
��12�֣���ҵ����������(��Ҫ�ɷ�ΪAl2O3��Fe2O3��)��ȡAl2O3��ұ������ԭ�ϣ������ε�ⷨ��õĴ����к�һ�����Ľ����ƺ���������Щ���ʿɲ��ô�����������ȥ��������β��������������ڸֲĶ�����������������ͼ��ʾ��
(��֪��NaCl�۵�Ϊ801�棻AlCl3��181������)
��1������Һ��ͨ�����CO2��������Ӧ�����ӷ���ʽΪ ��
��2������ǰ������������������������ʯӢɰ����ֹ����ʱ���Ƿֱ����������û���Ӧ�����µ����ʣ���������������Ӧ�Ļ�ѧ����ʽΪ ��
��3����Cl2����ͨ�������еĴ������壬�����������ϸ�����ȥ�����ݵ���Ҫ�ɷֳ�Cl2�����______����̬����ճ���������ϣ�����������γɸ����������п϶�����________��
��4�����������У�������Ϊ�����������ε��Һ����Ԫ����Ҫ��AlCl4����ʽ���ڣ��������ĵ缫��ӦʽΪ_____________��
��5���ֲĶ�������ʴ���ܻ�����ǿ����ԭ����_____________��
��12�֣�
��1�� AlO2�� ��2H2O ��CO2��Al(OH)3����HCO3����2�֣�
��2��Fe2O3��2Al Al2O3��2Fe��2�֣�
Al2O3��2Fe��2�֣�
��3��HCl��AlCl3����2�֣� NaCl��2�֣�
��4��Al-3e-��4Cl-= AlCl4-��2�֣�
��5�������γɵ�����������Ĥ�ܷ�ֹ�ֲĸ�ʴ�������ܵ�������Ĥ�������еĵ������Һ���ڲ�������� ��2�֣�
���������������1����Һ�к���AlO2�� ��ͨ��CO2������Al(OH)3���������ӷ���ʽΪ��AlO2�� ��2H2O ��CO2��Al(OH)3����HCO3��
��2�����ڸ��������£���Fe2O3��ԭΪFe����ѧ����ʽΪ��Fe2O3��2Al Al2O3��2Fe
Al2O3��2Fe
��3����Ϊ�����к���������������Cl2��Ӧ����AlCl3��H2��AlCl3��181���������������ݵ���Ҫ�ɷֳ�Cl2�����HCl��AlCl3�������к���Na����Cl2��Ӧ����NaCl��NaCl�۵�Ϊ801�棬���Ը����п϶�����NaCl��
��4��Alʧ��������AlCl4�����缫����ʽΪ��Al-3e-��4Cl-= AlCl4-
��5��Al�ڿ�������O2��Ӧ����Al2O3�������γɵ�����������Ĥ���ܷ�ֹ�ֲĸ�ʴ��
���㣺���⿼�黯ѧ�������̵ķ���������ʽ����д�����ԭ����


 ��ʿһ��ȫͨϵ�д�
��ʿһ��ȫͨϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����仯��������Ҫ��;����ۺ�������[Fe2(OH)n��SO4)3-n/2]m��һ������Ч��ˮ��������������������أ��������Ļ��ϼ�Ϊ+6����һ����Ҫ��ɱ����������ij��ѧ̽��С��������·����Ʊ��������ֲ�Ʒ��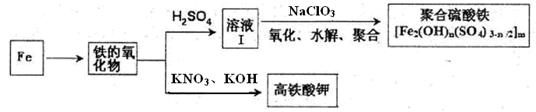
��ش��������⣺
��1������������������������Ļ��ϼۣ�Ӧʹ�õ��Լ��� �����ţ�
| A��ϡ���� | B��ϡ���� | C��KSCN��Һ | D�����Ը��������Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��֪A��G����ͼ��ʾ��ת����ϵ����������������ȥ��������A��GΪ���ʣ�D����ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����壬E��F������NaOH��Һ��Ӧ��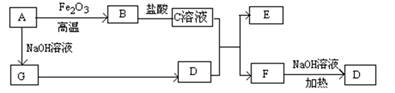
��ش��������⣺
��1��д��F�ĵ���ʽ ��
��2����C��Һ��D��Ӧ�����ӷ���ʽΪ ��
��F��Һ��NaOH��???���ȷ�Ӧ�Ļ�ѧ����ʽΪ ��
��3�����������ӷ���ʽ����C��ҺΪ�������� ��
��F��Һ������Ũ���ɴ�С��˳��Ϊ ��
��4����5.4gAͶ��200mL 2.0mol/Lij��Һ����G���ʲ������ҳ�ַ�Ӧ���н���ʣ�࣬�����Һ������ ������ţ�
A��HNO3��Һ B��H2SO4��Һ C��NaOH��Һ D��HCl��Һ
��5����1molN2��3molG�����������ݻ�Ϊ2L��ij�ܱ������н��з�Ӧ����֪�÷�ӦΪ���ȷ�Ӧ��ƽ��ʱ�����D�����ʵ���Ũ��Ϊa mol/L��
�������Ӧ����v(G)��1.2mol/(L��min)����v(D)�� mol/(L��min)
���������������������£�����ʼʱ����0.5molN2��1.5molG�ﵽƽ���D�����ʵ���Ũ�� ������ڡ�����С�ڡ����ڡ���a/2 mol/L��
�۸������µ�ƽ�ⳣ��Ϊ ���ú�a�Ĵ���ʽ��ʾ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
�ܵ����ʵ���Ϊ0.50 mol��Fe����Al�ۻ�����Ϊ�����������ݣ���һ���м�������
��ϡ���ᣬ�ڱ�״���²�������a L������һ���м�������������������Һ���ڱ�״���²�������b L����a+b����ֵ��������
| A��5.6 | B��7.3 | C��8.2 | D��11.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��70 g�������ƺ������ƵĻ�����98 gˮ��ַ�Ӧ����������������Һ����������Ϊ50%��
��1����ԭ������й������ƺ������Ƶ���������2���������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijС��ͨ��ʵ���о�Na2O2��ˮ�ķ�Ӧ?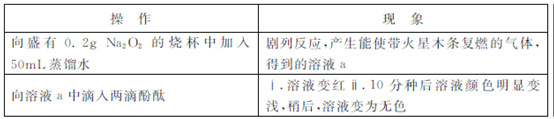
��1��Na2O2��ˮ��Ӧ�Ļ�ѧ����ʽ�� ?
��2��������Һ��ɫ��������Һa�д��ڽ϶��H2O2,H2O2���̪�����˷�Ӧ?
��ͬѧͨ��ʵ��֤ʵ��H2O2�Ĵ���:ȡ������Һa,�����Լ� (�ѧʽ)�����������?
����ͬѧ�������ϻ�Ϥ:��KMnO4(����ԭΪ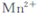 )���ԲⶨH2O2�ĺ���?
)���ԲⶨH2O2�ĺ���?
ȡ3mL��Һaϡ����15mL,��ϡH2SO4�ữ,����μ���0��0045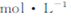 KMnO4��Һ,��������,��Һ��ɫ���ʿ�ʼ��������,���յ�ʱ������10mL KMnO4��Һ?
KMnO4��Һ,��������,��Һ��ɫ���ʿ�ʼ��������,���յ�ʱ������10mL KMnO4��Һ?
��KMnO4��H2O2��Ӧ�����ӷ���ʽ�� ?
����Һa�� ?
?
����Һ��ɫ���ʿ�ʼ���������ԭ������� ?
��3��Ϊ̽����������ԭ��,ͬѧ�Ǽ�������������ʵ��:
����H2O2��Һ�е������η�̪,��,����5��0��1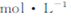 NaOH��Һ,��Һ�����Ѹ�ٱ���ɫ�Ҳ�������,10���Ӻ���Һ����ɫ,�ù�����������ЧӦ?
NaOH��Һ,��Һ�����Ѹ�ٱ���ɫ�Ҳ�������,10���Ӻ���Һ����ɫ,�ù�����������ЧӦ?
����0��1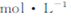 NaOH��Һ�е������η�̪��,��,��Һ���,10���Ӻ���Һ��ɫ�����Ա仯;�����Һ��ͨ������,��Һ��ɫ�����Ա仯?��ʵ���͢��У��ɵó��Ľ����� ?
NaOH��Һ�е������η�̪��,��,��Һ���,10���Ӻ���Һ��ɫ�����Ա仯;�����Һ��ͨ������,��Һ��ɫ�����Ա仯?��ʵ���͢��У��ɵó��Ľ����� ?
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijѧ����ʵ���ҳ������ᡢ��κͽ�������Ϊ��Ӧ�������һ���ײ���С���Թܺ�һ�����ƿ��װ����ͼ��ʾ��װ�á��Իش�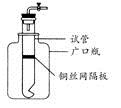
(1)���Թ���װ��ͭ˿�����壬���ø�װ�ÿ���ȡ��Щ���壿
(�����)��
(2)����ͭ˿�������Ϊ��˿�����壬���װ�ÿ�������ȡ�������壿
��
�÷�Ӧ�����ӷ���ʽ�ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ҹ�����ר�Һ�°����ڴ��£��Ľ��������ˡ������Ƽ����Ϊ�����Ƽҵ������ͻ�����ס�������������⣺
��1���������Ƽ���Ƶõġ���� (�ѧʽ)��
��2������������Ƽ��������Ҫ�Ĺ�ҵ�Ƽ�����б����У�����ȷ���� ��
| | | ��� | �����Ƽ |
| A | ԭ�� | ʳ�Ρ���������ʯ�� | ʳ�Ρ�������������̼ |
| B | ���ܵĸ����� | �Ȼ��� | �Ȼ�� |
| C | ѭ������ | ������������̼ | �Ȼ��� |
| D | ���� | ԭ���ã��豸���ӣ��ܺĸ� | ԭ�������ʸߣ��������� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼʵ��װ��������֤ijЩ���ʵ����ʡ����Թ�A��װ�������Ĺ���NaHCO3,DΪ�̶������ӲֽƬ���Իش��������⣺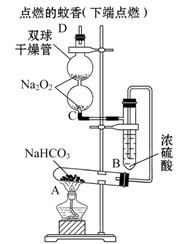
��1����ʵ���ʵ��Ŀ����__________________________________________��
��2����A�Թ��ڷ�����Ӧ�Ļ�ѧ����ʽ��_______________________________��
��3��Bװ�õ�������_________________________________________________��
��4����˫�������ڷ�����Ӧ�Ļ�ѧ����ʽΪ____________________________��
��5��ʵ��ʱ�۲쵽��ʵ��������_______________________________________��
����ʵ������˵��___________________________________________________��
��6������������ڵ�Na2O2����Na2O����ʵ��ʱ�۲쵽��ʵ��������___________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com