
”¾ĢāÄæ”æ[»ÆѧŅ»Ń”ŠŽ3: ĪļÖŹ½į¹¹ÓėŠŌÖŹ]
A”¢B”¢C”¢DĪŖŌŖĖŲÖÜĘŚ±ķÖŠĒ°ĖÄÖÜĘŚµÄĖÄÖÖŌŖĖŲ£¬ĒŅŌ×ÓŠņŹżŅĄ“ĪŌö“ó£¬C”¢DĶ¬ÖÜĘŚ£¬ĒŅA”¢B”¢C¾ł²»ŌŚĶ¬Ņ»ÖÜĘŚ£¬ĘäÖŠAÓėDĶ¬Ö÷×壬ĒŅøĆÖ÷×åÖŠÓŠČżÖÖ·Ē½šŹōŌŖĖŲ£¬BĪŖ·Ē½šŹōŌŖĖŲĒŅŌ×Ó×īĶā²ćÓŠ3¶Ō³É¶Ōµē×Ó£¬CŌŖĖŲĪ»ÓŚŌŖĖŲÖÜĘŚ±ķµŚ10ĮŠ”£Ēė»Ų“šĻĀĮŠĪŹĢā:
(1)CŌŖĖŲµÄŌ×ÓŠņŹżĪŖ________£¬»łĢ¬DŌ×ӵļņ»Æµē×ÓÅŲ¼Ź½ĪŖ________”£
(2)ÓėAĶ¬Ņ»ÖÜĘŚµÄĖłÓŠÖ÷×åŌŖĖŲÖŠµŚŅ»µēĄėÄÜŠ”ÓŚAµÄŌŖĖŲ¹²ÓŠ_______ÖÖ”£
(3) DŌŖĖŲæÉŠĪ³ÉDX3µÄĀ±»ÆĪļ£¬ŠŌÖŹŗĶ½į¹¹ÓėAX3ĄąĖĘ(X±ķŹ¾Ā±ĖŲŌŖĖŲ)£¬ŌŚĖ®ČÜŅŗÖŠĒæĮŅĖ®½ā£¬ĒėŠ“³öDCl3ÓėĖ®·“Ó¦µÄ»Æѧ·½³ĢŹ½:____________”£
(4) ČēĶ¼ĪŖD2B3µÄ·Ö×Ó½į¹¹Ķ¼£¬BŌӻƷ½Ź½ĪŖ____________”£
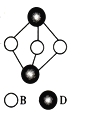
(5) ŹŌ±Č½ĻA”¢BŠĪ³ÉµÄ¼ņµ„Ēā»ÆĪļµÄČŪ·ŠµćµÄøßµĶ²¢½āŹĶ£ŗ________ (¼ņµ„Ēā»ÆĪļÓĆ·Ö×ÓŹ½±ķŹ¾)”£
(6) ŃŠ¾æ¾§Ģå½į¹¹µÄ×ī³£ÓĆ·½·ØŹĒ________£»ČēĶ¼ĪŖŅ»ÖÖŗ¬CŗĶDĮ½ÖÖŌŖĖ÷µÄĘ·ĢåµÄ¾§°ūĶ¼£¬³ŹĮł·½½į¹¹£¬¾§°ū²ĪŹżĪŖµ×Ćę±ß³¤ĪŖanm”¢øßĪŖbmm”£
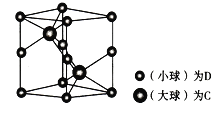
¢ŁøĆ¾§°ū×é³ÉĪļÖŹµÄ»ÆѧŹ½ĪŖ__________”£
¢ŚŅŃÖŖDŗĶCµÄĻą¶ŌŌ×ÓÖŹĮæ·Ö±šĪŖM1ŗĶM2,Čō¾§ĢåĆܶČĪŖ¦Ńg/cm3,NA±ķŹ¾°¢·ü¼ÓµĀĀŽ³£Źż£¬ÓĆŗĻĄķ±ķ“ļŹ½±ķŹ¾øĆ¾§ĢåµÄĆܶČĪŖ________g/cm3 (ĮŠ³ö¼ĘĖćŹ½¼“æÉ£¬²»±Ų»Æ¼ņ)”£
”¾“š°ø”æ 28 [Ar]3d104s24p3 5 AsCl3+3H2O=H3AsO3+3HCl sp3ŌÓ»Æ NH3£¾HCl£¬NH3“ęŌŚ·Ö×Ó¼äĒā¼ü£¬HCl·Ö×Ó¼äÖ»“ęŌŚ·¶µĀ»ŖĮ¦£¬Ēā¼ü×÷ÓĆĮ¦±Č·¶µĀ»ŖĮ¦Ē棬NH3µÄČŪ·ŠµćøßÓŚHCl X-ÉäĻßŃÜÉä NiAs»ņAsNi 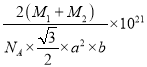
”¾½āĪö”æŹŌĢā·ÖĪö£ŗ±¾Ģāæ¼²éĪļÖŹ½į¹¹ÓėŠŌÖŹ£¬Éę¼°ŌŖĖŲµÄĶʶĻ£¬¼ņ»Æµē×ÓÅŲ¼Ź½µÄŹéŠ“£¬µŚŅ»µēĄėÄÜµÄ±Č½Ļ£¬ŌӻƷ½Ź½µÄÅŠ¶Ļ£¬ČŪ·ŠµćøßµĶµÄ±Č½Ļ£¬¾§°ūµÄ·ÖĪöŗĶ¼ĘĖć”£CŹĒĒ°ĖÄÖÜĘŚŌŖĖŲ£¬CŌŖĖŲĪ»ÓŚŌŖĖŲÖÜĘŚ±ķµŚ10ĮŠ£¬CĪŖNiŌŖĖŲ£¬Ni“¦ÓŚµŚĖÄÖÜĘŚ£¬C”¢DĶ¬ÖÜĘŚ£¬DŅ²“¦ÓŚµŚĖÄÖÜĘŚ£»A”¢B”¢C”¢DµÄŌ×ÓŠņŹżŅĄ“ĪŌö“ó£¬A”¢B”¢C¾ł²»ŌŚĶ¬Ņ»ÖÜĘŚ£¬AÓėDĶ¬Ö÷×åĒŅøĆÖ÷×åÖŠÓŠČżÖÖ·Ē½šŹōŌŖĖŲ£¬AĪŖNŌŖĖŲ£¬DĪŖAsŌŖĖŲ£»B“¦ÓŚµŚČżÖÜĘŚ£¬BĪŖ·Ē½šŹōĒŅŌ×Ó×īĶā²ćÓŠ3¶Ō³É¶Ōµē×Ó£¬BĪŖClŌŖĖŲ”£
£Ø1£©CĪŖNiŌŖĖŲ£¬NiµÄŌ×ÓŠņŹżĪŖ28”£DĪŖAs£¬AsµÄŌ×ÓŠņŹżĪŖ33£¬øł¾Ż¹¹ŌģŌĄķ£¬»łĢ¬AsµÄŗĖĶāµē×ÓÅŲ¼Ź½ĪŖ1s22s22p63s23p63d104s24p3£¬¼ņ»Æµē×ÓÅŲ¼Ź½ĪŖ[Ar] 3d104s24p3”£
£Ø2£©AĪŖN£¬N“¦ÓŚµŚ¶žÖÜĘŚ£¬µŚ¶žÖÜĘŚÖŠµŚŅ»µēĄėÄÜŠ”ÓŚNµÄŌŖĖŲÓŠLi”¢Be”¢B”¢C”¢O£¬¹²5ÖÖ”£
£Ø3£©AsCl3ŌŚĖ®ČÜŅŗÖŠĒæĮŅĖ®½āÉś³ÉH3AsO3ŗĶHCl£¬·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖAsCl3+3H3O=H3AsO3+3HCl”£
£Ø4£©ÓÉŹ¾ŅāĶ¼æɼūBŠĪ³É2øö¦Ņ¼ü£¬BÉĻ»¹ÓŠĮ½¶Ō¹Āµē×Ó¶Ō£¬BµÄ¼Ū²ćµē×Ó¶ŌŹżĪŖ4£¬BµÄŌӻƷ½Ź½ĪŖsp3ŌӻƔ£
£Ø5£©AµÄ¼ņµ„Ēā»ÆĪļĪŖNH3£¬BµÄ¼ņµ„Ēā»ÆĪļĪŖHCl”£ÓÉÓŚNH3“ęŌŚ·Ö×Ó¼äĒā¼ü£¬HCl·Ö×Ó¼äÖ»“ęŌŚ·¶µĀ»ŖĮ¦£¬Ēā¼ü×÷ÓĆĮ¦±Č·¶µĀ»ŖĮ¦Ē棬NH3µÄČŪ·ŠµćøßÓŚHCl”£
£Ø6£©ŃŠ¾æ¾§Ģå½į¹¹×ī³£ÓƵķ½·ØŹĒX-ÉäĻßŃÜÉ䔣
£Øa£©ÓĆ”°¾łĢÆ·Ø”±£¬DĪŖAs£ŗ8![]() +4
+4![]() =2£¬CĪŖNi£¬CČ«ŌŚ¾§°ūÄŚ£¬Ni£ŗ2£¬NiÓėAsµÄøöŹż±ČĪŖ2:2=1:1£¬øĆ¾§ĢåµÄ»ÆѧŹ½ĪŖNiAs»ņAsNi”£
=2£¬CĪŖNi£¬CČ«ŌŚ¾§°ūÄŚ£¬Ni£ŗ2£¬NiÓėAsµÄøöŹż±ČĪŖ2:2=1:1£¬øĆ¾§ĢåµÄ»ÆѧŹ½ĪŖNiAs»ņAsNi”£
£Øb£©øĆ¾§°ū³ŹĮł·½½į¹¹£¬¾§°ūµÄµ×Ć껿ĪŖa![]() 10-7cm
10-7cm![]() a
a![]() 10-7cm=
10-7cm=![]() a2
a2![]() 10-14cm2£¬¾§°ūµÄĢå»żĪŖ
10-14cm2£¬¾§°ūµÄĢå»żĪŖ![]() a2
a2![]() 10-14cm2
10-14cm2![]() b
b![]() 10-7cm=
10-7cm=![]() a2b
a2b![]() 10-21cm3£¬1mol¾§ĢåµÄĢå»żĪŖ
10-21cm3£¬1mol¾§ĢåµÄĢå»żĪŖ![]() a2b
a2b![]() 10-21cm3
10-21cm3![]() 2
2![]() NA£»¾§ĢåµÄ»ÆѧŹ½ĪŖNiAs»ņAsNi£¬1mol¾§ĢåµÄÖŹĮæĪŖ£ØM1+M2£©g£»¾§ĢåµÄĆܶČ=
NA£»¾§ĢåµÄ»ÆѧŹ½ĪŖNiAs»ņAsNi£¬1mol¾§ĢåµÄÖŹĮæĪŖ£ØM1+M2£©g£»¾§ĢåµÄĆܶČ=![]() =£ØM1+M2£©g
=£ØM1+M2£©g![]() £Ø
£Ø![]() a2b
a2b![]() 10-21cm3
10-21cm3![]() 2
2![]() NA£©=
NA£©=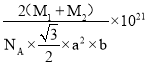 g/cm3”£
g/cm3ӣ


 æĪĢĆČ«½ā×Ö“Ź¾ä¶ĪĘŖÕĀĻµĮŠ“š°ø
æĪĢĆČ«½ā×Ö“Ź¾ä¶ĪĘŖÕĀĻµĮŠ“š°ø ²½²½øßæŚĖćĢāæØĻµĮŠ“š°ø
²½²½øßæŚĖćĢāæØĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĘū³µĪ²ĘųÖ÷ŅŖŗ¬ÓŠCO2 ”¢CO”¢SO2”¢NOx µČĪļÖŹ£¬Ī²ĘųÖŠCO”¢ĒāŃõ»ÆĪļ(NOx) ŃĻÖŲÓ°ĻģČĖĆĒµÄÉś»īŗĶ½”æµ£¬»Æѧ¹¤×÷Õ߶ŌµŖŃõ»ÆĪļµÄ“¦Ąķ×öĮĖ¹ć·ŗ¶ųÉīČėµÄŃŠ¾æ”£
£Ø1£©ĄūÓĆ¼×Ķ黹ŌNOx
¢ŁCH4(g)+4NO2(g)=4NO(g)+CO2(g)+2H2O(g)”÷H1=-574kJ/mo l£»
¢ŚCH4(g)+4NO(g)=2N2(g)+CO2(g)+H2O(g) ”÷H2=-1160kJ/mo l,
¼×ĶéÖ±½Ó½«NO2»¹ŌĪŖN2µÄČČ»Æѧ·½³ĢŹ½ĪŖ________________________________”£
£Ø2£©½«²»Ķ¬ĮæµÄCO(g)»żH2O(g)·Ö±šĶØČėĢå»żĪŖ2LµÄŗćČŻĆܱÕČŻĘ÷ÖŠ£¬½ųŠŠ·“Ó¦CO(g)+H2O(g)![]() CO2(g)+H2(g)£¬µĆµ½ČēĻĀČż×鏿¾Ż£ŗ
CO2(g)+H2(g)£¬µĆµ½ČēĻĀČż×鏿¾Ż£ŗ
ŹµŃé×é | ĪĀ¶Č”ę | ĘšŹ¼Įæ/mol | Ę½ŗāĮæ/mol | “ļµ½Ę½ŗāĖłŠčŹ±¼ä/min | ||
CO | H2O | H2 | CO | |||
1 | 650 | 4 | 2 | 1.6 | 2.4 | 6 |
2 | 900 | 2 | 1 | 0.4 | 1.6 | 4 |
3 | 900 | a | b | c | d | t |
¢ŁŹµŃé1ÖŠŅŌv(CO2) ±ķŹ¾µÄ·“Ó¦ĖŁĀŹĪŖ/span>_________(±£ĮōĮ½Ī»ÓŠŠ§Źż×Ö£¬ĻĀĶ¬)”£
¢ŚøĆ·“Ó¦ĪŖ________(Ģī”°ĪüČČ”±»ņ”°·ÅČČ”±) ·“Ó¦£¬ŹµŃé2Ģõ¼žĻĀµÄĘ½ŗā³£ŹżK=________”£
¢ŪČō“ļĘ½ŗāדĢ¬Ź±£¬ŹµŃé2 ÓėŹµŃé3ÖŠø÷ĪļÖŹµÄÖŹĮæ·ÖŹż·Ö±šĻąµČ£¬ĒŅt£¼4min,Ōņa”¢bÓ¦Āś×ćµÄ¹ŲĻµŹĒ_________________________(ÓĆŗ¬a”¢bµÄŹżŃ§Ź½±ķŹ¾)”£
£Ø3£©CO·ÖĪöŅĒµÄ“«øŠĘ÷æɲā¶ØĘū³µĪ²ĘųŹĒ·ń·ūŗĻÅŷűź×¼”£øĆ·ÖĪöŅĒµÄ¹¤×÷ŌĄķĄąĖĘÓŚČ¼ĮĻµē³Ų£¬ĘäÖŠµē½āÖŹŹĒŃõ»ÆīĘ(Y2O3) ŗĶŃõ»ÆļÆ(ZrO2) ¾§Ģ壬ÄÜ“«µ¼O2-£¬Ōņøŗ¼«µÄµē¼«·“Ó¦Ź½ĪŖ____________”£
£Ø4£©SO2æÉÓĆøĘ»ł¹ĢĮņ·“Ó¦³żČ„£¬·“Ӧɜ³ÉµÄCaSO4ŹĒŅ»ÖÖĪ¢ČÜĪļÖŹ£¬ĘäKsp=9.0”Į10-6”£Čō½«ÅضČĪŖ2”Į10-3mol/LµÄNa2SO4ČÜŅŗÓėµČĢå»żµÄCaCl2ČÜŅŗ»ģŗĻ£¬ŌņÉś³É³ĮµķĖłŠčCaCl2ČÜŅŗµÄ×īŠ”ÅضČĪŖ_____________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ1869Äź£¬¶ķ¹ś»Æѧ¼ŅĆŽŻĮŠ·ņÖĘ×÷³öĮĖµŚŅ»ÕÅŌŖĖŲÖÜĘŚ±ķ£¬½ŅŹ¾ĮĖ»ÆѧŌŖĖŲ¼äµÄÄŚŌŚĮŖĻµ£¬³ÉĪŖ»ÆѧŹ·ÉĻµÄÖŲŅŖĄļ³Ģ±®Ö®Ņ»”£ĻĀĮŠÓŠ¹ŲŌŖĖŲÖÜĘŚ±ķµÄĖµ·ØÕżČ·µÄŹĒ£Ø £©
A.ŌŖĖŲÖÜĘŚ±ķŗ¬ŌŖĖŲ×ī¶ąµÄ×åŹĒµŚ¢óB×å
B.ŌŖĖŲÖÜĘŚ±ķÓŠ18øö×å
C.µŚ¢ńA×åµÄŌŖĖŲČ«²æŹĒ½šŹōŌŖĖŲ
D.¶ĢÖÜĘŚŹĒÖøµŚŅ»”¢¶ž”¢Čż”¢ĖÄÖÜĘŚ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æÕÓĘųŹĒŅ»ÖÖÄÜŌ“£¬ĖüµÄÖ÷ŅŖ³É·ÖŹĒCH4£¬±ź×¼×“æöĻĀ£¬0.5 mol CH4ĶźČ«Č¼ÉÕÉś³ÉCO2ŗĶH2OŹ±£¬·Å³ö445 kJµÄČČĮ棬Ōņ±ķŹ¾CH4Č¼ÉÕČȵÄČČ»Æѧ·½³ĢŹ½ÕżČ·µÄŹĒ( )
A. ![]() CH4(g)£«O2(g)
CH4(g)£«O2(g) ![]() CO2(g)£«H2O(l) ¦¤H =445 kJ/mol
CO2(g)£«H2O(l) ¦¤H =445 kJ/mol
B. CH4£«2O2CO2£«2H2O ¦¤H=890 kJ/mol
C. CH4(g)£«2O2(g)CO2(g)£«2 H2O(g) ¦¤H =890 kJ/mol
D. CH4(g)£«2O2(g)CO2(g)£«2H2O(l) ¦¤H = -890 kJ/mol
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĻąĶ¬ÅØ¶ČµÄNaCl”¢MgCl2”¢AlCl3ČÜŅŗ£¬·Ö±šÓėAgNO3ČÜŅŗ·“Ó¦£¬µ±Éś³ÉµÄAgCl³ĮµķµÄÖŹĮæÖ®±ČĪŖ3£ŗ2£ŗ1Ź±£¬ČżÖÖČÜŅŗµÄĢå»ż±ČĪŖ £®
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æijŠĖȤŠ”×éÉč¼ĘČēĶ¼ĖłŹ¾×°ÖĆ½ųŠŠŌµē³ŲŌĄķµÄĢ½¾æ£¬ĻĀĮŠŠšŹö“ķĪóµÄŹĒ£Ø £©
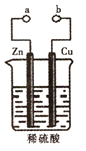
A. aŗĶbÓƵ¼ĻßĮ¬½ÓŹ±£¬µē×ÓÓÉZn¾µ¼ĻßĮ÷ĻņCu£¬ŌŁ¾Ļ”ĮņĖįĮ÷»ŲZn
B. aŗĶbÓƵ¼ĻßĮ¬½ÓŹ±ĶʬĪŖÕż¼«£¬·¢ÉśµÄ·“Ó¦Ź½ĪŖ£ŗ2H++2e-=H2”ü
C. aŗĶb²»Į¬½ÓŹ±£¬ŠæʬÉĻÓŠĘųĢåÉś³É£¬øĆ×°ÖĆ²»ÄÜŠĪ³ÉŌµē³Ų
D. ĪŽĀŪaŗĶbŹĒ·ńĮ¬½Ó£¬Ļ”ĮņĖį¾ł²ĪÓė·“Ó¦
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æÖ»ŅŖÓĆŅ»ÖÖŹŌ¼Į£¬æÉĒų±šNaCl”¢MgSO4”¢FeCl2”¢FeCl3ĖÄÖÖČÜŅŗ£¬ÕāÖÖŹŌ¼ĮŹĒ£Ø £©
A.HCl
B.NaOH
C.AgNO3
D.BaCl2
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ¼×Ķ¼±ķŹ¾µÄŹĒpH¶ŌÖ²ĪļŗĶČĖµÄµķ·ŪĆø»īŠŌµÄÓ°Ļģ£»ŅŅĶ¼±ķŹ¾µÄŹĒ3ÖÖĶŃĒāĆø(A”¢B”¢C)µÄ»īŠŌŹÜĪĀ¶ČÓ°ĻģµÄĒéæö”£ĻĀĮŠŠšŹöÕżČ·µÄŹĒ(””””)

A. “Ó¼×Ķ¼ÖŠæÉŅŌÖŖµĄpH=6Ź±Ö²Īļµķ·ŪĆøµÄ»īŠŌ×īøß
B. “ÓŅŅĶ¼ÖŠĪŽ·ØÖŖµĄĆøCµÄ×īŹŹĪĀ¶Č
C. “Ó¼×Ķ¼ÖŠæÉŅŌÖŖµĄČōĻø°ūÓÉĖįŠŌ±ä³É¼īŠŌŹ±,µķ·ŪĆøµÄ»īŠŌÖš½„Éżøß
D. “ÓŅŅĶ¼ÖŠæÉŅŌÖŖµĄĆø»īŠŌĪĀ¶Č·¶Ī§×ī¹ćµÄŹĒB
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ
A”¢B”¢D”¢E”¢FĪŖŌ×ÓŠņŹżŅĄ“ĪŌö“óµÄĒ°ĖÄÖÜĘŚŌŖĖŲ£¬ĘäÖŠAµÄ×īĶā²ćµē×ÓŹżŹĒĘäÄŚ²ćµē×ÓŹżµÄ2±¶£¬B”¢D”¢EĪŖĶ¬ÖÜĘŚŌŖĖŲ£¬BŌ×ÓµÄŗĖĶāµē×Ó×ÜŹżŹĒĘäĪ“³É¶Ōµē×ÓŹżµÄ5±¶£¬EŌ×Ó×īĶā²ćÓŠ1øöĪ“³É¶Ōµē×Ó£¬FŌ×ÓŗĖĶāÓŠ22ÖÖŌĖ¶ÆדĢ¬µÄµē×Ó”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)FŌŖĖŲĪ»ÓŚÖÜĘŚ±ķ___________Ēų£¬Ęä¼Ūµē×ÓÅŲ¼Ķ¼ĪŖ£ŗ___________”£
(2)B”¢D”¢EČżÖÖŌŖĖŲÖŠ£¬µŚŅ»µēĄėÄÜ×īŠ”µÄŹĒ_______________________ (ĢīŌŖĖŲ·ūŗÅ)£»Š“³öAD2µÄµČµē×ÓĢå___________ (·Ö×ÓŗĶŅõĄė×Óø÷Š“Ņ»ÖÖ)”£
(3)AO2ŗĶDO2ČŪµćøߵďĒ___________£¬ŌŅņŹĒ___________”£
(4)BµÄµ„ÖŹŗĶEµÄµ„ÖŹ·“Ó¦æÉŠĪ³ÉŌ×ÓøöŹż±ČĪŖ1”Ć3µÄ»ÆŗĻĪļM£¬MµÄĮ¢Ģå¹¹ŠĶĪŖ___________£¬ÖŠŠÄŌ×ÓµÄŌӻƹģµĄĄąŠĶĪŖ___________”£
(5)ŌŖĖŲFÓŠĮ½ÖÖĶ¬ĖŲŅģŠĪĢ壬³£ĪĀĻĀŹĒĮł·½¶Ń»ż£¬øßĪĀĻĀŹĒĢåŠÄĮ¢·½¶Ń»ż”£ČēĶ¼ĖłŹ¾ŹĒF¾§ĢåµÄŅ»ÖÖ¾§°ū£¬¾§°ū²ĪŹża=0.295nm£¬c=0.469nm£¬ŌņøĆF¾§ĢåµÄĆܶČĪŖ___________g”¤cm-3(ÓĆNA±ķŹ¾°¢·ü¼ÓµĀĀŽ³£ŹżµÄÖµ£¬ĮŠ³ö¼ĘĖćŹ½¼“æÉ)”£

²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com