

| A���ô��п̶ȵ��ձ����Ƴ�100 mL������Һ�� | B������Ͳ��ȡ25.00 mL������Һ�� | C����������ƿ�С� | D��������ˮϴ�ӵζ��ܺ�װ��KMnO4����Һ���øñ���Һ�ζ�����������(E)����Һ��ɵ��Ϻ�ɫʱ��ֹͣ�ζ�����30���ڲ���ɫ��(F)��ȡ������ζ��������ĵ�KMnO4����Һ��������������е�FeԪ�غ�������ָ����ʵ������д����������ı��________________________�� |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

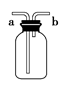

| A��ͼ������1mol/LNaCl��Һ���ݲ��� | B��ͼ��a�ڽ������ռ�Cl2��CO2������ |
| C��ͼ��̽��NaHCO3�����ȶ��� | D��ͼ�������ᾧ����Ȼ��ƾ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������ʱ��Ӧʹ������е�ˮ����ȫ���ɺ���ֹͣ���� |
| B��ij�����ڼ���ϡ�����������ɫ��ζ����ʹ����ʯ��ˮ�������壬֤���ù���һ������CO32- |
| C��ȡ��Na2CO3��Һʱ������ȡ�����࣬Ϊ�˲��˷ѣ��ְѹ������Լ������Լ�ƿ�� |
| D����Ũ��������һ�����ʵ���Ũ�ȵ�ϡ����ʱ����Ũ��������ˮ��Ӧ��ȴ�����²���ת�Ƶ�����ƿ�С� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

A����0.1mol��L��1 Na2CO3��Һ�У������η�̪��dz��ɫ���Ⱥ��ɫ���˵������ˮ�ⷴӦ�����ȷ�Ӧ | B���ò�˿պȡ����ij��Һ������ɫ��Ӧ������ɫ�ܲ����۲쵽�������ɫ������Һһ���Ǽ�����Һ |
| C����ijFeCl2��Һ�м���KSCN��Һ���۲쵽��Һ��Ѫ��ɫ��˵������Һ��FeCl2��ȫ�������� | |
| D����ij��Һ�м���ϡ���ᣬ����������ͨ�����ʯ��ˮ��ʯ��ˮ����ǣ�����Һһ������CO32�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ��� | V (Na2CO3)/mL | ������� | | ��� | ��Ӧ�¶�/�� | ������� |
| 1 | 2.8 | �ࡢ��ɫ | | 1 | 40 | �ࡢ��ɫ |
| 2 | 2.4 | �ࡢ��ɫ | | 2 | 60 | �١�dz��ɫ |
| 3 | 2.0 | �϶ࡢ��ɫ | | 3 | 75 | �϶ࡢ��ɫ |
| 4 | 1.6 | ���١���ɫ | | 4 | 80 | �϶ࡢ��ɫ(������ɫ) |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ˮ���հ���ʱ�ĵ����� |
| B��ʯ�ͷ���ʱ�����¶����õ��¶�֭ |
| C�����Ҵ��Ʊ���ϩʱ���õ��¶ȼ� |
| D����NaOH��Һ��Fe2(SO4)3��Һ�Ʊ�Fe(OH)3��ʱ������NaOH��Һ�Ľ�ͷ�ι� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

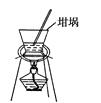
| A������ǰû�н�����ƿ�е�ˮ������ |
| B��̼����ʧȥ�˲��ֽᾧˮ�� |
| C��̼���ƾ��岻�������л����Ȼ��ƣ� |
| D������̼���ƾ���ʱ�����������⣻ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����


�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com