
һ������������ȼ�ϵ�ؾ��и߷���Ч�ʶ��������ӡ�����Li2CO3��Na2CO3�������λ����������ʣ�һ��ͨCO���壬��һ��ͨO2��CO2�Ļ�����壬����650��ʱ������ȼ�ϵ�أ������ܷ�Ӧ��2CO+O2=2CO2��������˵������ȷ���� �� ��
A.ͨCO��һ���ǵ�ص�����
B.�����缫��Ӧ�ǣ�O2+2CO2+4e�D=2CO32�D
C.��������CO32�D�����ʵ����ڹ���ʱ���ֲ���
D.��������������Ӧ
 С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д� ��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
��У��ʦ������ҵ���Ӻ����Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��X��Y��Z�Ƕ�����Ԫ�ص����ֳ��������X��ˮ��Ӧ������һ�־��л�ԭ�ԵIJ��ȶ��Ķ�Ԫ�ᣬ����Ļ�ѧʽ��
��X��Y��Z�Ƕ�����Ԫ�ص����ֳ��������X��ˮ��Ӧ������һ�־��л�ԭ�ԵIJ��ȶ��Ķ�Ԫ�ᣬ����Ļ�ѧʽ��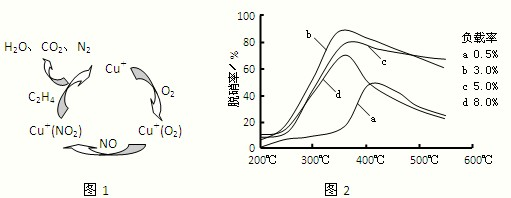
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| n |
| 3 |
| n |
| 3 |
| V/s | 0 | 50 | 100 |
| c��N2O3��/mol?L-1 | 5.00 | 3.52 | 2.48 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
��16�֣�����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
��1����֪��N2(g)+O2(g)=2NO(g) ��H=+180.5kJ/mol
N2(g)+3H2(g) 2NH3(g) ��H=��92.4kJ/mol
2H2(g)+O2(g)=2H2O(g) ��H=��483.6kJ/mo
����17g��������������ȫ����һ�����������ˮ�������ų�������Ϊ ��
��2��ij����С���о����������������������£��ı���ʼ�����������ʵ�����N2(g)+3H2(g)
2NH3(g)��Ӧ��Ӱ�졣ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ�����
��ͼ����T2��T1�Ĺ�ϵ�ǣ�T2 T2������ڡ������ڡ������ڡ�����ȷ������
�ڱȽ���a��b��c����������ƽ��״̬�У���Ӧ��N2��ת������ߵ��� ������ĸ����
������ʼ��ϵ�м���N2�����ʵ���Ϊ molʱ����Ӧ�İٷֺ�������������ݻ�ΪH��n=3mol��Ӧ�ﵽƽ��ʱH2��ת����Ϊ60%����������£�T2������Ӧ��ƽ�ⳣ��K= ��
��3��N2O3��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע��
��һ���¶��£��ں����ܱ�������N2O3�ɷ������з�Ӧ��
2N2O34NO2(g)+O2 ��H>0�±�Ϊ��Ӧ��T1�¶��µIJ���ʵ������
| Vs | 0 | 500 | 1000 |
| c(N2O3)/mol��L��1 | 5.00 | 3.52 | 2.48 |
��500s��NO2��ƽ����������Ϊ .
������H2��O2��������Na2CO3��ɵ�ȼ�ϵ�أ����õ�ⷨ�Ʊ�N2O3��װ����ͼ��ʾ������YΪCO2��
д��ʯīI�缫�Ϸ�����Ӧ�ĵ缫��Ӧʽ ��
�ڵ���������N2O3�ĵ缫��ӦʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011���㽭ʡ��У����2�����������ۣ���ѧ���� ���ͣ������
��.��˹�����������Ϳ�ѧ�о����к���Ҫ�����塣��Щ��Ӧ�ķ�Ӧ����Ȼ��ֱ�Ӳ�ã�����ͨ����ӵķ����ⶨ���ָ������е�5����Ӧ(�ɰ�����HCl��ˮ�Ʊ�NH4C1ˮ��Һ)�����жϷ�Ӧ�ܵķ�Ӧ�ȣ���H�� ��
�� NH3(g) + HCl(g) = NH4Cl(s) ��H=��176kJ��mol�C1
�� NH3(g) + H2O(l) = NH3(aq) ��H=��35.1 kJ��mol�C1
�� HCl(g) + H2O(l) = H Cl(aq) ��H=��72.3 kJ��mol�C1
Cl(aq) ��H=��72.3 kJ��mol�C1
�� NH4C1(s) + H2O(1) = NH4C1(aq)
�� NH3(aq) + HCl(aq) = NH4C1(aq) ��H= ��52.3 kJ��mol�C1
HCl(aq) = NH4C1(aq) ��H= ��52.3 kJ��mol�C1
��.N2O5��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע������H2��O2��������Na2CO3��ɵ�ȼ�ϵ�أ����õ�ⷨ�Ʊ�N2O5��װ����ͼ��ʾ������YΪCO2��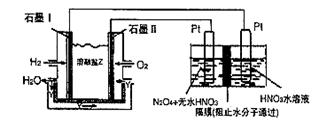
д��ʯīI�缫�Ϸ�����Ӧ�ĵ缫��Ӧʽ ��
�ڵ���������N2O3�ĵ缫��ӦʽΪ ��
��.����������SO2����������������NOx���������ǻ�����ѧ�о����ȵ㡣
���������������Ļ��������� ��
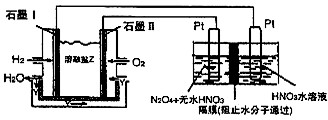
(2)Ŀǰ����ѧ�������о�һ������ϩ��Ϊ��ԭ����������NO��ԭ��������������ʾ��ͼ����ͼ1�����������¶ȡ������ʣ�����ɸ�д����������������Ĺ�ϵ��ͼ2��ʾ��
��д��������ԭ���ܷ�Ӧ�Ļ�ѧ����ʽ�� ����Ϊ�ﵽ�������Ч����Ӧ��ȡ�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��������ʯ��ɽ��2010�����һģ�������ۺϣ���ѧ���� ���ͣ������
��16�֣�����N2��H2����ʵ��NH3�Ĺ�ҵ�ϳɣ������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
��1����֪��N2(g)+O2(g)=2NO(g) ��H=+180.5kJ/mol
N2(g)+3H2(g)  2NH3(g) ��H=��92.4kJ/mol
2NH3(g) ��H=��92.4kJ/mol
2H2(g)+O2(g)=2H2O(g) ��H=��483.6kJ/mo
����17g��������������ȫ����һ�����������ˮ�������ų�������Ϊ ��
��2��ij����С���о����������������������£��ı���ʼ�����������ʵ�����N2(g)+3H2(g) 
2NH3(g)��Ӧ��Ӱ�졣ʵ������ͼ��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ�����

��ͼ����T2��T1�Ĺ�ϵ�ǣ�T2 T2������ڡ������ڡ������ڡ�����ȷ������
�ڱȽ���a��b��c����������ƽ��״̬�У���Ӧ��N2��ת������ߵ��� ������ĸ����
������ʼ��ϵ�м���N2�����ʵ���Ϊ molʱ����Ӧ�İٷֺ�������������ݻ�ΪH��n=3mol��Ӧ�ﵽƽ��ʱH2��ת����Ϊ60%����������£�T2������Ӧ��ƽ�ⳣ��K= ��
��3��N2O3��һ�������������������ʺ��Ʊ��ܵ����ǵĹ�ע��
��һ���¶��£��ں����ܱ�������N2O3�ɷ������з�Ӧ��
2N2O3 4NO2(g)+O2 ��H>0�±�Ϊ��Ӧ��T1�¶��µIJ���ʵ������
4NO2(g)+O2 ��H>0�±�Ϊ��Ӧ��T1�¶��µIJ���ʵ������
|
Vs |
0 |
500 |
1000 |
|
c(N2O3)/mol��L��1 |
5.00 |
3.52 |
2.48 |
��500s��NO2��ƽ����������Ϊ .
������H2��O2��������Na2CO3��ɵ�ȼ�ϵ�أ����õ�ⷨ�Ʊ�N2O3��װ����ͼ��ʾ������YΪCO2��

д��ʯīI�缫�Ϸ�����Ӧ�ĵ缫��Ӧʽ ��
�ڵ���������N2O3�ĵ缫��ӦʽΪ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com