
| 50mL���� | 50mL���� | 50mL���� | |
| m������ | 9.2g | 15.7g | 27.6g |
| V��CO2������״���� | 2.24L | 3.36L | 3.36L |
| A�� | 15.7 g�����ǡ����������ȫ��Ӧ | |
| B�� | NaHCO3ռ���������ʵ�������Ϊ50% | |
| C�� | ��������ʵ���Ũ��Ϊ3.0 mol/L | |
| D�� | ��������9.2 gʱ������� |
���� ����ͬ�������ڵڶ���ʵ������������ڵ�һ��ʵ�飬˵����������9.2 gʱ����������ڶ��������������������ȣ�˵���ڶ���������ȫ��Ӧ������3.36L������̼��Ҫ����������Ϊ$\frac{3.36L}{22.4L/mol}$��9.2g=13.8g��15.7g���ʵڶ������������ʣ�ࣻ
��һ��ʵ���л������ȫ��Ӧ�����������������ӦH++HCO3-�TCO2��+H2O�����ɶ�����̼�����ʵ���Ϊ$\frac{2.24L}{22.4L/mol}$=0.1mol����9.2 g�������NaHCO3��KHCO3�����ʵ����ֱ�Ϊxmol��ymol������̼Ԫ���غ��֪n��CO2��=n��NaHCO3��+n��KHCO3���������û���������з��̼���x��y��ֵ����������������NaHCO3������������
�ڶ���ʵ�����β��㣬�������ʣ�࣬�ڶ���ʵ���з�����ӦH++HCO3-�TCO2��+H2O�����ɶ�����̼�����ʵ���Ϊ$\frac{3.36L}{22.4L/mol}$=0.15mol���ɷ���ʽ��֪n��HCl��=n��CO2�����ݴ˼�����������ʵ���Ũ�ȣ�
��� �⣺A������ͬ�������ڵڶ���ʵ������������ڵ�һ��ʵ�飬˵����������9.2 gʱ����������ڶ��������������������ȣ�˵���ڶ���������ȫ��Ӧ������3.36L������̼��Ҫ����������Ϊ$\frac{3.36L}{22.4L/mol}$��9.2g=13.8g��15.7g���ʵڶ������������ʣ�࣬��A����
B��D����һ��ʵ���л������ȫ��Ӧ�����������������ӦH++HCO3-�TCO2��+H2O�����ɶ�����̼�����ʵ���Ϊ$\frac{2.24L}{22.4L/mol}$=0.1mol��
��9.2 g�������NaHCO3��KHCO3�����ʵ����ֱ�Ϊxmol��ymol������̼Ԫ���غ��֪��x+y=0.1mol��
���������Ϊ9.2g������84x+100y=9.2g���������̽�ã�x=0.05 mol��y=0.05 mol����������̼�����Ƶ����ʵ�������Ϊ��$\frac{0.05mol}{0.05mol+0.05mol}$��100%=50%����B��ȷ��
C���ڶ���ʵ�����β��㣬�������ʣ�࣬�ڶ���ʵ���з�����ӦH++HCO3-�TCO2��+H2O�����ɶ�����̼�����ʵ���Ϊ$\frac{3.36L}{22.4L/mol}$=0.15mol���ɷ���ʽ��֪n��HCl��=n��CO2��=0.15mol����������ʵ���Ũ��Ϊ$\frac{0.15mol}{0.05L}$=3mol/L����C��ȷ��
D������ͬ�������ڵڶ���ʵ������������ڵ�һ��ʵ�飬˵����������9.2 gʱ�����������D��ȷ��
��ѡA��
���� ���⿼������ļ��㣬��Ŀ�Ѷ��еȣ��ؼ��DZȽ��������ݣ����ݶ�����̼������仯����������ж��Ƿ���ȫ��Ӧ������ѧ�������ݵķ�������������˼ά�������飮



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ������Ϊ17��������Ϊ20���ȣ�Cl��ԭ�ӣ�${\;}_{17}^{20}$Cl | |
| B�� | CO2����ʽ��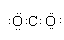 | |
| C�� | þ���ӵĽṹʾ��ͼ�� | |
| D�� | CCl4���ӵı���ģ�ͣ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ˮ����ͭ��SO2������ | B�� | NH3��Cu������ | ||
| C�� | ���ᡢ���ᡢ���� | D�� | ��ˮ��ʳ�Ρ��ƾ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 1 | B�� | 2 | C�� | 3 | D�� | 4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ���� | K+ | Na+ | NH4+ | SO42- | NO3- | Cl- |
| Ũ��/mol��L-1 | 4��10-5 | 6��10-5 | 2��10-5 | 4��10-5 | 3��10-5 | 2��10-5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| Ԫ�� | ���ֽṹ֪ʶ | �������� |
| X | X�ĵ�����˫ԭ�ӷ��ӹ��ɣ���������14������ | X�ж����������XO��XO2��X2O4�ȣ�ͨ�������XO2��X2O4���� |
| Y | Yԭ�ӵĴ�������������������������һ�� | Y���γɶ�����̬�⻯�� |
| Z | Zԭ�ӵ���������������4 | ZԪ�ص���������ϼ���������ϼ۴����͵���6 |
| W | Wԭ�ӵ���������������2n-3��nΪԭ�Ӻ�����Ӳ����� | ��ѧ��Ӧ��Wԭ����ʧȥ���������γ�Wn+ |
 ��Z�ĵ�����W�ĵ���һ�������¿ɷ�����Ӧ���ɻ�����A��A��������ѧ������Ϊ���ۼ���
��Z�ĵ�����W�ĵ���һ�������¿ɷ�����Ӧ���ɻ�����A��A��������ѧ������Ϊ���ۼ����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | NO2ֻ��ԭ������ | B�� | NO2��������� | ||
| C�� | NO2ֻ������������ | D�� | O3��O2��Ϊͬ���칹�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | H2 | B�� | O2 | C�� | CO | D�� | CH4 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com