
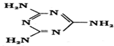
 �������е�ԭ�ӹ���ӻ�������sp2��sp3��l mol�����谷�����к�15mol�Ҽ���
�������е�ԭ�ӹ���ӻ�������sp2��sp3��l mol�����谷�����к�15mol�Ҽ���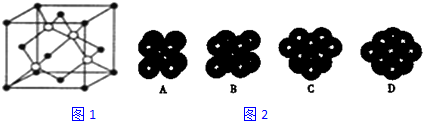
���� I����1�����ӽṹ���Գƣ�������ɵ����IJ��غϵķ���Ϊ���Է��ӣ����ṹ�Գ���������ɵ������غϵķ���Ϊ�Ǽ��Է��ӣ����ݼ۲���Ӷ����ж��ӻ���ʽ��
��2��ÿ8������46��H2O���ӹ��ɿ�ܣ���������6��CH4���Ӻ�2��H2O���ӣ����ж�ÿ8������48��H2O���Ӻ�6��CH4���ӣ��ݴ��ƶϿɱ�ʾ��ȼ����ƽ����ɵĻ�ѧʽ��
��1�������谷�����У������ϵ�Nԭ�ӳ�3���� ������һ���µ��Ӷԣ����ϵ�Nԭ�ӳ�2���� ������һ���µ��Ӷԣ��ӻ������=�� ����+�µ��Ӷ������ݴ�ȷ��Nԭ���ӻ���ʽ�����������谷�����к��Ц� �����㣻
��2��Ԫ��λ�ڵ�������VIII�壬���̬ԭ�ӵ�δ�ɶԵ��������̬̼ԭ�ӵ�δ�ɶԵ�������ͬ��Cԭ�ӵĵ����Ų�Ϊ1s22s22p2��δ�ɶԵ�����Ϊ2�����Ԫ��ΪNi��
��3��Ni�ļ۵�����Ϊ10��ÿ�������ṩһ�����Ӷԣ�����10+2n=18���㣻
��4����ͼ2��֪��̼ԭ��Ϊ���������ѻ���ΪABC�Ͷѻ���
��5�����ݾ�̯�����㾧����C��Siԭ����Ŀ���������㾧���к���C��Siԭ������������㾧��������������Ŀռ�������=$\frac{������C��Siԭ�������}{�������}$��100%��
��� �⣺I����1��ˮ���ӵĿռ乹��ΪV�ͽṹ��������ɵ����IJ��غϣ����ڼ��Է��ӣ�
����Ŀռ乹��Ϊ�������壬�ṹ�Գƣ�������ɵ������غϣ����ڷǼ��Է��ӣ�
ˮ����������ԭ��O�ļ۲���Ӷ���=2+$\frac{6-1��2}{2}$=4������ԭ��Ϊsp3�ӻ���
����������ԭ��C�۲���Ӷ���=4+$\frac{4-1��4}{2}$=4����̼ԭ��Ϊsp3�ӻ���
��ѡ��CE��
��2��������ÿ8����ֻ��6��������CH4���ӣ�����2����������H2O������䣬���6��������6��������ӣ�ˮ����46+2=48����48��H2O������Ȼ��ˮ����ƽ��������ɿɱ�ʾΪCH4•8H2O��6CH4•48H2O��
�ʴ�Ϊ��CH4•8H2O��6CH4•48H2O��
��1�������谷�����У������ϵ�Nԭ�Ӻ���3�� �� ����һ���µ��Ӷԣ����Բ�ȡsp3�ӻ������ϵ�Nԭ�Ӻ���2�� �� ����һ���µ��Ӷԣ����Բ�ȡsp2�ӻ���һ�������谷�����к���15���� ��������1mol�����谷������ �� ��Ϊ15mol���ʴ�Ϊ��sp2��sp3��15��
��2��Ԫ��λ�ڵ�������VIII�壬���̬ԭ�ӵ�δ�ɶԵ��������̬̼ԭ�ӵ�δ�ɶԵ�������ͬ��Cԭ�ӵĵ����Ų�Ϊ1s22s22p2��δ�ɶԵ�����Ϊ2�����Ԫ��ΪNi�����̬ԭ�ӵ�M������Ų�ʽΪ3s23p63d8��
�ʴ�Ϊ��3s23p63d8��
��3��Ni�ļ۵�����Ϊ10��ÿ�������ṩһ�����Ӷԣ���10+2n=18����n=4���ʴ�Ϊ��4��
��4����ͼ2��֪��̼ԭ��Ϊ���������ѻ���ΪABC�Ͷѻ���ѡ��D���ϣ��ʴ�Ϊ��D��
��5�����ݾ�̯�����㾧����Cԭ����Ŀ=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4��Siԭ����Ŀ=4�������߳�Ϊa cm�������Ϊa3cm3��̼ԭ��ֱ��Ϊb cm������Cԭ�������=4��$\frac{4}{3}$���С���$\frac{1}{2}b$��3cm3=$\frac{2}{3}$��b3cm3����ԭ��ֱ��Ϊc cm������Siԭ�������=4��$\frac{4}{3}$���С���$\frac{1}{2}c$��3cm3=$\frac{2}{3}$��c3cm3���ʾ�����C��Siԭ�������=$\frac{2}{3}$��b3cm3+$\frac{2}{3}$��b3cm3=$\frac{2}{3}$�У�b3+c3��cm3���ʾ����Ŀռ�������=$\frac{\frac{2}{3}�У�{b}^{3}+{c}^{3}��}{{a}^{3}}$��100%=$\frac{2�У�{b}^{3}+{c}^{3}��}{3{a}^{3}}$��100%��
�ʴ�Ϊ��$\frac{2�У�{b}^{3}+{c}^{3}��}{3{a}^{3}}$��100%��
���� �����Ƕ����ʽṹ�Ŀ��飬�漰���Ӽ��ԡ��ӻ�������������㡢��̬�뼤��̬����������Ų�����ѧ������֪ʶ�㣬�ѵ��Ǿ������㣬�ؼ�����ȷ����������������ӣ���Ҫѧ������һ���Ŀռ���������ѧ������������Ŀ�Ѷ��еȣ�


 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ��ͼΪ�ס����������ʵ��ܽ������ͼ�������ͼ�ش��������⣮
��ͼΪ�ס����������ʵ��ܽ������ͼ�������ͼ�ش��������⣮�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �����¶ȣ�����Ӧ�������ӣ�����Ӧ���ʼ�С | |
| B�� | �����¶������ڷ�Ӧ�������ӣ��Ӷ����̴ﵽƽ���ʱ�� | |
| C�� | ����Ӧ���ܱ���ϵ�ڽ��У�ͨ��He������ϵѹǿ����Ӧ�������� | |
| D�� | ����Ӧ�ں�ѹ��ϵ�ڽ��У�ͨ��He������ϵѹǿ���䣬��Ӧ���ʲ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ɫ����������Һ��MnO4-��K+��C1-��SO42- | |
| B�� | ʹ��̪������Һ��K+��Na+��NO3-��Cl- | |
| C�� | �μ�KSCN�Ժ�ɫ����Һ��NH4+��K+��Cl-��I- | |
| D�� | 0.1 mol•L-1NaHCO3��Һ��Na+��Ba2+��NO3-��OH- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| 1 | 2 | 3 | 4 | 5 | 6 | �� |
| �� | C2H6 | C5H12 | C8H18 | �� | �� | �� |
| A�� | C16H34 | B�� | C17H36 | C�� | C26H54 | D�� | C27H56 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Aԭ�ӵ�������������Bԭ�ӵ������������� | |
| B�� | Aԭ�ӵĵ��Ӳ�����Bԭ�ӵĵ��Ӳ����� | |
| C�� | 1mol A��ϡ�������û���������1mol B�û��������� | |
| D�� | �����£�A�ܴ���ˮ���û�����������B���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 3 | B�� | 4 | C�� | 5 | D�� | 6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
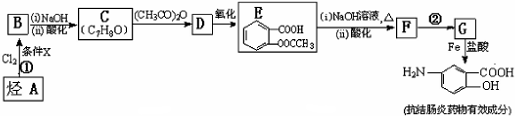
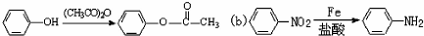
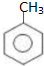 ��
��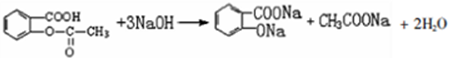 ��
��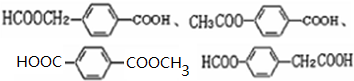 ��
��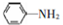 �ױ��������������������ʱ������һ��ȡ��������ȡ����������ڶ�λ�����������������Ȼ�ʱ��ȡ���ڼ�λ���ݴ�д����AΪԭ�Ϻϳɻ�����
�ױ��������������������ʱ������һ��ȡ��������ȡ����������ڶ�λ�����������������Ȼ�ʱ��ȡ���ڼ�λ���ݴ�д����AΪԭ�Ϻϳɻ�����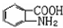 �ĺϳ�·��
�ĺϳ�·��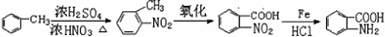 �������������п��᳦��ҩ����Ч�ɷֵĺϳ�·�ߵı��﷽ʽ���⣩
�������������п��᳦��ҩ����Ч�ɷֵĺϳ�·�ߵı��﷽ʽ���⣩�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com