧ����Һ���Թ��У��μ� | �� ������� ������ �� �� �����3������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ҵ�������ų������ı���[��Ҫ���� ��
�� ��
�� ��
��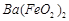 ��]��ij��Ҫ����
��]��ij��Ҫ����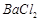 ��
��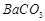 ��
��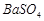 �Ļ��������ñ�����ȡ
�Ļ��������ñ�����ȡ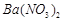 ���䲿�ֹ����������£�
���䲿�ֹ����������£�
��1�����ܺ���Һ��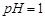 ��
��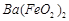 ��
��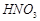 �ķ�Ӧ��ѧ����ʽΪ ��
�ķ�Ӧ��ѧ����ʽΪ ��
��2������ʱͨ�����Ʒ�Ӧ�¶Ȳ�����70�棬�Ҳ�ʹ��Ũ���ᣬ
ԭ���� �� ��
��3���ó���ϱ���ʵ�ʣ�ѡ�õ�XΪ ���ѧʽ�����к͢�ʹ��Һ�� �������ӷ��ţ���Ũ�ȼ��٣��к͢��������Һ����仯�ɺ��ԣ���
��4������������ϴ�ӵ�Ŀ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
һѧ�����������ʵ�鷽������NaCl��CaCl2���ֹ�������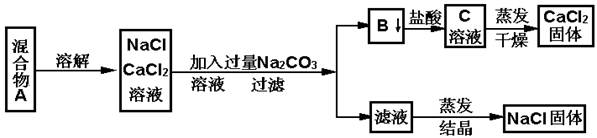
��պͻش��������⣺
��1��B������ ��д��ѧʽ��
��2������ʵ�鷽������õ���NaCl���������������ʣ�����Ϊ����������ijһ�������ë������һ������ȷ��Ʒ���Ӧ�� ��
��3����Ҫ�ⶨ����Ʒ��NaCl��CaCl2�������ȣ��ɳ��������B���ʺ���һ���ʵ����������������� ��
��4����д����B��C�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com